Treatment
With autoimmune/immune-mediated dermatoses, there are two therapeutic approaches that can be utilized for treatment: immunosuppression or immunomodulation (Table 1a, b and c). Disease type and severity determines the approach. Most dogs with discoid lupus erythematosus, rabies vaccine-induced cutaneous vasculitis, pinnal margin vasculitis, and symmetric lupoid onychodystrophy will respond favorably to, and can be maintained on, immunomodulatory drugs. Other diseases such as pemphigus foliaceus, erythema multiforme, systemic lupus, and various other vasculitides will need immunosuppressive therapies.
Table 1a. Commonly used drugs for the treatment of cutaneous autoimmune and immune-mediated diseases. 1,24,25
Immunomodulatory drugs may take time to effect an improvement (generally seen within 3-4 weeks of starting therapy) so if the clinical signs are severe, a tapering course of high dose glucocorticoids can be utilized initially to obtain rapid control, along with a chosen immunomodulatory drug. Once remission is achieved the immunomodulatory drug can be continued as maintenance. Note that both the glucocorticoids and the immunomodulatory drug should be given initially since the latter class of drug can take time to be effective: this will help prevent relapse of the disease once steroids are tapered. The primary benefit of immunomodulatory drugs is that they have less serious adverse side effects and decreased health impact.
When immunosuppressive therapy is utilized, the most commonly used drug is a glucocorticoid. Initially, high doses are needed to achieve remission, and then tapered to the lowest possible dose that will maintain remission with minimal adverse systemic effects. In many autoimmune diseases, adjunctive therapies are necessary in order to permit the glucocorticoid dose to be lowered to a level which minimizes adverse side effects. In the more severe cases, it is not unusual to combine several different immunosuppressant drugs to achieve and maintain remission. As many of these medications can have adverse side effects on the liver and bone marrow, blood monitoring every 2-3 weeks for the first several months is recommended, with maintenance monitoring every 4-6 months. If significant changes in the blood parameters are noted, the offending drug should be discontinued and replaced with another medication. The most commonly utilized adjunctive medications include azathioprine, cyclosporine, mycophenolate mofetil, cyclophosphamide, and chlorambucil. In more severely affected dogs, supportive care for open wounds, fluid corrective therapy and monitoring of serum protein levels may be necessary. Use of human intravenous immunoglobulin (hIVIg) has shown promise in treating severe autoimmune dermatoses when other treatments are failing 6.
Topical therapies can be useful with more localized lesions or for sporadic flare-ups. The most commonly used topicals include betamethasone or tacrolimus. Betamethasone has the benefit of rapidly controlling inflammation and disease symptoms but can induce dermal atrophy with chronic use; therefore, transition to tacrolimus is prudent if a topical is needed for long-term use.
Table 1b. Commonly used drugs for the treatment of cutaneous autoimmune and immune-mediated diseases. 1,24,25
There are four phases to consider in the treatment of cutaneous autoimmune dermatoses: induction phase, transition phase, maintenance phase and determining cure 1. With the induction phase, the goal is to stop the inflammatory component as quickly as possible and suppress the immunologic response directed towards the skin. In this phase, higher doses of medications are normally necessary. If an acceptable response is not noted in a timely manner, another treatment regimen should be considered; i.e., alternative medications chosen or additional medications added to the current treatment regimen. In the transition phase, drugs are tapered to minimize side effects and adverse reactions. When drug combinations are utilized, those with the greatest side effects – such as glucocorticoids – are the first to be tapered. Medications are slowly reduced, often over several weeks or months, until an acceptable maintenance dose of medications is achieved, or until signs recur. If this happens, the medications are increased until remission is again achieved, then tapered down to the last dose that maintained the patient’s symptoms under acceptable control (the maintenance phase). A “cure” is considered for cases of immune-mediated dermatoses that have achieved remission and are successfully controlled with maintenance therapy but do not then recur after cessation.
Cessation of maintenance therapy in a patient that has been well-controlled is a difficult decision to make, especially if the initial disease was severe. This decision should be one that is mutually agreed between the clinician and owner; it is essential that the client is well-informed, with the realization that if the patient relapses, achieving remission a second time may be more difficult. When to discontinue maintenance therapy depends on the type of disease, whether or not a trigger was identified and removed, and the risk to the patient if therapy is discontinued. In many cases, maintenance therapy for 8-12 months is recommended before cessation 1. In animals where the risk of recurrence outweighs the benefit of discontinuing therapy, the drugs can be maintained life-long with appropriate lab work monitoring.
Future vaccinations are often discouraged in cases of autoimmune dermatoses, even in those where vaccination is not a known trigger. Concern lies with the idea that vaccination may stimulate a broad, non-specific immune response, possibly initiating recrudescence of the autoimmune disease 7. The author prefers to discontinue rabies vaccination and monitor titer levels for distemper and parvovirus. If titers are not sufficient to maintain proper immunity, a risk vs. benefit assessment should be performed before considering re-vaccination.
Table 1c. Commonly used drugs for the treatment of cutaneous autoimmune and immune-mediated diseases. 1,24,25
Specific diseases
Pemphigus foliaceus
The most common autoimmune skin disorder in the dog, pemphigus foliaceus (PF) is a pustular to crusting autoimmune dermatitis. PF affects the epidermis, targeting various adhesion molecules, especially desmosomes, which hold keratinocytes together. In human PF, the desmoglein-1 glycoprotein (DSG1) in the desmosome is the primary target of autoantibodies 8 and the same glycoprotein was previously suspected to be the primary target in dogs 9,10; however, it is now believed to be a minor autoantigen 11, with current evidence suggesting that desmocollin-1 is a major autoantigen in canine PF 12.
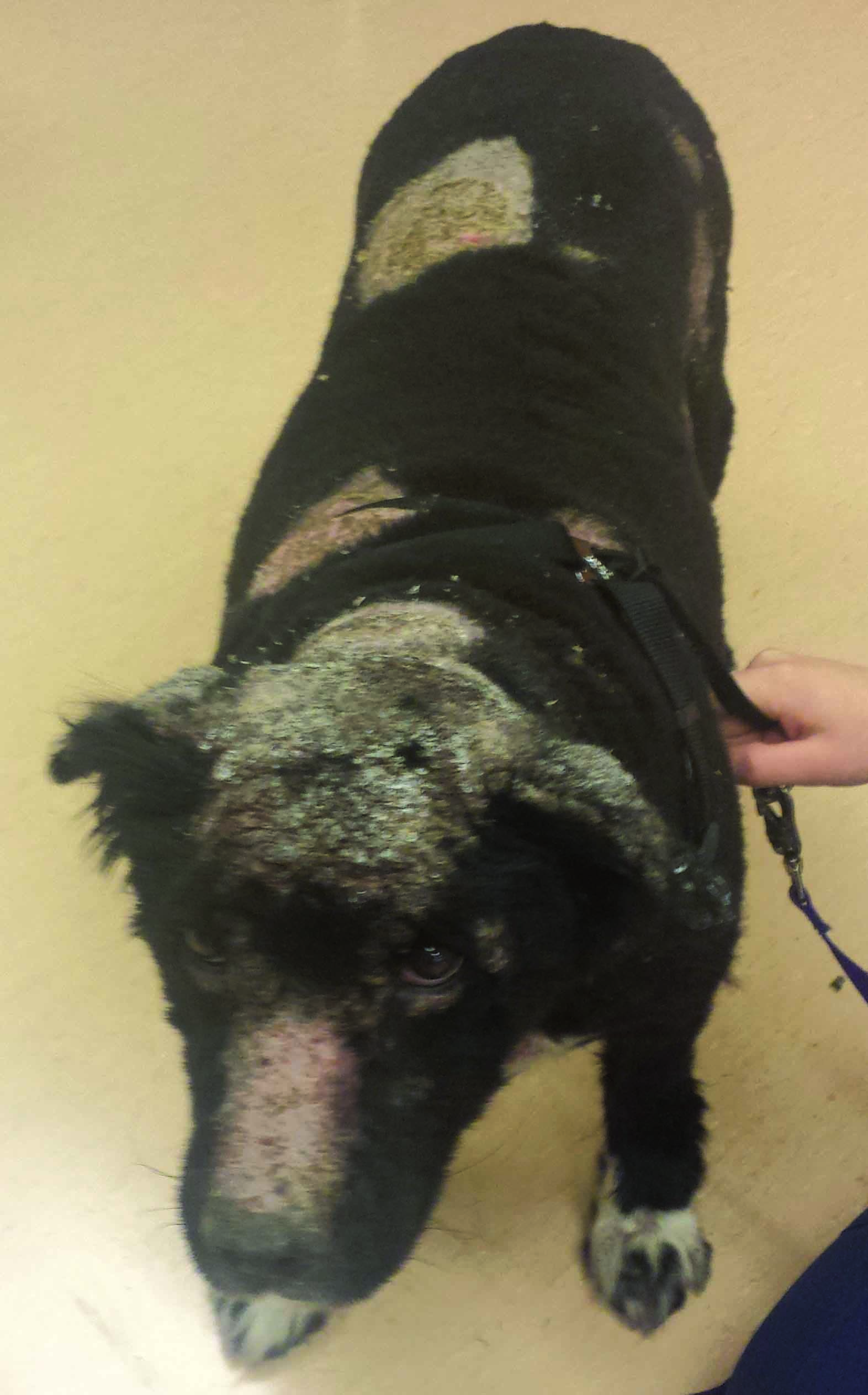
Genetic factors appear to play a role in the development of PF, with Akitas and Chow Chows the breeds considered most at risk 10. Triggers include chronic allergic skin disease and drugs (antibiotics, NSAIDs, topical flea spot-ons), but the most important one is ultraviolet light 1,10. The initial lesion is a macule that rapidly progresses to pustules, which are often large and coalesce. The pustules are frequently fragile and easily ruptured, resulting in crusting. As a result, crusts are the most common clinical sign 1,9,10. Erosions may be noted; ulcerations are rare but can be present in cases complicated with a deep pyoderma. Canine PF is often characterized by crusting, initially involving the face (especially dorsal muzzle and nasal planum, peri-ocular region and pinnae), and subsequently progressing to a generalized form (Figure 2).
Cytology of an intact pustule or of the skin below a crust will often reveal the presence of numerous non-degenerate neutrophils surrounding individual or rafts of acantholytic keratinocytes, which appear as large, rounded basophilic nucleated keratinocytes (Figure 1). Histological evaluation reveals subcorneal pustules containing neutrophils and variable numbers of eosinophils, and acantholytic keratinocytes 13. Treatment often involves high doses of steroids with an adjunctive immunosuppressant and topicals for localized treatment.
Figure 2. Pemphigus foliaceus: (a) Honey-colored crusting involving the dorsal muzzle and nasal planum is depicted. Mild erosion of the nasal planum can be seen underlying the lifted crust. (b) A more generalized presentation of pemphigus foliaceus.
© Amy Shumaker
Discoid lupus erythematosus
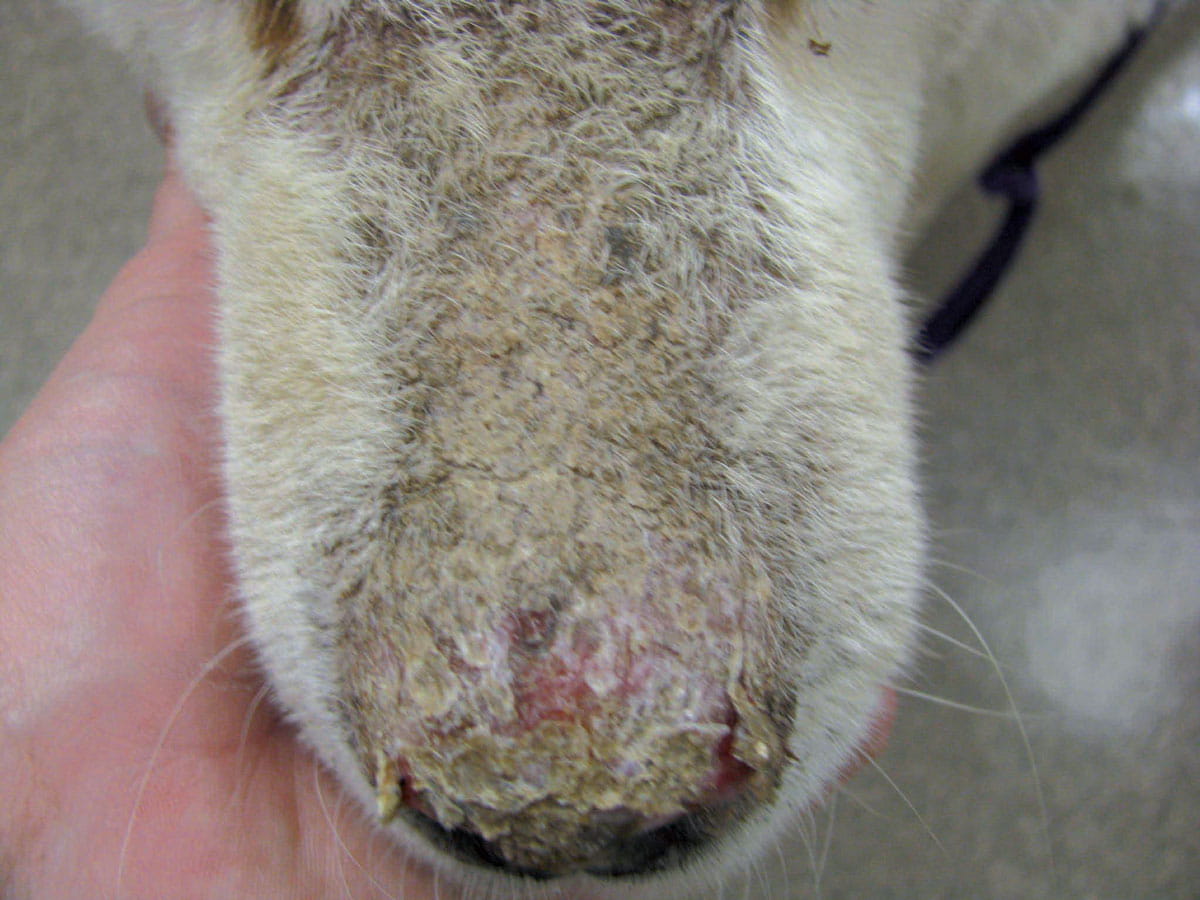
Also referred to as “collie nose” or cutaneous lupus erythematosus, discoid lupus erythematosus (DLE) is a benign ulcerative disease without systemic manifestations 1. DLE is generally localized to the nasal planum, but can involve the sun-exposed areas of the pinnae and peri-ocular region and there are reports of generalized variants 14. The most common clinical sign is initial loss of the cobblestone architecture of the nasal planum progressing to depigmentation and scaling (Figure 3). With chronicity, erosions, ulcerations and crusting occur. Annular to polycyclic hyperpigmented plaques involving the neck, trunk and extremities may be seen in the generalized variant cases.
Histopathology reveals interface basal cell degeneration (apoptosis), with a moderate pleocellular lichenoid infiltrate of the dermis 13. As this disease can closely mimic mucocutaneous pyoderma both clinically and histopathologically, cytologic evaluation of the nasal planum below a crust can be useful; treatment of mucocutaneous pyoderma is recommended if bacteria are present.
In most cases of DLE, the use of potent immunosuppressants are unnecessary; a systemic immunomodulatory approach utilizing a tetracycline (doxycycline, minocycline) and niacinamide coupled with topical therapy (topical steroids, tacrolimus) is often successful at controlling the disease. In refractory or severe cases, high doses of corticosteroids may be needed initially. In the reported generalized variants, hydroxychloroquine or cyclosporine were effective treatments 14,15. As sunlight plays a significant role in DLE, it is important that sun exposure be minimized with avoidance and use of sunscreens. Vitamin E supplementation (400 IU daily) may also be helpful.