Pathogenesis
Multiple factors are involved in the pathogenesis of Malassezia dermatitis, such as the host’s corneocyte adherence mechanisms and the prevalence of concurrent symbiotic organisms, as well as the host’s immune response.
Adherence to canine corneocytes may be an important factor in the pathogenesis of Malassezia dermatitis in some dogs. Yeast cell walls, which consist of chitin, glucan, chitosan, and mannan 1, contain trypsin-sensitive proteins or glycoproteins that contribute to adherence to canine corneocytes. M. pachydermatis also specifically expresses adhesins that bind to mannosyl-bearing carbohydrate residues on the canine corneocyte. This adherence mechanism, however, does not apparently play a role in the pathogenesis of Malassezia dermatitis in Basset Hounds (which are prone to Malassezia overgrowth) but seems to be significant in other breeds 10.
M. pachydermatis appears to have a symbiotic relationship with commensal staphylococci species, although suggestions that Malassezia dermatitis is associated with prior antibiotic therapy have not been substantiated. The two organisms produce growth factors and micro-environmental alterations that are mutually beneficial; thus, there are increased numbers of Staphylococcus pseudintermedius or S. intermedius on dogs with concurrent Malassezia 1,4,8. In fact, 40% of dogs with Malassezia overgrowth are diagnosed with staphylococcal pyoderma due to the symbiotic relationship between the two organisms 3,11.
The yeast may elicit a spectrum of immunological responses in the host. The humoral response is stimulated, exemplified by higher numbers of antibodies found against more antigens in dogs with Malassezia versus healthy dogs 12,13. However, the elevated IgA and IgG levels found in dogs with Malassezia dermatitis does not appear to offer any additional protection against infection with the yeast. Cell-mediated immunity may play a larger role in protection against disease than humoral immunity. For example, Basset Hounds appear to have decreased lymphocyte responses to Malassezia versus healthy dogs which do not get overgrowth of the yeast 14.
Another type of immunological response that can occur in dogs is a hypersensitivity or inflammatory reaction. The preceding reactions to yeast products and antigens appear to be the main culprit in the pathogenesis of Malassezia dermatitis, since the yeast itself remains in the upper level of the epidermis 4,8. As the yeast adheres to the canine corneocytes, it secretes various substances including zymosan, urease, proteases, phosphohydrolase, phospholipases (especially phospholipase A2), lipoxygenases, phosphatases, glucosidase, galactosidase, and leucine arylamidase. These virulence factors cause alteration of the local pH, proteolysis, lipolysis, complement activation, and eicosanoid release in the skin, thus inciting the inflammatory response and pruritus 1,4,8. Furthermore, higher levels of Malassezia-specific IgE to allergens of 45, 52, 56 and 65 kDa have been found in atopic dogs compared to normal dogs, further substantiating the hypersensitivity potential of yeast 15.
Factors that predispose to pathogenicity
Factors that may predispose M. pachydermatis to become pathogenic, rather than remain commensal, may include any of the following: increased humidity, skin folds, endocrine diseases, keratinization disorders, genetic predisposition, immunologic dysfunction, hypersensitivity diseases, and increased numbers of symbiotic staphylococci.
Humidity may be important as Malassezia organisms appear to be more common in otic canals and skin folds and prevalence increases in humid climates 1. Endocrine diseases, such as hypothyroidism, primary and iatrogenic hyperadrenocorticism, and diabetes mellitus may allow increased availability of nutrients and growth factors for the yeast. This may be due to changes in cutaneous fatty acid concentrations, abnormal keratinocyte lipogenesis, and alterations in sebaceous gland function 16,17. American Cocker Spaniels, Shih Tzus, English Setters, West Highland White Terriers, Basset Hounds, Toy and Miniature Poodles, Boxers, Australian and Silky Terriers, Cavalier King Charles Spaniels, Dachshunds, and German Shepherd dogs appear to be at a higher risk for Malassezia dermatitis, suggesting a genetic component to the disease 4,6,8. Dysfunction in secretory IgA or cell-mediated immunity may also contribute to pathogenicity in some dogs 2,4. For example, Basset Hounds with Malassezia dermatitis have decreased in vitro lymphocyte blastogenic response to M. pachydermatis antigen when compared to healthy Bassets, indicating a cell-mediated immune dysfunction 14. Hypersensitivity diseases, such as flea allergy dermatitis, cutaneous adverse food reaction, and atopic dermatitis, may also predispose dogs to Malassezia dermatitis due to incitation of the inflammatory cascade and resultant pruritus.
In summary, any dermatoses that produces disruption of the stratum corneum barrier, whether mechanical (due to pruritus), or biochemical (due to endocrinopathies, keratinization or immunologic disorders), may potentially allow Malassezia virulence factors to gain exposure to the subcorneal immune system, resulting in pathogenicity of the yeast.
Diagnosis
Clinical presentation
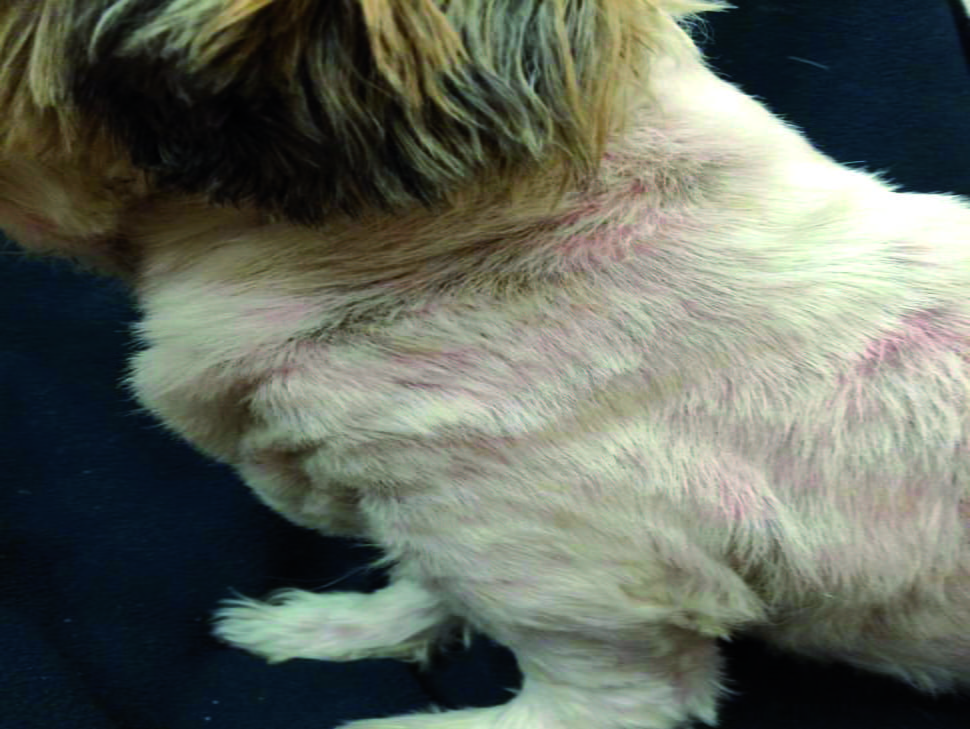
Dermatological lesions of Malassezia dermatitis may be localized (Figure 2) or generalized. They commonly manifest in warm, moist areas such as lip folds, otic canals, axillae, groin, ventral neck, medial thighs, interdigital skin, perianal and perivulvar regions, and other intertriginous areas (Figure 3). Concurrent dermatoses, such as staphylococcal pyoderma, allergies, or keratinization disorders, are seen in 70% of affected dogs 1,4. Appearance of the lesions commonly starts in the humid summer months – which is also the peak time for seasonal allergies – and may persist through the winter months. Historically, patients lack a response to glucocorticoids.