Feline lungworm and heartworm
Lungworm and heartworm are an underestimated but potentially serious threat for many cats, as this article reveals.
Systemic hypertension is sometimes called the “silent killer” as it is usually asymptomatic until severe and often irreversible organ damage occurs.
The organs most vulnerable to damage from systemic hypertension are the eyes, brain, heart, and kidneys, because of their rich arteriolar blood supply.
Accurate, repeatable and reliable blood pressure measurement in a stress-free animal is essential for the correct diagnosis and proper management of cats with hypertension.
Indirect blood pressure measurement methods are most commonly used in veterinary clinics and, if performed using a strict protocol, can give reliable results.
Systemic hypertension, which is defined as a persistent increase in systemic blood pressure, has become an increasingly common condition seen in veterinary practice today. This is mainly due to the growing awareness among veterinarians that hypertension is a disease that impacts both the lifespan and the quality of life of dogs and cats – although the increased incidence of systemic hypertension may also be a consequence of their improved life expectancy, as the frequency of diseases specific to old age, including hypertension, has increased. A significant contribution to this awareness has been the evolution in recent years of reliable devices for measuring blood pressure, which are now much more readily available to practitioners.
Alice M. Rădulescu
Systemic hypertension can be caused by a number of factors:
Situational hypertension, also called “white coat” hypertension, is a transient increase in blood pressure from activation of the sympathetic branch of the autonomic nervous system due to arousal or anxiety. The “white coat” effect will typically increase systolic blood pressure (SBP) by 15-20 mmHg 1, but greater increases (or even decreases) may sometimes occur 2. It is essential to be aware of this effect because the neurohormonal changes in a cat associated with the stress of a clinic visit can lead to a misdiagnosis of hypertension. However, this may not be easy to identify, because the effects of anxiety on blood pressure are unfortunately not predictable for individual animals.
Secondary hypertension, which is a persistent increase in blood pressure in animals suffering from other diseases known to cause hypertension, is the most diagnosed form in small animals. The most common conditions associated with systemic hypertension are kidney disease, endocrinopathies (hyperthyroidism, hyperadrenocorticism, diabetes, primary hyperaldosteronism), and pheochromocytoma. Secondary hypertension can also occur in animals that have received medication or ingested toxic substances that have a hypertensive effect, such as glucocorticoids, mineralocorticoids, erythropoiesis stimulants, phenylpropanolamine, cocaine, or methamphetamine.
Idiopathic hypertension is a persistent pathological increase in blood pressure in the absence of any identifiable underlying cause. It is nowadays more frequently diagnosed than before, with recent studies showing an incidence of about 13-20% in cats 3,4,5.
There are differences in the onset, progression, and prognosis for the different types of hypertension. Secondary hypertension is found mainly in older cats (ten years plus) and associated with conditions specific to old age, such as chronic kidney disease, hyperthyroidism, and hyperaldosteronism. Once the underlying cause has been identified and treated, the hypertension may resolve. Primary (idiopathic)hypertension is much less commonly diagnosed in small animals, but can be found in cats of any age. Because there is no identifiable underlying disease, the aim here is to use medication to control the blood pressure and prevent damage to the target organs. In humans "white coat hypertension" is considered a risk factor for subsequent hypertensive lesions, and the issue of its treatment has been raised. There is little evidence for this in animals, so there is currently no justification for treating feline situational hypertension.
Also called the “silent killer”, systemic hypertension is often asymptomatic until severe organ damage occurs. The organs most vulnerable to systemic hypertension are the eyes, brain, heart, and kidneys, because of their rich arteriolar supply 6. The damage is collectively referred to as target organ damage (TOD).
Ocular damage syndrome in animals with systemic hypertension is called hypertensive choroidopathy and retinopathy 7. In cats with high blood pressure, ocular lesions are frequently noted, with a reported prevalence between 68.1-100% in affected animals 4,8. Signs that are easy for the owner to note, including hyphema (Figure 1) and fixed mydriatic pupils (Figure 2), are often the reason for presentation to the veterinarian. However, the most common lesion diagnosed is exudative retinal detachment, with an increased risk when systolic blood pressure exceeds 180 mmHg. Other lesions, such as retinal hemorrhage, multifocal retinal edema, retinal vessel tortuosity, retinal perivascular edema, papillary edema, vitreous hemorrhage, secondary glaucoma, and retinal degeneration have all been observed. Diagnosis of ocular damage requires an ophthalmic evaluation with funduscopic examination.

Figure 1. Bleeding into the anterior chamber of the left eye in a cat with systemic hypertension (unilateral hyphema).
© Alice M. Rădulescu

Figure 2. Severe mydriasis in a cat with systemic hypertension presented for sudden blindness due to bilateral retinal detachment.
© Alice M. Rădulescu
Systemic hypertension can lead to brain damage from edema and hemorrhage, and is classified as hypertensive encephalopathy 9. Neurological signs associated with systemic hypertension have been reported both in cats 4,8 and dogs 10, although cats appear to be more prone. Hypertensive encephalopathy is more likely to occur with sudden increases in blood pressure, or where values are persistently greater than 180 mmHg. The clinical signs observed are typical of intracranial disease and can include altered mentation, vocalization, disorientation, ataxia, head tilt, nystagmus, torpor, convulsions, or even coma. Confirmation of brain damage requires neurological examination and specialist diagnostics such as magnetic resonance imaging.
The kidneys are one of the preferred targets of systemic hypertension, but many affected animals also have concomitant chronic kidney disease, and it can be difficult to determine which condition came first. In the case of systemic hypertension, failure of the local blood pressure control system leads to increased intraglomerular capillary pressure, followed by proteinuria, which promotes the development of glomerulosclerosis, which in turn can exacerbate the initial hypertension 10. Protocols for evaluating hypertensive renal patients should include measurement of blood pressure, urine analysis, quantitative evaluation of proteinuria or albuminuria 11, measurement of serum SDMA and creatinine, radiography, and abdominal ultrasound. The IRIS (International Renal Interest Society) subclassification based on proteinuria using the Urine Protein:Creatinine ratio (UPC) (Table 1) may help determine whether antihypertensive therapy is indicated for individual cases.
Table 1. IRIS subclassification of feline chronic kidney disease based on proteinuria status.
| Proteinuric status | UPC ratio |
|---|---|
| Non-proteinuric | <0.2 |
| Borderline proteinuric | 0.2-0.4 |
| Proteinuric | >0.4 |
The heart is a target organ in both hypertensive cats 12 and dogs 13. Chronic increases in afterload caused by systemic hypertension lead to compensatory myocardial hypertrophy in an attempt to normalize wall stress. Dogs are more likely to show diffuse and symmetric left ventricular concentric hypertrophy, proximal aortic bulb dilation, and aortic insufficiency, whilst cardiac changes detectable by echocardiography in affected cats include concentric hypertrophy of the left ventricular wall and interventricular septum, septal hypertrophy in the subaortic region (Figure 3), and dilation of the proximal aorta. At clinical examination any gallop sound, arrhythmia or murmur is strongly suggestive of heart damage, but additional tests such as thoracic radiography, electrocardiography and echocardiography are needed for confirmation.

Figure 3. An ultrasound scan showing localized septal hypertrophy at the base of the interventricular septum in a cat with hyperthyroidism and systemic hypertension. This change may be seen also in normal older cats.
© Alice M. Rădulescu
There are several situations in which it is recommended that blood pressure (BP) should be measured. Systemic hypertension is considered a disease of elderly animals, and therefore it is recommended to assess BP as part of any routine screen in older pets. A recent study found that the estimated incidence risk of systemic hypertension for cats older than nine years is 23.7% 14, so routine measurement should start at this point. Blood pressure should also be checked for any patient with evidence of a condition that is associated with hypertension, or when a drug that may cause secondary hypertension has been administered. In particular, blood pressure assessment should always be performed in cats with clinical manifestations compatible with target organ damage, and in this situation a single high value may be sufficient to confirm the diagnosis of systemic hypertension. Box 1 shows an algorithm for decision-making when investigating a cat that shows signs of target organ damage that may be related to hypertension, or that has clinical signs suggestive of a condition which can cause systemic hypertension.
Animals with a suspected diagnosis of idiopathic hypertension should have their blood pressure measured several times to rule out situational hypertension, typically 5-7 times within the session and averaged. The first value obtained is usually ignored, as are individual readings which are much higher or lower than the majority of the results. If there is any doubt about the accuracy of the measurements, it is recommended to repeat the procedure after acclimatization or to reschedule for another day. In patients with conditions that can lead to systemic hypertension, regular blood pressure monitoring is advisable to detect any development over time; the author recommends doing this at eight-week intervals. This approach is also crucial for assessing the response to antihypertensive treatment.
Box 1. A diagnostic algorithm for cats where there is target organ damage or evidence of an underlying condition that can cause systemic hypertension 15.
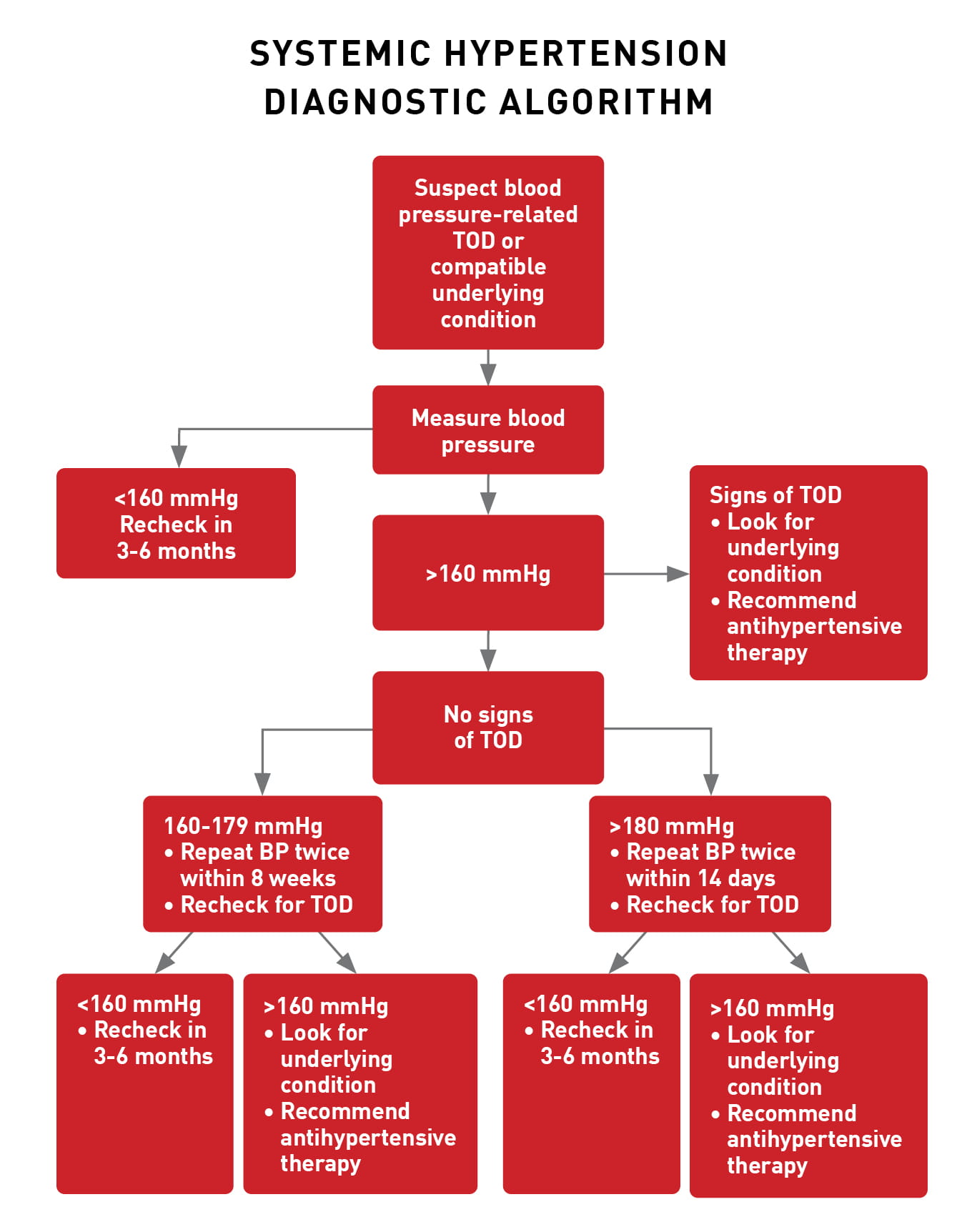
Alice M. Rădulescu
Accurate, repeatable, and reliable blood pressure measurement is essential for the correct diagnosis and proper management of patients with hypertension, and a standardized staging system has been developed for the diagnosis of high blood pressure and the risk of developing target organ damage 15 (Table 2). Direct measurement by arterial catheterization is the gold standard for assessing blood pressure, but this is an invasive and impractical method for daily use in conscious patients, so indirect methods such as Doppler sphygmomanometry and high definition oscillometry (HDO) are commonly used in the veterinary clinic.
Table 2. ACVIM classification for risk categories for target organ damage (TOD) 15.
| Risk category | SBP (mmHg) | TOD Risk |
|---|---|---|
| Normotensive | <140 | Minimal |
| Prehypertensive | 140-159 | Low |
| Hypertensive | 160-179 | Moderate |
| Severely hypertensive | >180 | High |
Direct blood pressure measurement involves placing a catheter in an artery and connecting it to a pressure transducer, the most common site for catheter placement in small animals being the dorsal pedal artery. After preparing the puncture site, palpate the pulse and insert the catheter in line with the vessel and at an angle of 45 ̊ (Figure 4). Pulsed and rapid blood flow through the catheter indicates successful placement in the artery. The catheter should then be fully advanced into the vessel and the stylus removed. Note that arterial catheters require a secure bandage to prevent dislodgement and bleeding, and should be clearly labeled (Figure 5) to avoid confusion with intravenous lines. Regular flushing of the arterial line is also required to ensure permeability and accuracy. The catheter is connected via a saline-filled tube to a pressure transducer placed at the same height as the heart. Once connected to the patient, the transducer must be calibrated to zero and then connected to a monitor; this will display a continuous pressure wave and numerical readouts of systolic (SAP), mean (MAP), and diastolic (DAP) blood pressures (Figure 6). From a clinical point of view, direct monitoring of arterial BP is indicated for animals in shock, hemodynamically unstable, at high anesthetic risk, severely hypertensive, requiring sympathomimetic support, or benefiting from mechanical ventilation 16.

Figure 4. For direct measurement of blood pressure, a catheter is placed in a distal artery, usually the dorsal pedal artery.
© Alice M. Rădulescu

Figure 5. The arterial catheter must be securely bandaged once in place, and clearly labeled to avoid confusion with intravenous lines.
© Alice M. Rădulescu

Figure 6. A monitor displays the readout as a continuous pressure wave, as well as systolic, mean and diastolic blood pressures.
© Alice M. Rădulescu
Obtaining repeatable, reliable, and accurate blood pressure measurements by indirect methods is not easy, as several factors related to the device, patient, and operator can influence the results obtained. However, studies have shown a good correlation between direct and indirect methods, allowing practitioners to use the latter option, which is non-invasive, relatively inexpensive, and easy to perform.
Indirect measurement methods are based on detecting the return of pulsating blood flow after occlusion of the artery with an inflatable cuff. The choice of cuff is critical to ensure accurate readings; its width should be about 40% of the limb circumference. If the cuff is too large, the values will be underestimated; if too small, the values will be overestimated 17. Ideally the cuff should be positioned at the same level as the heart.
In order to obtain accurate blood pressure values, it is very important that measurement is done using a standard protocol (Table 3). This protocol must consider not only the practical aspects of the assessment and the type of device used, but also the environment in which the measurements are made, and how data collected are recorded. Blood pressure measurement should be common practice in veterinary clinics, but standardization is essential for practitioners to obtain meaningful results.
Table 3. A standard protocol for indirect blood pressure measurement (from 15).
|
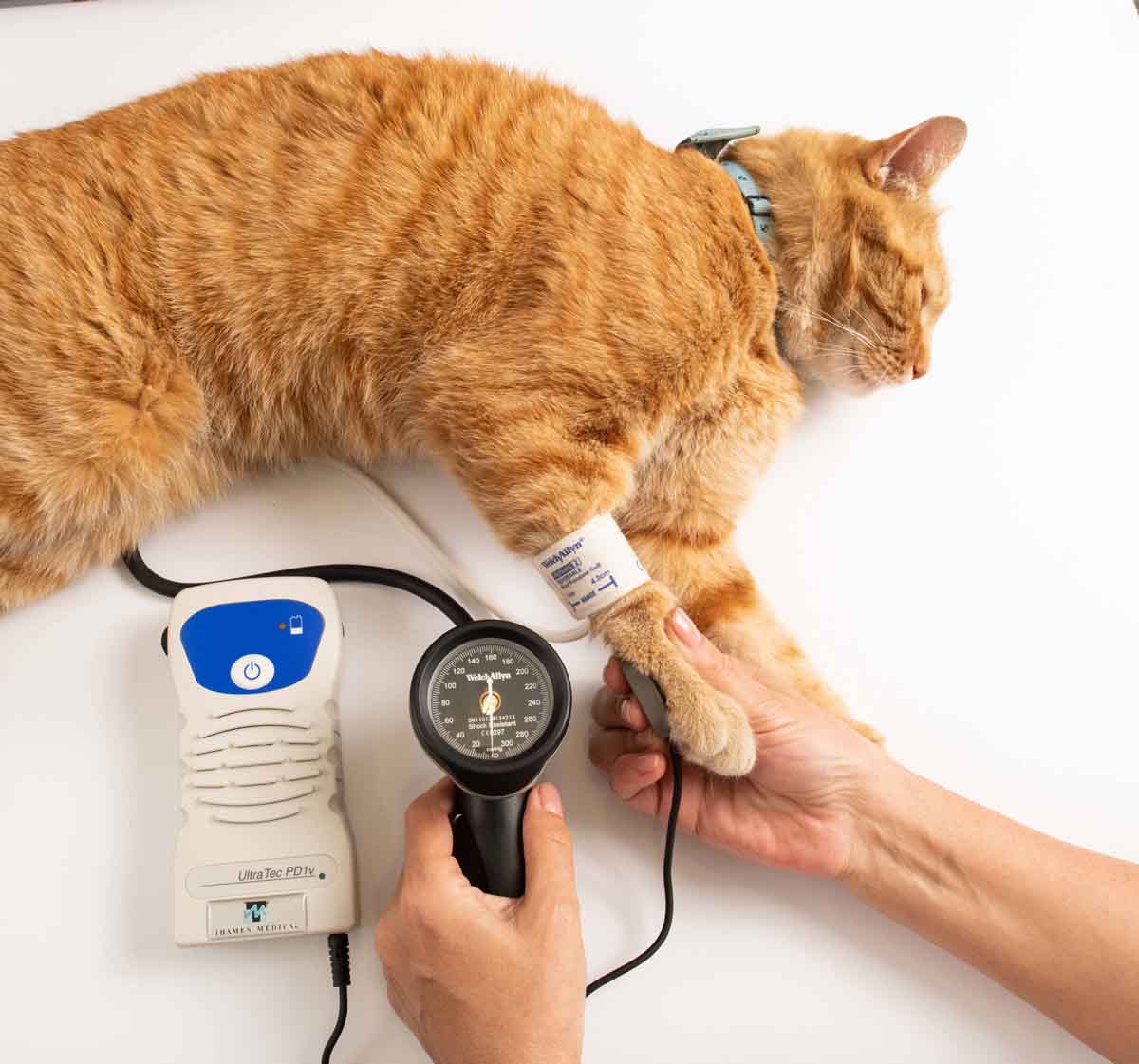
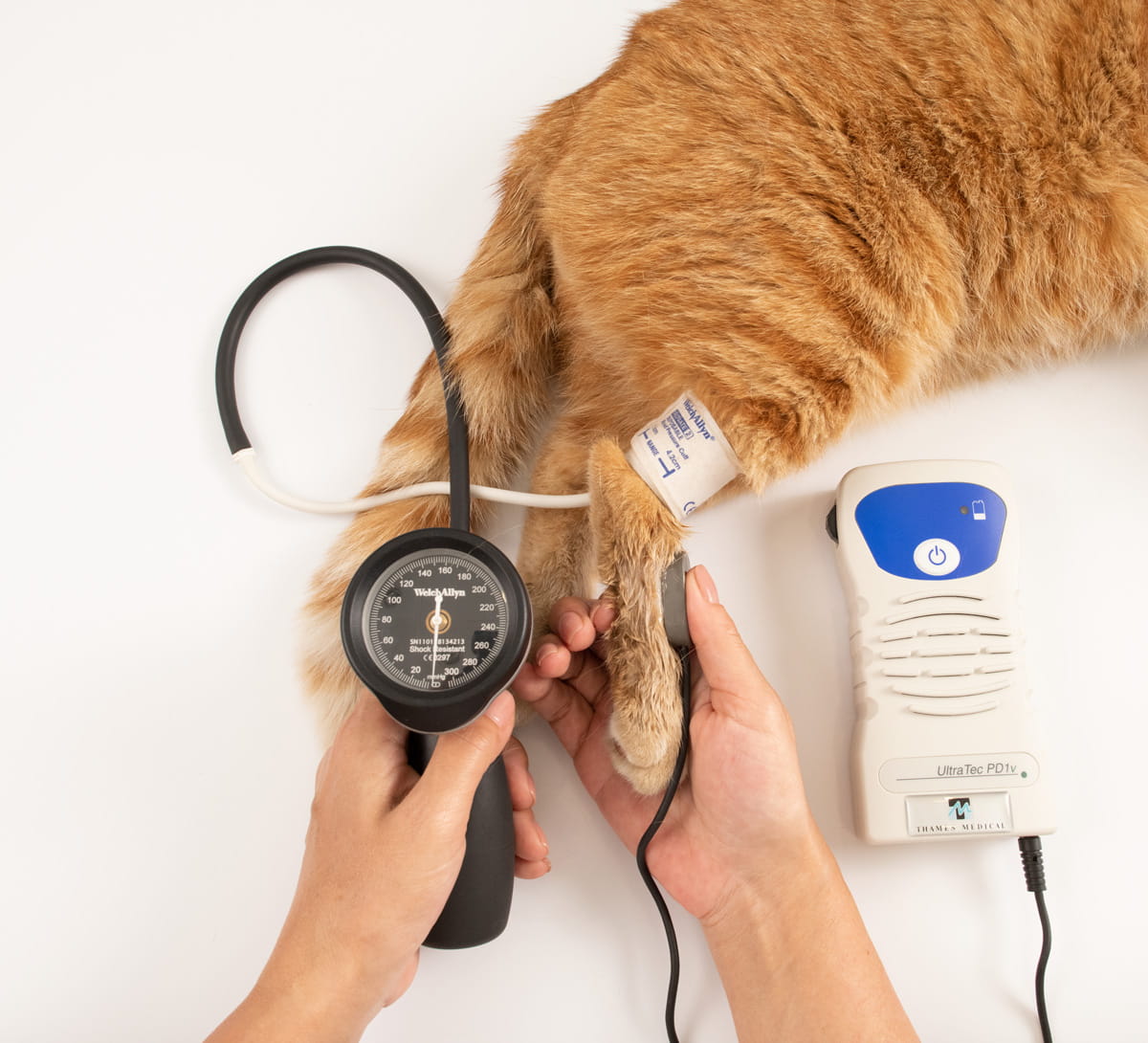
The Doppler method is currently recommended by most authors due to its speed and simplicity 18, and in addition the values obtained have good correlation with those from direct catheterization 19. The technique detects arterial blood flow via an ultrasound transducer connected to an amplifier and a speaker or headphones. The probe is placed over a distal artery below the inflatable cuff which is connected to an aneroid manometer. The most common placement sites are on the palmar aspect of the metacarpal region (ulnar artery) or the dorsal aspect of the metatarsal region (dorsal pedal artery) (Figure 7). Because detection of blood flow requires excellent contact between the probe and the skin, it may be necessary to first shave the chosen site and degrease the skin with alcohol. The inflatable cuff is positioned immediately above the prepared area and ultrasound gel is applied to the probe, which is then held using light pressure over the artery and parallel to the blood flow. Once blood flow is audible, the cuff should be inflated until the pulse can no longer be heard, and then deflated slowly while observing the manometer. The point at which the pulse first becomes audible is the systolic pressure value.
 |
| a |
 |
| b |
Figure 7. Blood pressure measurement using the Doppler technique with placement of the transducer over the ulnar artery in the forelimb (a) and the dorsal pedal artery in the hindlimb (b).
© Alice M. Rădulescu
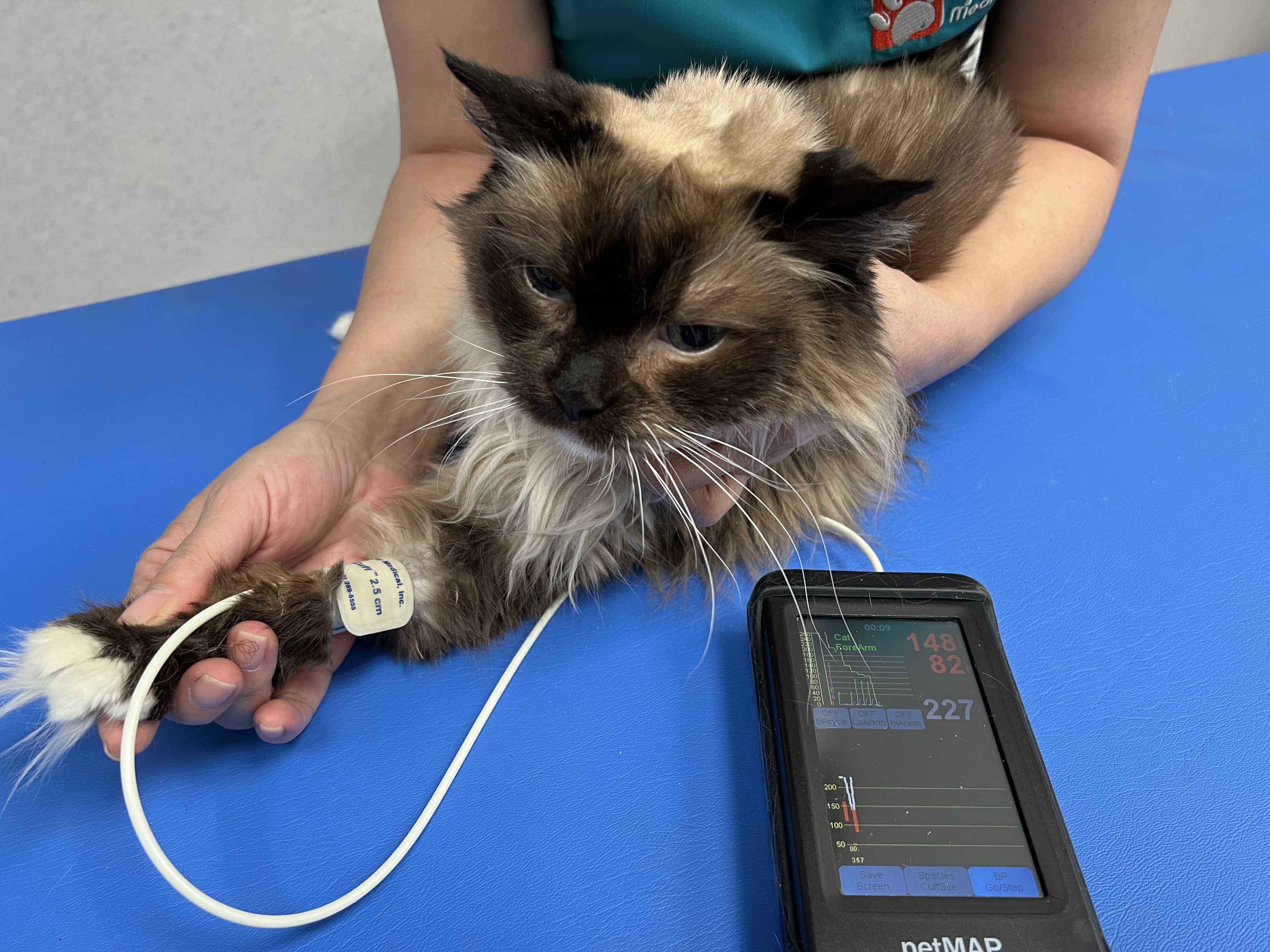
The oscillometric method is based on detecting oscillations (periodic fluctuations) inside the cuff produced by pulsations of the arterial wall. As the cuff is deflated, the oscillations increase rapidly, peak at MAP, and then rapidly decrease at DAP. The device screen will display SAP, MAP, DAP and pulse rate, usually alongside a graph of the detected oscillations which helps confirm the accuracy of the measurement. Most devices will typically measure MAP and employ programmed algorithms to calculate the SAP and DAP.
The technique is very simple, but the choice of cuff size, cuff placement, and animal position play a significant role in the accuracy of the results. The most common cuff placements are in the forelimb (using the radial artery), in the metatarsal region of the hind limb (the caudal saphenous artery) or at the tail base (the coccygeal artery) (Figure 8).
 |
| a |
 |
| b |
Figure 8. Blood pressure measurement with the high definition oscillometry (HDO) technique showing placement of the cuff at the level of the forelimb in the radial region (a) and at the base of the tail (b).
© Alice M. Rădulescu
The recommended approach for assessing a potential hypertensive cat involves reliable measurements of blood pressure and identifying possible target organ damage. Once a diagnosis of systemic hypertension has been established, further investigation should be performed to detect the underlying condition and establish appropriate treatment, which is beyond the scope of this paper; however, it is not enough simply to start a hypertensive cat on medication to lower its blood pressure without first determining the etiology wherever possible.
Marino CL, Cober RE, Lazbik MC, et al. White-coat effect on systemic blood pressure in retired racing greyhounds. J. Vet. Intern. Med. 2011;25:861-865.
Belew AM, Barlett T, Brown SA. Evaluation of the white-coat effect in cats. J. Vet. Intern. Med. 1999;13:134-142.
Jepson RE, Elliott J, Brodbelt D, et al. Effect of control of systolic blood pressure on survival in cats with systemic hypertension. J. Vet. Intern. Med. 2007;21:402-409.
Maggio F, DeFrancesco TF, Atkins CE, et al. Ocular lesions associated with systemic hypertension in cats: 69 cases (1985-1998). J. Am. Vet. Med. Assoc. 2000;217(5):695-702.
Elliott J, Barber PJ, Syme HM, et al. Feline hypertension: clinical findings and response to antihypertensive treatment in 30 cases. J. Small Anim. Pract. 2001;42:122-129.
Henik RA: Systemic hypertension and its management. Vet. Clin. North Am. Small Anim. Pract. 1997;26(6):1355-1372.
Crispin SM, Mould JR. Systemic hypertensive disease and the feline fundus. Vet. Ophthal. 2001;4(2):131-140.
Littman MP. Spontaneous systemic hypertension in 24 cats. J. Vet. Intern. Med. 1994;8(2):79-86.
Brown CA, Munday JS, Mathur S, et al. Hypertensive encephalopathy in cats with reduced renal function. Vet. Pathol. 2005;42(5):642-649.
Jacob F, Polzin DJ, Osborne CA, et al. Association between initial systolic blood pressure and risk of developing a uremic crisis or of dying in dogs with chronic renal failure. J. Am. Vet. Med. Assoc. 2003;222(3):322-329.
Bacic A, Kogika MM, Barbaro KC, et al. Evaluation of albuminuria and its relationship with blood pressure in dogs with chronic kidney disease. Vet. Clin. Pathol. 2010;39(2):203-209.
Chetboul V, Lefebre HP, Pinhas C, et al. Spontaneous feline hypertension: clinical and echocardiographic abnormalities, and survival rate. J. Vet. Intern. Med. 2003;17(1):89-95.
Misbach C, Gouni V, Tissier R, et al. Echocardiographic and tissue Doppler imaging alterations associated with spontaneous canine systemic hypertension. J. Vet. Intern. Med. 2011;25(5):1025-1035.
Conroy M, Chang YM, Brodbelt D, et al. Survival after diagnosis of hypertension in cats attending primary care practice in the United Kingdom. J. Vet. Intern. Med. 2018;32(6):1846-1855.
Acierno MJ, Brown S, Coleman AE, et al. ACVIM consensus statement: Guidelines for the identification, evaluation, and management of systemic hypertension in dogs and cats. J. Vet. Intern. Med. 2018;32(6):1803-1822.
Cooper E, Cooper S. Direct systemic arterial blood pressure monitoring. In: Burkitt Creedon JM, Davis H, eds. Advanced Monitoring and Procedures for Small Animal Emergency and Critical Care. Iowa, Willey-Blackwell, 2012;122-133.
Geddes LA, Whistler BS. The error in indirect blood pressure measurement with the incorrect size of cuff. Am. Heart J. 1978;96(1):4-8.
Jepson RE, Hartley V, Mendl M, et al. A comparison of CAT Doppler and oscillometric Memoprint machines for non-invasive blood pressure measurement in conscious cats. J. Feline Med. Surg. 2005;7(3):147-152.
Binns SH, Sisson DD, Buoscio DA, et al. Doppler ultrasonographic, oscillometric sphygmomanometric, and photoplethysmographic techniques for non-invasive blood pressure measurement in anesthetized cats. J. Vet. Intern. Med. 1995;9(6):405-414.
Alice M. Rădulescu
Dr. Rădulescu graduated in 1999 from Bucharest before going on to gain a master’s degree in Veterinary Medicine in 2005 Read more
Lungworm and heartworm are an underestimated but potentially serious threat for many cats, as this article reveals.
Diaphragmatic hernia is a common consequence of traumatic injury in cats and may be fatal if not promptly diagnosed and treated; this article reviews the pathophysiology and treatment of the condition.
Chylothorax is a differential for any cat with pleural effusion; here Elizabeth Rozanski describes her preferred approach to such cases.
Asthma is a common disease in cats, but it can mimic many other pathologies, and the diagnosis and treatment of the condition can be challenging, as this paper explains.
