Petfood: how to prevent insect infestation
Insect contamination of petfoods is a potential problem in tropical countries; this paper offers an overview of the situation and how the risks can be minimized
Issue number 33.2 Other Scientific
Published 06/09/2023
Also available in Français , Deutsch , Italiano , Português , Español and ภาษาไทย
The “Shrinking Kitty” is a common presentation in the small animal clinic; this article offers a logical approach to the cat with unexplained weight loss.
© Shutterstock

Unexplained weight loss should be investigated promptly, as it usually indicates a significant change in the cat’s health.
Subtle changes in the routine laboratory data may hold key clues regarding the cause of unexplained weight loss.
Gastrointestinal disease is a common cause of weight loss in cats but may not be accompanied by overt signs of digestive tract dysfunction such as vomiting or diarrhea.
The cat’s nutritional needs must be evaluated and addressed pending determination of the underlying cause of weight loss.
A cat with unexplained weight loss is a familiar scenario within the small animal consult room, and such cases can be both challenging and frustrating for the clinician. This article will outline a logical approach to the situation where no obvious diagnosis is apparent, with an emphasis on a cost-conscious, stepwise approach; a typical scenario is shown in Box 1.
Box 1. The cat with unexplained weight loss – a typical presentation.
PresentationIt is 10 am on a busy Monday morning, and Mrs. Smith arrives at the clinic with her 8-year-old, male (castrated) cat, Freddie, for his annual wellness visit. Freddie’s physical examination is unremarkable except for grade 2 periodontal disease. Findings on routine lab work* are within reference ranges, but he has lost 0.5 kg since his last visit. *Complete blood count; serum biochemical profile; urinalysis with sediment examination; fecal flotation; total thyroxine concentration; FeLV and FIV testing Follow-up on FreddieOn further questioning, it was established that Freddie was maintained on a dry, senior diet, offered ad libitum. The owner was unaware of any changes in intake, but was unable to specify the amount consumed. No vomiting or changes in stool consistency had been noted. However, the cat’s serum albumin had decreased from 3.6 g/dL a year ago to 3.1 g/dL (reference interval: 2.8-3.6 g/dL). Due to concerns about underlying GI disease, serum folate, cobalamin and PLI* were measured. All values were within the reference interval, but cobalamin was judged to be questionable at 388 µg/dL (normal serum cobalamin levels will vary depending on the reference laboratory used). Findings on abdominal ultrasonography were unremarkable; the overall appearance and thickness of the small intestinal wall were normal. After a thorough discussion with the owner, the decision was made to anesthetize Freddie for dental prophylaxis with intraoral radiographs, along with upper and lower GI endoscopic examination and biopsy of the stomach, duodenum, ileum, and colon. The dental procedure confirmed and addressed mild periodontal disease. Histopathology of the stomach and duodenum identified a mild lymphoplasmacytic infiltrate; a histiocytic infiltrate with intralesional yeast (appearance consistent with Histoplasma capsulatum) was reported in the ileal and colonic biopsies. The diagnosis of histoplasmosis was subsequently confirmed with an enzymatic immunoassay performed on a urine sample, and Freddie was treated successfully with itraconazole. *pancreas-specific lipase immunoreactivity |
Owners can become accustomed to abnormal behaviors and may discount the significance of clinical signs such as vomiting. It is not unusual for an owner to say “Fluffy seems fine” when talking about a cat that vomits on a weekly basis. Similarly, an owner may fail to recognize the importance of a subtle change in stool consistency or an increase in urine output. Occasional coughing may be attributed to hairballs and not reported to the veterinary care team. A careful review of the patient’s history, using open-ended questions to encourage more detailed responses, may highlight issues that could explain the weight loss and merit further investigation.
It can often be very useful to repeat the physical examination in a cat with unexplained weight loss, as subtle abnormalities may be initially overlooked or discounted. If the muscle condition score was not initially noted, this should be established and recorded. It can be difficult to examine the oral cavity in an awake cat, but inspection of the teeth and gum line may reveal significant pathology. It is however important to bear in mind that a cursory visual examination does not rule out significant and impactful dental disease; the extent of painful conditions such as tooth resorption may only be determined with radiographic studies (Figure 1) 1. A thorough ophthalmologic examination should be performed, looking for signs of uveitis or chorioretinitis. These are non-specific findings, but are often seen in cats with fungal or protozoal infections (Figure 2) 2. Any skin or subcutaneous nodules should be carefully examined, particularly if noted in proximity to the mammary chain. It is also prudent to watch the cat ambulate to look for signs of joint, spinal, or neurological disease; many of us examine our feline patients on the table and may therefore fail to notice changes in gait, coordination, or muscle strength.
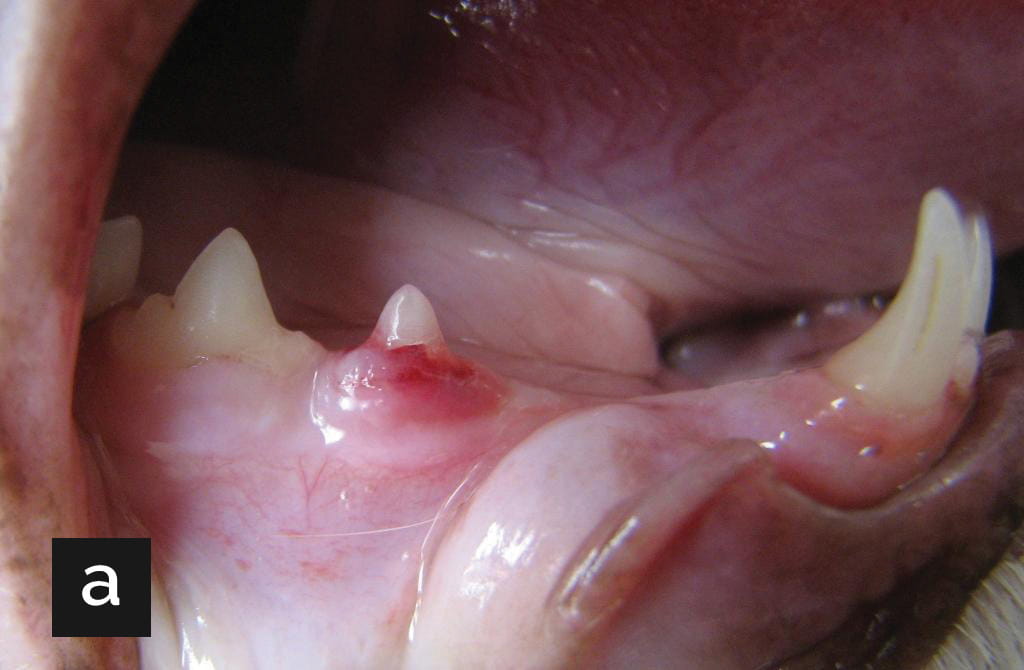
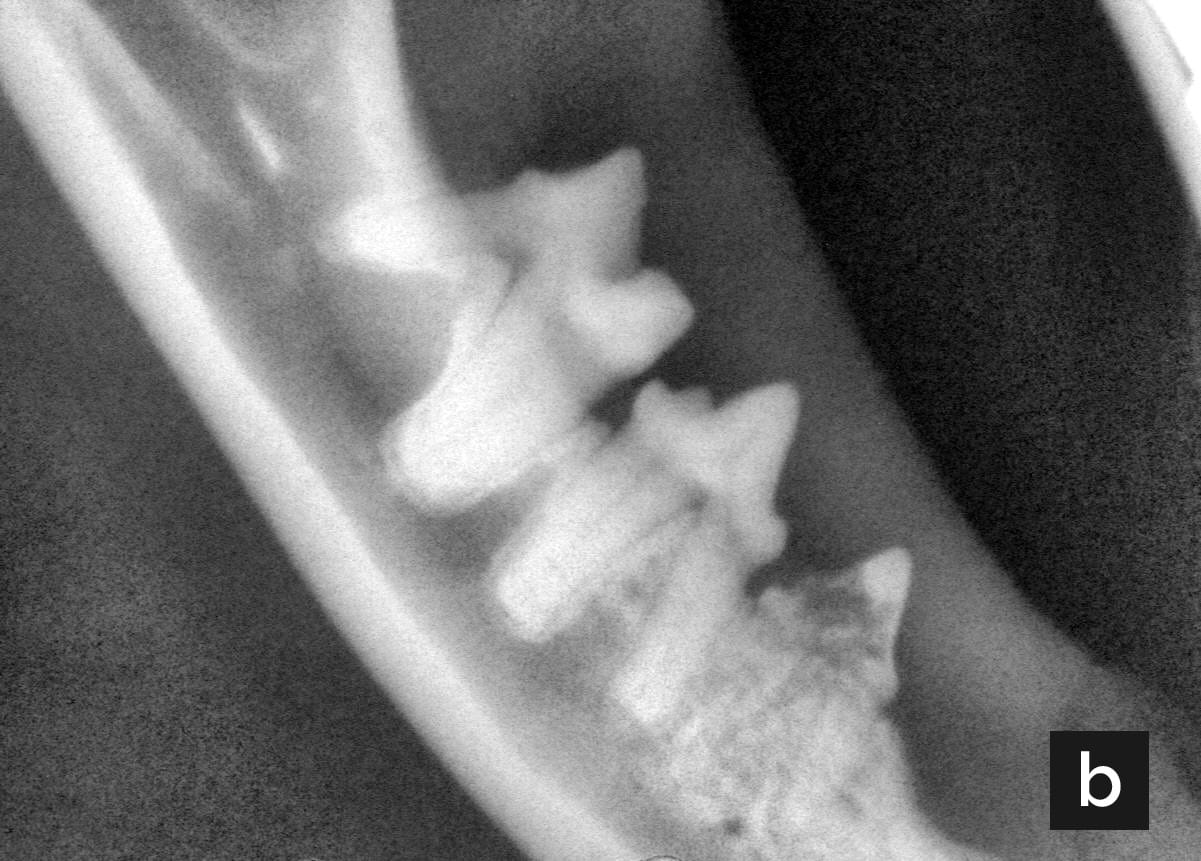
Figure 1. (a) The right mandible of an 8-year-old cat with unexplained weight loss. Note the area of hyperemia and erosion on the rostral side of tooth 407. (b) Radiographic image of the same patient. Note the lucent lesion in the caudal portion of tooth 407, and the loss of root structures; these changes are consistent with type 2 tooth resorption.
© Courtesy of Dr. Bert Dodd, Texas A&M University

Figure 2. All senior cats should be given a full ophthalmology assessment in the consult room, as this can reveal signs of systemic disease. This cat with systemic histoplasmosis had an active retinitis visible on ophthalmoscopic examination.
Courtesy Dr. Luc Vallone, Texas A&M University
Detailed information should be collected regarding the amount and type of food offered and consumed daily, with the goal of establishing the cat’s actual caloric intake. Unfortunately, collecting this piece of information can be difficult, as many cats are offered ad libitum dry food and the owner can have a limited understanding of how much is actually consumed. Under these circumstances, the owner should be asked to carefully measure or weigh out a daily allowance and determine the amount that remains uneaten, over several 24-hour periods.
It is also important to ask questions about changes in food preferences or feeding behaviors. Owners will sometimes describe their cat as “hungrier than usual” or “having a good appetite” if the cat exhibits food-seeking behaviors such as rubbing against the owner’s legs or vocalizing at established mealtimes. Although these activities suggest an interest in eating, it is still important to find out how much the cat takes in. Some cats will continue to “ask” for (and consume) treats or canned food but decrease their intake of kibble-based diets. Inattentive owners may think their cat’s intake is robust, when in fact the total calories consumed are inadequate.
It can be particularly difficult to determine a specific cat’s intake in multiple cat households. Under these circumstances, the owner should pay close attention to the group dynamics, as a more assertive cat may make it more difficult for a timid individual to get adequate access to food 3. Cats are thought to prefer to eat small, frequent meals when alone and unobserved; even in the absence of overt inter-cat aggression, the presence of other animals may limit food intake. Consideration should also be given to the location of the cat’s food; if this is up on a counter and the cat has orthopedic disease, it may take in less calories simply because accessing the food causes discomfort or requires significant effort. Food bowls placed next to noisy machinery, such as the washing machine, may also be problematic.
The actual calories needed to maintain a stable body weight vary from cat to cat, and will depend on age, sexual status, and activity level. For a sedentary, neutered adult cat, 40-66 kcal/kg body weight/day is a useful starting point, but this number should be regarded as a rough guide. Basal/resting energy requirement (RER) may also be calculated using a non-linear formula: body weight (kg)0.75 x 70. For the average middle-aged cat, the RER should be multiplied by a factor of 1.2 to 1.4 to determine the actual daily (or maintenance) energy requirement 4. If the cat is underweight, the ideal body weight should be used to determine the true daily energy requirement.
In the author’s experience, unexplained weight loss (i.e., weight loss in a cat with essentially unremarkable routine lab work) accompanied by adequate or robust caloric intake is uncommon. Diagnoses to consider under these circumstances include early hyperthyroidism, inflammatory bowel disease (IBD) and exocrine pancreatic insufficiency (Table 1). Additionally, some cats with cachexia related to cancer or chronic infection will experience weight loss despite an adequate food intake; however, hyporexia is more commonly noted under these circumstances 5. Patients with cachectic disorders may be recognized by the loss of muscle rather than adipose tissue; this pattern suggests changes in metabolism that are driven in part by increases in inflammatory cytokines such as tumor necrosis factor-alpha, and interleukins 1 and 6. The diagnostic possibilities for cats with unexplained weight loss and decreased intake are broader, and a more extensive list of differentials should be considered for these patients (Table 2).
Table 1. Diagnostic considerations in cats with unexplained weight loss despite adequate or excessive caloric intake.
| Disorder | Initial diagnostic considerations |
| Hyperthyroidism | Measure free thyroxine +/- feline thyroid stimulating hormone |
| Exocrine pancreatic insufficiency | Measure fasted trypsin-like immunoreactivity |
| Inflammatory/infiltrative bowel disease | Measure serum folate and cobalamin (B12) |
| Cachectic disorders (less likely) | Thoracic radiographs; abdominal ultrasonography |
Table 2. Diagnostic considerations in cats with unexplained weight loss and inadequate caloric intake.
| Disorder | Initial diagnostic considerations |
| Prehension disorders/oral discomfort | Observe feeding behaviors; sedated oral examination |
| Psychological stress | Assess environment, feeding practices, overall husbandry |
| Chronic kidney disease | Measure SDMA*; check blood pressure; quantify proteinuria; ultrasonography |
| Pancreatitis | Measure pancreas-specific lipase activity; ultrasonography |
| Inflammatory/infiltrative bowel disease | Measure serum folate and cobalamin (B12); ultrasonography |
| Idiopathic hypercalcemia | Measure ionized calcium; measure parathyroid hormone and parathyroid-related protein |
| Cachectic disorders (e.g., neoplasia; chronic infection). | Thoracic radiography; abdominal ultrasonography |
*SDMA = symmetric dimethylarginine
The “minimum database” for a feline patient with weight loss should include a complete blood count, serum biochemical profile with electrolytes, and urinalysis. A fecal floatation should be performed in the cat that has access to the outdoors. If the patient is 7 years or older, total thyroxine concentration should also be measured. As per recommendations from the American Association of Feline Practitioners, cats with evidence of systemic disease should also be tested for feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV).
Although this is a very comprehensive approach, patients with significant organic disease may have “normal” bloodwork. It is therefore important to closely evaluate the available data and compare – whenever possible – with historical findings (see Table 3). Some analytes, such as creatinine and albumin, will remain remarkably consistent over many years in healthy cats; looking for trends rather than simply focusing on “abnormal” values can therefore be very helpful. For example:
Table 3. Key laboratory parameters to trend in cats with unexplained weight loss and normal laboratory data.
| Parameter | Diagnostic considerations |
| Albumin | A decrease from baseline suggests GI disease Consider measurement of serum folate and cobalamin concentrations |
| Creatinine | An increase from baseline suggests CKD (in a euvolemic patient) Consider measurement of systolic blood pressure and renal ultrasonography |
| Thyroxine | An increase from baseline or a value within the upper half of the reference range suggests hyperthyroidism Consider measurement of free T4 +/- feline-specific thyroid stimulating hormone |
| Calcium | A robust total calcium suggests hypercalcemia Consider measurement of ionized calcium |
| Eosinophil count | A robust count is suggestive of lymphoma, mast cell tumor, protozoal or fungal infection, eosinophilic IBD Consider abdominal ultrasonography +/- infectious disease testing |
If the minimum data base does not provide any useful clues, the author will prioritize the investigation of GI tract function, and measure serum folate and cobalamin concentrations. Hypofolatemia indicates absorptive dysfunction in the duodenum, but this finding is a relatively insensitive indicator of disease, and a normal value does not discount inflammatory or neoplastic changes in this segment of the bowel 14. Serum cobalamin (B12) concentrations appear to be more useful, and subnormal values indicate ileal disease, intestinal dysbiosis, or exocrine pancreatic insufficiency. In the author’s practice, a concentration < 400 ng/L (reference range: 290-1,500 ng/L) is considered significant. Cobalamin deficiency may in turn impact appetite, so prompt recognition and management of hypocobalaminemia is important 15.
Another useful consideration is chronic pancreatitis. Affected cats may not vomit or exhibit overt signs of abdominal discomfort; instead, food intake is variably decreased. Although a definitive diagnosis requires pancreatic histopathology, a presumptive clinical diagnosis is usually based on a combination of signs (which may be limited to hyporexia), along with findings on abdominal ultrasonography and/or measurement of pancreas-specific lipase immunoreactivity. An increase in this parameter is strongly indicative of active pancreatic acinar cell damage; however, this may not be the only cause of a patient’s weight loss, and the possibility of concurrent GI tract disease or other systemic disorders should be considered 16. Additionally, pancreatic inflammation may wax and wane, so a result within the reference range does not exclude the possibility of cyclical bouts of inflammation; repeated measurement of pancreas-specific lipase may be helpful under these circumstances.
In canine patients, C-reactive protein is recognized as an emerging biomarker for inflammatory conditions, and increased concentrations have been associated with numerous disorders 17. This is a positive acute phase protein, and concentrations increase substantially (often > 20-fold) in response to inflammation, injury or neoplasia in dogs. Unfortunately, C-reactive protein does not seem to be useful in cats and it is not regarded as a reliable indicator of underlying inflammatory or neoplastic disease in this species. In one study, serum concentrations did not differentiate clinically normal cats from post-operative patients 18.
Audrey K. Cook
In the author’s opinion, an ultrasonographic examination of the abdomen is often a high yield diagnostic test in a cat with unexplained weight loss. Particular attention should be paid to the GI tract, and both the overall thickness of the various sections of the bowel, and the ratio of the thickness of the mucosal versus the muscularis layer, should be determined. As a general rule, any segment of the small intestine with wall thickness > 3 mm is a concern; similarly, a prominent muscularis can be indicative of pathology 19. Diffuse thickening of the muscularis is sometimes seen in clinically normal cats, but is more often associated with IBD or infiltrative GI conditions such as small cell lymphoma and histoplasmosis (Figure 3). Other intra-abdominal conditions causing low grade discomfort and secondary hyporexia such as chronic pancreatitis may be identified with ultrasonography, although subtle changes in echogenicity may be difficult to appreciate without a high-end machine or substantial experience (Figure 4) 16. Problems affecting the kidneys (e.g., a ureteral obstruction or hydronephrosis) can be easily identified with ultrasonography and may well cause enough chronic discomfort to impact food intake 20. Bear in mind that if the contralateral kidney is unaffected, serum creatinine and urine specific gravity will be within the reference ranges.

Figure 3. Ultrasonographic image of the jejunum in a cat with unexplained weight loss and subnormal serum cobalamin concentrations. Although the overall thickness of the bowel is within normal limits, the muscularis layer is more prominent than expected. This cat was subsequently diagnosed with small cell lymphoma via endoscopic biopsies.
© Courtesy Diagnostic Imaging Service, Texas A&M University

Figure 4. Ultrasonographic image of the left limb of the pancreas in a cat with unexplained weight loss and intermittently increased pancreas-specific lipase immunoreactivity. The pancreas is enlarged and hypoechoic, and the surrounding mesentery is hyperechoic.
© Courtesy Diagnostic Imaging Service, Texas A&M University
If appropriate, changes in size or echogenicity of organs such as liver, spleen, and lymph nodes should prompt the collection of cytologic specimens by fine needle aspiration.
Thoracic radiographs are an appropriate next step if abdominal ultrasonography does not provide additional direction. The fact that clinical signs of respiratory disease are lacking should not be used to discount the possibility of conditions such as pulmonary neoplasia that could account for weight loss 21. A three-view study (left and right lateral images and a ventrodorsal image) should be performed, and interpretation by a board-certified radiologist may be helpful (Figure 5).
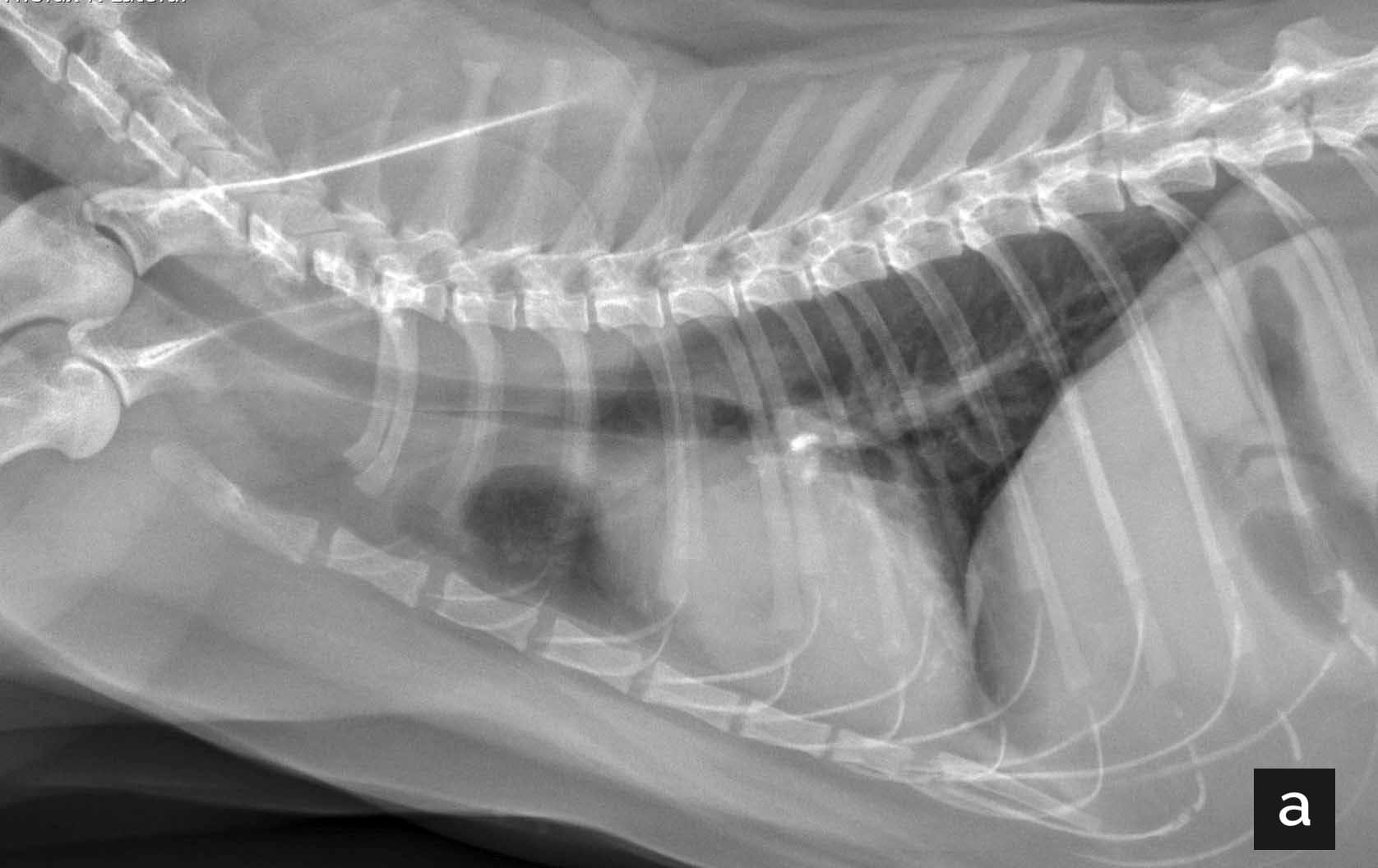
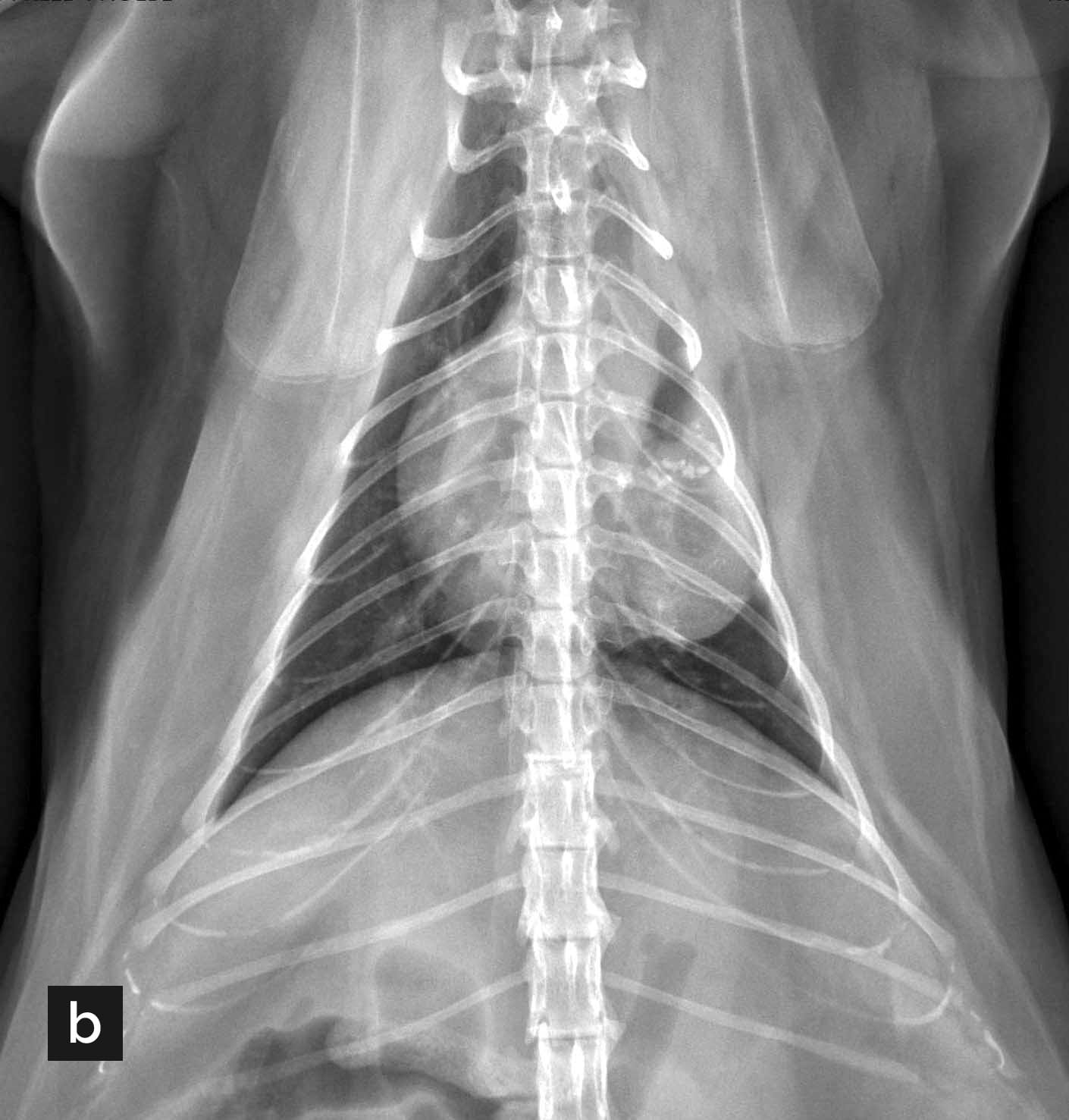
Figure 5. (a) Right lateral thoracic radiograph of an 11-year-old cat with unexplained weight loss. A mineralized density is noted just caudal to the carina. (b) Ventrodorsal thoracic radiograph of the same cat. A soft tissue mass is evident in the left thorax, which effaces the cardiac silhouette and the lateral body wall. There are multifocal angular mineral opacities throughout the mass, which are most numerous dorsally, and there is a large, rounded, gas opacity within the mass. There is a mild rightward mediastinal shift. Cytologic findings were consistent with carcinoma.
© Courtesy Diagnostic Imaging Service, Texas A&M University
If food intake is clearly suboptimal, efforts must be made to increase caloric intake while diagnostics are completed; appropriate approaches include offering alternative foods and administering appetite stimulants. New foods should ideally be energy dense and highly palatable; digestibility may also be important if there are concerns about underlying GI dysfunction.
Cats have complex behaviors regarding food preferences and are often reluctant to consume an unfamiliar diet. For example, switching a cat accustomed to an exclusively dry diet to a feeding plan based on canned food can be difficult; the cat may nibble at the new food but fail to consume adequate amounts. Simply changing the shape or flavor of a kibbled diet can also be problematic if the cat is not used to novel foods. As a general rule, the old diet should be offered alongside one or two new options, with each new food served on a separate dish and tested for 24 hours before any decision is made. It is very important that the amount of food eaten is carefully measured, so that caloric intake can be monitored appropriately.
Increasing the palatability of a familiar or novel food by adding a small amount of a highly flavored protein (e.g., tuna or salmon) may encourage intake. In the short term (2-3 weeks), feeding a home-cooked diet is unlikely to be harmful. However, prolonged dependence on an unbalanced diet may have significant consequences; input from a nutritionist is warranted under these circumstances.
Over the last decade, various medications have been used off-label to encourage food intake in cats. Many of these had questionable safety and efficacy. Fortunately, there are now two products licensed in many countries to specifically address weight loss in cats. Mirtazapine can be administered transdermally on the inner side of the pinna q24H at a dose of 2 mg/cat 22. This drug is thought to improve appetite by increasing central norepinephrine levels, along with antagonism of specific serotonin receptor subtypes. In a study looking at its effect in cats with ≥ 5% unintended weight loss, the treated group gained an average of 3.9% body weight in 2 weeks, while those in the placebo group gained just 0.4% 22. Transdermal mirtazapine is generally well tolerated, although skin irritation at the site of application has been reported. Overdose can result in vocalization, agitation, and vomiting, and the recommended dose or frequency should be decreased in any cat with significant hepatic or renal disease.
Capromorelin is a ghrelin receptor agonist, and a liquid oral formulation is licensed in the USA for use in cats, at a dose of 2 mg/kg q24H 23. Ghrelin is a peptide hormone secreted by the gastric mucosa; serum levels increase during the interprandial interval and stimulate food-seeking behaviors. This product is specifically approved for use in feline CKD and has been shown to promote weight gain in cats with this condition. In an eight-week study, treated cats averaged a weight gain of > 5%; animals in the placebo group lost an average of 1.6% of body weight 24. Ghrelin has significant anti-inflammatory properties, and additionally stimulates growth hormone secretion. Side effects include salivation, and occasional cats may experience a temporary decrease in heart rate and blood pressure following administration of the first few doses.
If adequate food intake cannot be maintained with these simple interventions, more aggressive steps (e.g., placement of an esophageal feeding tube) should be considered.
Even small decreases in the weight of a feline patient can signal a significant underlying disorder; cats (much like people) tend to maintain a stable weight or slowly gain over time unless caloric intake is deliberately restricted, or disease arises. Unexplained weight loss therefore requires a logical and thorough evaluation. Supportive care to encourage adequate intake should be instituted while diagnostics are pursued.
Audrey K. Cook
Dr. Cook graduated with distinction from Edinburgh University in Scotland and completed her residency in small animal internal medicine at the University of California, Davis Read more
Insect contamination of petfoods is a potential problem in tropical countries; this paper offers an overview of the situation and how the risks can be minimized
Mistakes happen to all of us in veterinary practice; this article looks at how different people react in different ways when things go wrong and – importantly – discusses how we can best cope with mistakes.
Cats are living longer and better lives; how can we ensure that the healthcare we offer them is optimal? This article offers some hints.
OA in cats remains underdiagnosed and undertreated, despite its widespread prevalence; this paper looks at how we can surmount the challenge of early diagnosis, leading to better treatment interventions.
