Feline lungworm and heartworm
Lungworm and heartworm are an underestimated but potentially serious threat for many cats, as this article reveals.
Issue number 32.2 Other Scientific
Published 08/12/2022
Also available in Français , Deutsch , Italiano and Español
Diaphragmatic hernia is a common consequence of traumatic injury in cats and may be fatal if not promptly diagnosed and treated; this article reviews the pathophysiology and treatment of the condition.

Traumatic diaphragmatic hernia (TDH) should be considered in any cat presented following suspected major trauma.
TDH may present in the absence of respiratory compromise, particularly in more chronic cases.
Diagnosis of TDH can usually be made by thoracic radiography.
Stabilization of patients prior to surgery is paramount, but the prognosis is generally good, with recent studies suggesting mortality rates below 15%.
Traumatic diaphragmatic hernias (TDH) in cats are most frequently the result of blunt trauma, with vehicular accidents accounting for up to 90% of cases where the cause of the trauma is known 1,2,3. Patients may present with life-threatening respiratory compromise, and prompt diagnosis and initial stabilization, followed by appropriate surgical treatment, are crucial to maximize the probability of survival. With this, the prognosis is good in the majority of cases.
The diaphragm is located at the caudal thoracic aperture, dividing the thorax from the abdomen. Approximately 20% of the diaphragm is a central Y-shaped tendon, with the remainder consisting of muscle, divided into the pars lumbalis dorsally, the pars sternalis ventrally and the pars costalis bilaterally. The pars lumbalis arises from the cranioventral border of L4 and the body of L3, forming the right and left crura; the right crus is substantially thicker than the left. The pars costalis inserts obliquely across ribs 8-13, and the pars sternalis inserts on the xiphoid process. The diaphragm is innervated by the paired phrenic nerves, with the blood supply via the main phrenic arteries and cranial phrenic veins. The diaphragm is pierced dorsally by the aortic hiatus, allowing passage of the aorta, the thoracic duct and the azygous and hemiazygos veins between the thorax and abdomen. In the mid-diaphragm between the paired lumbar muscles, the esophagus and the left and right vagus nerves pass through the esophageal hiatus, while the caudal vena cava and right phrenic nerve enter the abdomen via the caval foramen in the central tendon 4.
Contraction of the diaphragm and external intercostal muscles during inspiration increases the volume of the thoracic cavity by displacing the abdominal contents caudally and everting the caudal ribs. This leads to a reduction of intra-thoracic pressure, expansion of the lungs and movement of air into the lungs. Relaxation of the intercostal muscles and diaphragm during expiration produces a rise in intrathoracic pressure and air moves out of the lungs. Modification of intra-abdominal pressure by diaphragm contraction also has a role in the expulsion of urine, feces and vomitus.
Traumatic diaphragmatic hernias may result from direct or indirect injury. Direct injury from penetration of the diaphragm by a sharp object or projectile is common in human patients but rare in companion animals 5,6. Indirect injury occurs when sudden compression of the abdominal cavity with an open glottis results in an increased abdominal to thoracic pressure gradient, causing stretching and subsequent rupture of the diaphragm 7. The diaphragmatic muscle is substantially weaker than the central tendon and therefore ruptures more easily: in one study of 23 cats with TDH, all tears occurred in the diaphragmatic muscle 8. Herniation likely occurs with equal frequency on the left and right in cats: around 15% of animals have ventral tears or multiple sites of herniation 1. In contrast to dogs, where radial and circumferential diaphragmatic tears are diagnosed with equal frequency, approximately 60% of tears in cats are circumferential and only 20% have a radial morphology 9.
Once a diaphragmatic tear is present, abdominal viscera are likely to move into the thoracic cavity (Figure 1). The most commonly herniated organs are the liver (55-82% of cases), small intestine (47-71%), stomach (34-53%), spleen (32-65%) and omentum (6-58%). The large intestine (6-16%), gallbladder (0-38%), pancreas (0-10%) and kidney (0-13%) herniate less frequently 1,2,3,8,9.
Herniation may cause cardiorespiratory compromise secondary to pulmonary compression by abdominal viscera or pleural fluid, or can lead to incarceration or strangulation of the herniated organs. Liver herniation may initiate hepatic venous stasis and necrosis: obstruction of hepatic venous outflow leads to lymph extravasation and resultant pleural effusion. Intestinal incarceration may result in obstruction, whilst strangulation can cause necrosis and perforation. Extrahepatic biliary tract obstruction may develop following herniation of the gall bladder 10. Herniation of the stomach can cause dilatation with or without volvulus 11,12 and urothorax can occur secondary to kidney herniation 13.

Figure 1. Right lateral radiograph of a cat showing disruption of the diaphragmatic silhouette (blue arrow), cranial displacement of the right kidney and stomach (green arrow), and ventral displacement of the caudal lung lobes (red arrow).
© Catrina Pennington
TDH are more common in cats with outdoor access (and thus have a greater probability of suffering blunt trauma) and more frequently seen in male cats; the average age at presentation is two to five years, with a reported range of 3 months to 14 years 1,2,3,8,9,14. Domestic short-hair cats are most commonly affected, likely due to their over-representation in the population. Although signalment may increase the index of suspicion for TDH, it should be considered as a differential diagnosis in any animal presenting after known or suspected trauma.
Diagnosis is based on history, clinical signs and diagnostic imaging.
There may be a history of recent trauma (e.g., a road traffic accident) but the causative event is often unknown. Cats may present acutely or chronically: 20-30% of affected animals may present more than 14 days after the initiating trauma 1,2,3 because of delayed herniation due to occlusion of small tears by omentum, progressive herniation of abdominal contents over time exacerbating respiratory compromise, or incarceration of herniated organs and pleural effusion developing secondary to contraction and fibrosis of the hernia ring during healing 15.
In acute cases respiratory abnormalities (dyspnea, tachypnea and cyanosis) are the most frequently reported history, although up to 10% of cases may present with vomiting or anorexia and no respiratory signs 9. The history in chronic cases is more variable; up to 50% of cats are reported to show dyspnea or tachypnea at home, while 33% are said to have gastrointestinal signs (anorexia, obstipation or vomiting). Fifty percent of chronic TDH cases with gastrointestinal signs have no history of tachypnoea or dyspnea, whilst coughing is the presenting complaint in 13% of cases, but 19% of chronic cases are actually reported to be found incidentally whilst the cats are under investigation for other problems 14.
Over 70% of acute cases exhibit tachypnea and/or dyspnea at examination. Other less common clinical findings include muffled heart sounds, increased or decreased lung sounds, reduced palpable abdominal contents, abdominal pain, hypothermia and orthopneic posture 1,2,9. Acute cases may also have clinical signs relating to hypovolemic shock, such as tachycardia, poor quality peripheral pulses, prolonged capillary refill time, abnormal mentation and pallor. Appropriate stabilization must be performed prior to further investigation.
In chronic TDH situations, decreased heart and/or lung sounds are the most common clinical finding in 75% of cases (Figure 2), while only 56% of affected cats are tachypneic or dyspneic on clinical examination 14. Concurrent soft tissue injuries are identified in approximately 40% of cats diagnosed with TDH 2: their presence (along with other signs of trauma, e.g., scuffed nails, broken teeth and orthopedic injuries) can increase the index of suspicion for TDH in cases without a complete recent history.

Figure 2. In chronic traumatic diaphragmatic hernia (TDH) decreased heart and/or lung sounds may be found in up to 75% of cases, thus careful thoracic auscultation is advisable during the clinical examination.
© Shutterstock
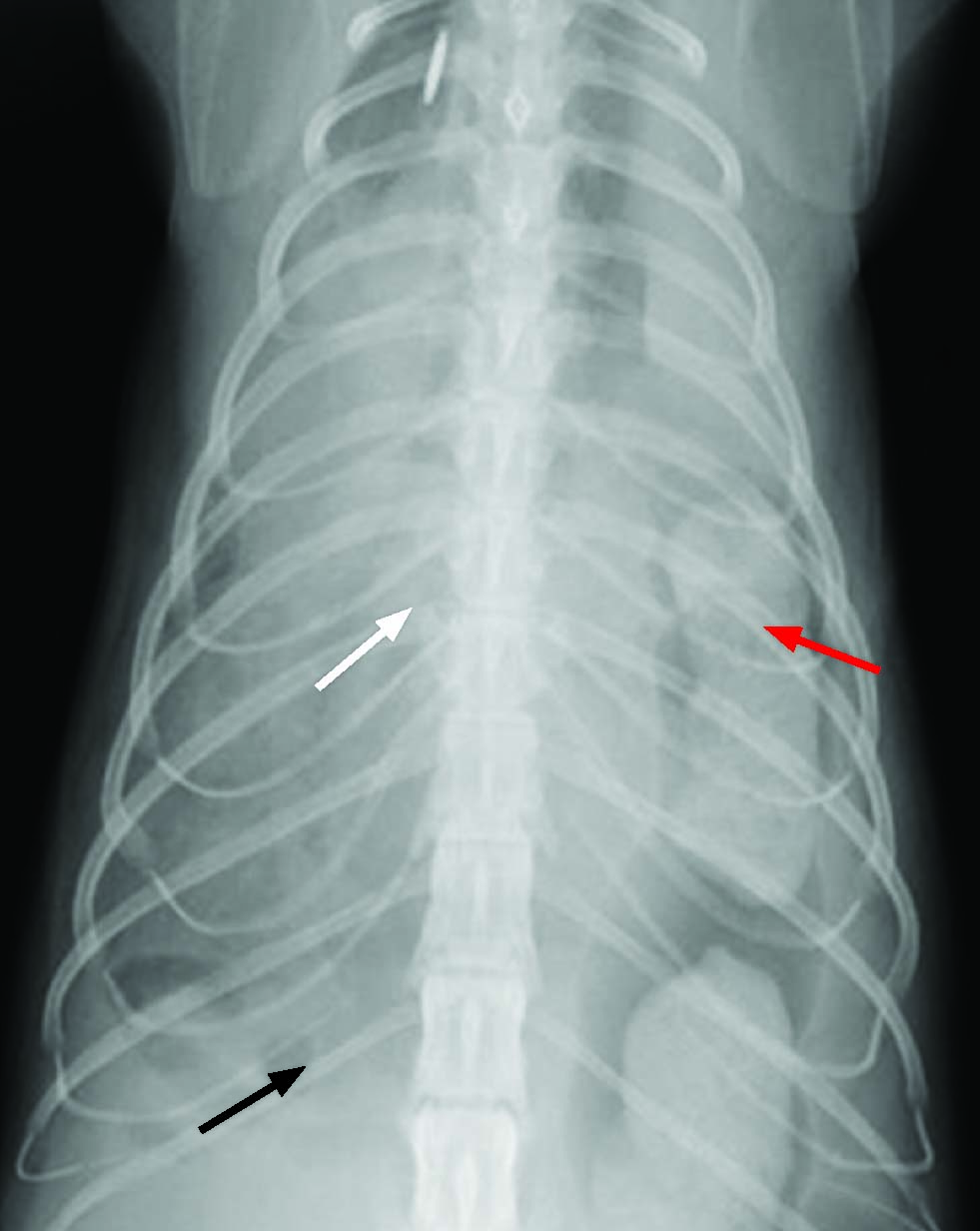
Orthogonal thoracic radiographs should be taken of any cat suspected to have suffered severe trauma (Figure 3). Care must be taken during radiographic positioning to avoid undue stress which may exacerbate respiratory compromise, so patients should be handled gently with minimal restraint. In many cases induction of general anesthesia, which allows the airway to be secured by endotracheal intubation, is preferable to heavy restraint or sedation.
 |
| a |
 |
| b |
Figure 3. Orthogonal thoracic radiographs should be taken of any cat suspected to have suffered severe trauma. Here dorsoventral (a) and right lateral (b) views of a cat demonstrate loss of the normal diaphragmatic line (black arrows) with effacement of the cardiac silhouette (white arrows) and extensive migration of abdominal organ contents within the thoracic cavity (red arrows).
© Catrina Pennington
Up to 97% of patients show disruption of the normal diaphragmatic silhouette on lateral thoracic radiographs 16, but it is important to remember that this can be caused by conditions other than TDH (e.g., congenital peritoneopericardial diaphragmatic hernia). Increased soft tissue / fluid density in the thorax with pleural fissure lines may be present if there is pleural effusion (Figure 4), while cranial displacement of the liver or stomach, effacement of the cardiac silhouette, increased density of the ventral lung fields, intestinal gas shadows within the thorax, and reduced lung volume may also be seen 9. Radiographic evidence of concurrent orthopedic injuries are reported in up to 14% of cats 9, with rib fractures present in 6% of cases 14.
The sensitivity of thoracic ultrasonography for the diagnosis of diaphragmatic hernias has been shown to be 90% in one study: findings included identification of abdominal contents within the thoracic cavity and loss of the diaphragmatic line 14 (Figure 5). Ultrasonography is a useful technique in animals where pleural effusion complicates radiographic diagnosis.

Figure 4. Left lateral radiograph of a cat showing loss of the normal diaphragmatic line, effacement of the cardiac silhouette (blue arrow) and material of a soft tissue/fluid opacity within the thorax and abdomen, consistent with pleural and peritoneal effusion (green arrows).
© Catrina Pennington

Figure 5. An ultrasound image of a cat showing disruption of the lung-diaphragm interface and presence of hepatic tissue within the thorax.
© Catrina Pennington
Routine hematology and serum biochemistry screens should be performed to guide pre-operative stabilization (e.g., to determine if a blood transfusion may be required). Hematology may reveal anemia and leucocytosis with or without a left shift. Serum biochemistry may reveal various electrolyte derangements, elevations in alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP) and bilirubin associated with herniation of the liver 1,9,14 and hypoproteinemia 1,14.
Although previous studies reported that animals receiving surgery in the first 24 hours following injury, as well as those that had surgery over a year after herniation, were more likely to have a poor outcome, recent studies have found no significant association between survival and the timing of hospitalization or surgery after the initial trauma 1,2. Because of this (and the risk of respiratory decompensation with progressive herniation of abdominal contents or dilation of a herniated stomach) the current recommendation is to perform surgery as soon as shock, hypoxia, hypovolemia, hemorrhage, pulmonary contusions and cardiac arrhythmias have been addressed sufficiently for the patient to be safely anesthetized.
Patients should be premedicated with an appropriate agent prior to induction of anesthesia. Individual protocols should be determined based on consideration of the patient to be anesthetized and the experience of the clinician, however commonly used agents include benzodiazepines and opioids 17. It is important to monitor patients closely following administration of premedicant agents, as sudden deterioration in respiratory function may occur following positional change. Pre-oxygenation prior to induction is strongly recommended, with provision of oxygen via a flow-by technique being typically better tolerated than use of a facemask. General anesthesia should be induced via a fast-acting, injectable technique and maintained via inhalational agents delivered in oxygen. Normal diaphragmatic function is required for spontaneous ventilation under general anesthesia, therefore intermittent positive pressure ventilation (IPPV) is required. Airway pressures should be maintained around 15cm H2O 18, and manometry is recommended to reduce the risk of inadvertent volutrauma.
Alistair Freeman
The patient should be positioned in dorsal recumbency and the ventral surface clipped and aseptically prepared from the mid-thorax to caudal to the pubis: this will allow a caudal sternotomy and/or caudal extension of the celiotomy if required. If possible, incline the operating table to raise the patient’s head and promote caudal displacement of the abdominal contents: this will increase exposure of the diaphragm, improve ventilation and facilitate reduction of herniated viscera 6.
A recommended list of equipment is given in Box 1. A ventral midline celiotomy is performed, from the xiphoid process to midway between the umbilicus and pubis. Median sternotomy is rarely indicated but may be required to increase exposure of the diaphragm and herniated viscera, either in chronic cases where thoracic adhesions have formed, or in cases with concurrent thoracic trauma. The falciform ligament should be excised and appropriate self-retaining retractors, such as Balfour or Gosset abdominal retractors (Figure 6) placed to improve visualization of the cranial abdomen. The cranial abdominal contents are retracted caudally and laterally (or exteriorized) to improve visualization of the diaphragm and to locate the hernia. Abdominal swabs moistened with sterile saline are helpful to protect organs during retraction, and the process is easier if a surgical assistant is present. Even if there is an obvious diaphragmatic defect on initial inspection, the entire diaphragm must be thoroughly explored, as bilateral or multiple tears frequently occur, and dorsal tears and those at the aortic hiatus are easily missed due to concealment by the liver 5.
Box 1. Equipment list for surgical repair of traumatic diaphragmatic hernia (TDH).
|

Figure 6. Use of Balfour self-retaining retractors will improve visualization of the cranial abdomen.
© Catrina Pennington
Gentle traction is applied to reduce any herniated viscera, using careful handling to avoid damage: the liver and spleen in particular may be engorged and friable. Some herniated viscera may not be reducible due to swelling or the formation of adhesions to the pleura; with this scenario, improved access to the caudal thorax can be obtained by incising the diaphragm to enlarge the hernia ring (avoiding transection of the phrenic nerves and trauma to any of the structures crossing the diaphragm) or performing a caudal sternotomy. Early fibrinous adhesions may be broken down by gentle digital manipulation: this minimizes the risk of hemorrhage. Adhesions that have been present for more than 1-2 weeks can be divided by sharp dissection, cautery or transection of the involved organ using an appropriate technique 19. After the organs are returned to the abdomen they should be inspected for rupture, necrosis, torsion or other injury, with damaged tissues repaired or resected as required. The thoracic cavity should be lavaged thoroughly prior to herniorrhaphy.
Debridement of the edges of the hernia ring is only required in chronic cases where adhesions between the diaphragm and herniated viscera are present. The defect should be closed using a synthetic non-absorbable monofilament suture in an interrupted or continuous pattern: the latter option minimizes the risk of injury to abdominal viscera from cut ends of suture. Preplacing the most dorsal sutures and starting suturing at the dorsal (deepest) end of the hernia can facilitate visualization. When closing tears involving the aortic or esophageal hiatus or caval foramen it is important to avoid narrowing these structures by careful placement of sutures and by taking smaller bites of tissue. Excessive tension is rarely a concern in repair of simple, acute tears; in more complex or chronic tears temporary approximation with stay sutures can facilitate closure. Very large defects may not be amenable to primary closure and may require supplementation with muscle or omental flaps, autogenous grafts and incorporation of synthetic implants 20,21,22,23,24,25.
Placement of a thoracostomy tube allows evacuation of the residual pneumothorax prior to the final diaphragmatic suture placement; the tube is connected to a three-way tap and air is removed via gentle suction until a light negative pressure is achieved (Figure 7). Alternatively insert a narrow gauge thoracostomy or feeding tube through the diaphragmatic defect prior to final suture tightening; the end should exit the celiotomy incision to allow postoperative drainage of pneumothorax if required.

Figure 7. A thoracostomy tube connected to a three-way tap will allow evacuation of residual pneumothorax post-surgery.
© Catrina Pennington
Attempts to eliminate pneumothorax by over-inflation of the lungs at closure may lead to intrapulmonary hemorrhage and pulmonary edema. Rapid re-expansion of the lungs increases the risk of reperfusion injury, so reinflation should therefore be gradual. In addition, excessive or prolonged expansion of the lungs following closure of the diaphragmatic defect increases intrathoracic pressure, which in turn increases resistance to venous return and leads to a fall in cardiac output. Atelectic lobes which do not reinflate following mechanical ventilation at physiological pressures should be addressed postoperatively with drainage via a thoracic catheter, aiming for a gentle negative pressure: gradual reinflation reduces the risk of reperfusion injury.
Finally, the abdomen should be lavaged with sterile saline before performing routine closure. Evacuate air from the abdomen by applying gentle pressure during closure of the linea alba to help restore the normal pleural-peritoneal pressure gradient.
Prior to anesthetic recovery, position the patient in both lateral and sternal recumbency and aspirate the thoracostomy tube until a gentle negative pressure is achieved. Maintain the tube until production falls below 0.5 mL/kg of fluid and 1 mL/kg of air over 24 hours 8.
The patient’s vital signs (respiratory rate and pattern, mucous membrane color, capillary refill time, rectal temperature) should be closely monitored postoperatively, with an objective pain scoring system used to ensure analgesia is adequate: this is particularly important as pain can cause hypoventilation and hypoxia secondary to reduced thoracic expansion. Intravenous fluid therapy should be maintained until voluntary intake resumes, and other supportive care (e.g., active warming and supplementary oxygen) should be given as required.
Up to 50% of cats will have complications following herniorrhaphy 1; these include surgical site infection, dehiscence of the surgical wound or herniorrhaphy site, delayed hemorrhage, cardiac dysrhythmias, organ rupture, cardiac arrest, pneumothorax and reinflation pulmonary edema 1,2,3,8,9,14. Pneumothorax due to iatrogenic trauma to the lungs during adhesion disruption, or secondary to reinflation injury, is a rare complication 14: mild cases may be self-resolving, but others may require lung lobectomy to address persistent leakage. Abdominal compartment syndrome is a concern in canine and human patients after herniorrhaphy but has not been documented in cats.
The most frequent causes of death in the postoperative period are cardiac arrest 1 and re-expansion pulmonary edema 8,9. Edema is thought to result from increased alveolar capillary permeability secondary to both pulmonary overinflation and inflammation due to reperfusion injury 26. Affected patients can be treated with oxygen supplementation, diuresis and bronchodilation, but the prognosis is guarded.
Between 85.3% and 100% of cats with TDH survive to discharge 1,2,3,8. Older age, lower respiratory rates on presentation, concurrent injury 1 and increased surgical/anesthetic duration 3 are associated with a poorer prognosis. Animals who are oxygen dependent at any time during hospitalization are 5 times more likely to die postoperatively, and those with concurrent soft tissue or orthopedic injuries are 7.3 times more likely to die. As previously discussed, timing of surgery has not been consistently associated with mortality, and reported survival rates after surgical correction of chronic hernias are up to 100% 14.
Catrina Pennington
Traumatic diaphragmatic hernia should be considered as a differential in any cat with a known or suspected traumatic injury, particularly those with respiratory pattern changes. Chronic cases may present with predominantly gastrointestinal signs, but careful thoracic auscultation may detect abnormalities in these animals. Radiography is the imaging modality of choice and is warranted in any cat presenting following trauma, or in animals with either increased or decreased respiratory sounds on thoracic auscultation. Surgery should be performed as soon as the patient is stabilized, with both surgeon and owner prepared for a period of intensive care following the procedure. However, the prognosis is generally good, with cardiac arrest and re-expansion pulmonary edema being the major causes of postoperative mortality.
Schmiedt CW, Tobias KM, Stevenson MA. Traumatic diaphragmatic hernia in cats: 34 cases (1991-2001). J. Am. Vet. Med. Assoc. 2003;222(9):1237-1240.
Gibson TW, Brisson BA, Sears W. Perioperative survival rates after surgery for diaphragmatic hernia in dogs and cats: 92 cases (1990-2002). J. Am. Vet. Med. Assoc. 2005;227(1):105-109.
Legallet C, Thieman Mankin K, Selmic LE. Prognostic indicators for perioperative survival after diaphragmatic herniorrhaphy in cats and dogs: 96 cases (2001-2013). BMC Vet. Res. 2017;13(1):16.
Evans H, de Lahunta A. Miller’s Anatomy of the Dog 4th ed. St Louis: Elsevier Saunders; 2013;250-252.
Caywood D. Non-cardiac surgical diseases of the thorax. Vet. Clin. North Am. Small Anim. Pract. 1987;17(2):255-270.
Rashid F, Chakrabarty MM, Singh R, et al. A review on delayed presentation of diaphragmatic rupture. World J. Emerg. Surg. 2009;4:32.
Sangster G, Ventura VP, Carbo A, et al. Diaphragmatic rupture: a frequently missed injury in blunt thoracoabdominal trauma patients. Emerg. Radiol. 2007;13(5):225-230.
Sullivan M, Reid J. Management of 60 cases of diaphragmatic rupture. J. Small Anim. Pract. 1990;31:425-430.
Garson HL, Dodman NH, Baker GJ. Diaphragmatic hernia; analysis of fifty-six cases in dogs and cats. J. Small Anim. Pract. 1980;21(9):469-481.
Cornell K, Jakovljevic S, Waters D, et al. Extrahepatic biliary obstruction secondary to diaphragmatic hernia in two cats. J. Am. Anim. Hosp. Assoc. 1993;29;502-507.
Formaggini L, Schmidt K, De Lorenzi D. Gastric dilatation-volvulus associated with diaphragmatic hernia in three cats: clinical presentation, surgical treatment and presumptive aetiology. J. Feline Med. Surg. 2008;10(2):198-201.
Bredal WP, Eggertsdottir AV, Austefjord O. Acute gastric dilatation in cats: a case series. Acta Vet. Scand. 1996;37(4):445-451.
Stork CK, Hamaide AJ, Schwedes C, et al. Hemiurothorax following diaphragmatic hernia and kidney prolapse in a cat. J. Feline Med. Surg. 2003;5(2):91-96.
Minihan AC, Berg J, Evans KL. Chronic diaphragmatic hernia in 34 dogs and 16 cats. J. Am. Anim. Hosp. Assoc. 2004;40(1):51-63.
Boudrieau S, Muir W. Pathophysiology of traumatic diaphragmatic hernia in dogs. Comp. Cont. Educ. Pract. Vet. 1987;9:379.
Sullivan M, Lee R. Radiological features of 80 cases of diaphragmatic rupture. J. Small Anim. Pract. 1989;30;561-566.
Wilson DV: Anesthesia for patients with diaphragmatic hernia and severe dyspnea. Vet. Clin. North. Am. Small Anim. Pract. 1992;22(2):456-459.
Dyson DH. Positive pressure ventilation during anesthesia in dogs: Assessment of surface area derived tidal volume. Can. Vet. J. 2012;53(1):63-66.
Henderson RA. Controlling peritoneal adhesions. Vet. Surg. 1982;11;30-33.
Rosenkrantz J, Cotton E. Replacement of left hemidiaphragm by a pedicled abdominal muscular flap. J. Thorac. Cardiovasc. Surg. 1964;48;912-920.
Touloukian R. A “new” diaphragm following prosthetic repair of experimental hemidiaphragmatic defects in the pup. Ann. Surg. 1978;187;47-51.
Schairer B, Keeley J. Experimental use of homologous fascia lata to repair diaphragmatic defects in dogs. Surg. Gynecol. Obstet. 1957;105;564-568.
Bright R, Thacker H. The formation of an omental pedicle flap and its experimental use in the repair of a diaphragmatic rent in the dog. J. Am. Anim. Hosp. Assoc. 1982;18;283-289.
Neville W, Clowes G. Congenital absence of hemidiaphragm and use of a lobe of liver in its surgical correction. Arch. Surg. 1954;69;282-290.
Furneaux R, Hudson M. Autogenous muscle flap repair of a diaphragmatic hernia. Feline Pract. 1976;6;20-24.
Sivrikoz MC, Tuncozgur B, Cekmen M, et al. The role of tissue reperfusion in the re-expansion injury of the lungs. Eur. J. Cardiothorac. Surg. 2002;22(5):721-727.
Alistair Freeman
Dr. Freeman graduated from the University of Edinburgh in 1994 and remained there to complete a Junior Clinical Training Scholarship Read more
Catrina Pennington
Dr. Pennington graduated from the University of Edinburgh in 2014 and initially worked in small animal practice before undertaking a rotating internship, followed by an orthopedic internship at a private Veterinary Specialist Hospital Read more
Lungworm and heartworm are an underestimated but potentially serious threat for many cats, as this article reveals.
Chylothorax is a differential for any cat with pleural effusion; here Elizabeth Rozanski describes her preferred approach to such cases.
Asthma is a common disease in cats, but it can mimic many other pathologies, and the diagnosis and treatment of the condition can be challenging, as this paper explains.
Lymphoma is one of the most common neoplasms of cats, and the mediastinal form can be a major differential for a cat that presents with respiratory signs, as James Elliott describes.
