Feline lungworm and heartworm
Lungworm and heartworm are an underestimated but potentially serious threat for many cats, as this article reveals.
This article contains sensitive photo that may be harmful for young children
Published 26/10/2022
Also available in Français , Deutsch , Italiano , Español and Українська
How do you deal with a cat with fainting episodes? Luca Ferasin offers a step-by-step guide for the clinician.
© Shutterstock

Syncopal events are characterized by rapid onset, short duration and a spontaneous and complete recovery, although transient disorientation may be observed for several minutes in some cases.
Syncope in cats is commonly associated with primary cardiovascular conditions, although some non-cardiovascular abnormalities can also be implicated.
The therapeutic approach to a cat with syncope depends largely on the underlying cause, but the transitory nature of syncopal episodes makes diagnosis a real challenge.
Many cats that experience syncope are misdiagnosed with epilepsy, and may undergo expensive – and often unnecessary – diagnostic investigations.
Syncope, or fainting, is defined as a sudden, unexpected, and unprovoked loss of consciousness followed by spontaneous recovery. Affected cats lose postural control and fall to the ground, remaining unresponsive throughout the event. However, syncope is a transient condition, usually only lasting for a few seconds, and is always followed by complete recovery. Syncopal events in people may be preceded by palpitations, dizziness, light-headedness, sweating, nausea, or even visual disturbances but, for obvious reasons, such premonitory signs cannot be communicated by pets and are therefore not reported in syncopal cats, although some pet carers will describe their cat vocalizing or “yowling” just prior to an event. A syncopal event can give the impression of sudden death, and carers who witness it are often deeply concerned, a situation comparable to the distress of parents when they see their children fainting, because they feel insecure and anxious as they are unable to help and assist their loved ones. Furthermore, cats with a history of syncope may be at increased risk of sudden death 1. Some cats may occasionally experience milder events and show signs of “pre-syncope” (or lipothymia), characterized by falling, weakness or unsteady gait without a complete loss of consciousness. Nevertheless, the diagnostic approach and clinical management of such cases should be the same as that for investigation and treatment of complete syncopal events.
Although several conditions can lead to syncope, this is ultimately the result of a transient global cerebral hypoperfusion. In people, irrespective of the precise underlying cause, a sudden cessation of cerebral blood flow for 6-8 seconds and/or a decrease in systolic blood pressure to 60 mmHg or less has been shown to be sufficient to cause complete loss of consciousness 2. Critical physiological values that can induce syncope in cats are not available, but it is not unrealistic to believe that they may be similar to those reported in people.
Feline syncope is often associated with primary cardiovascular conditions, although some non-cardiovascular abnormalities are also in the list of differential diagnoses. However, orthostatic, metabolic and psychological causes of syncope, which represent common causes of fainting in people, are not typically described in veterinary medicine. Iatrogenic arterial dilation may occur after administration of drugs that induce vasodilation, such as nitroprusside, acepromazine, or amlodipine. All the abnormalities listed above can potentially induce cerebral hypoperfusion and, ultimately, loss of consciousness. A summary of potential causes of syncope is reported in Table 1.
Table 1. Potential causes of syncope in cats.
| Cardiovascular |
|
| Vascular |
|
| Non-cardiovascular |
|
* - uncertain if this occurs in cats
In the veterinary literature, the reported cases of feline syncope have been associated with pathological variations of heart rate, such as prolonged periods of bradycardia due to sinus arrest 3 or paroxysmal atrio-ventricular (AV) block with ventricular standstill 4. Syncope in cats has also been reported with paroxysmal ventricular (VT) 5,6 or supraventricular (SVT) tachycardia 7. An episode of “tetanic seizure”, possibly associated with paroxysmal atrial tachycardia, has been described in a Persian cat, although a confirmative association between syncope and tachycardia was not demonstrated in this case 8. Other syncopal reports in cats have been associated with right outflow obstruction secondary to heartworm disease 9, increases in intra-thoracic and intra-abdominal pressure with subsequent reduced venous return during defecation (situational syncope) 10, aortic dissecting aneurysm associated with systemic arterial hypertension 11, and congenital cardiac defects 12. To the best of the author’s knowledge, confirmed cases of vasovagal (neurocardiogenic) syncope in cats, which is one of the most common causes of syncope in humans, have not been reported in the veterinary literature, although the author has documented a few cases during his clinical activity.
The brain needs a constant and adequate cerebral blood flow to function, and any significant reduction or interruption, even for a few seconds, can lead to syncope. Cerebral blood flow is maintained by various mechanisms affecting arterial blood pressure (BP), which is the result of cardiac output (CO) multiplied by the total peripheral resistance (TPR) (i.e., BP = CO x TPR). In turn, CO is determined by the volume of blood pumped by the heart (stroke volume, SV) per unit of time (heart rate, HR) (i.e., CO = SV x HR). Therefore, changes in either SV, HR or TPR will affect arterial blood pressure and, ultimately, cerebral perfusion.

Figure 1. The relationship between cardiac output (CO) and heart rate (HR) 26. The interval “N” represents physiological variations of heart rate (e.g., during exercise) where the cardiac output remains almost unchanged, as increased HR reduces the stroke volume (SV) in a harmonized manner (CO = SV x HR). However, when HR increases above the physiological range (interval “T” = tachycardia) the diastolic time decreases significantly and becomes insufficient to provide adequate ventricular filling and SV, consequently compromising the CO. Vice versa, when HR drops excessively (interval “B” = bradycardia), the ventricular filling, despite a prolonged diastolic time, does not increase proportionally due to the limited ventricular compliance, which compromises the SV and, eventually, CO.
© Luca Ferasin
Physiological variations in heart rate have little effect on CO. Indeed, when heart rate increases, the stroke volume decreases accordingly, since the diastolic interval is shortened, with decreased time available for ventricular filling and hence the volume of blood ejected by the heart on the following contraction. Conversely, a physiological reduction in heart rate is accompanied by a greater diastolic period and associated ventricular filling, causing increased SV. However, when changes in heart rate become pathological (i.e., tachycardia and bradycardia), CO drops significantly with both conditions. Indeed, rapid and sustained tachycardia can critically reduce diastolic time to the point that stroke volume becomes severely affected. Conversely, a profound bradycardia, despite the increased ventricular filling, can lead to a reduced CO (Figure 1). Examples of syncopal events induced by rapid tachycardia and profound bradycardia are reported in Figures 2 and 3 respectively.
 |
| a |
 |
| b |
 |
| c |
Figure 2. Nine-year-old male neutered Devon Rex cat referred with a long history of syncopal episodes, occurring approximately every 3-4 months. Resting and 24-hour Holter ECG recordings, thoracic radiographs and echocardiograms were inconclusive. An implantable loop recorder (a) was therefore surgically inserted subcutaneously on the left side of the chest to allow continuous ECG monitoring (b). A few weeks later the cat experienced another syncopal episode and the carer managed to trigger the patient activator device. The ECG trace during the syncopal event showed rapid supraventricular tachycardia (SVT) at a rate of 430 bpm (black arrowhead) (c). The cat was treated with sotalol (10 mg PO BID) and did not experience any further fainting episodes.
© Luca Ferasin

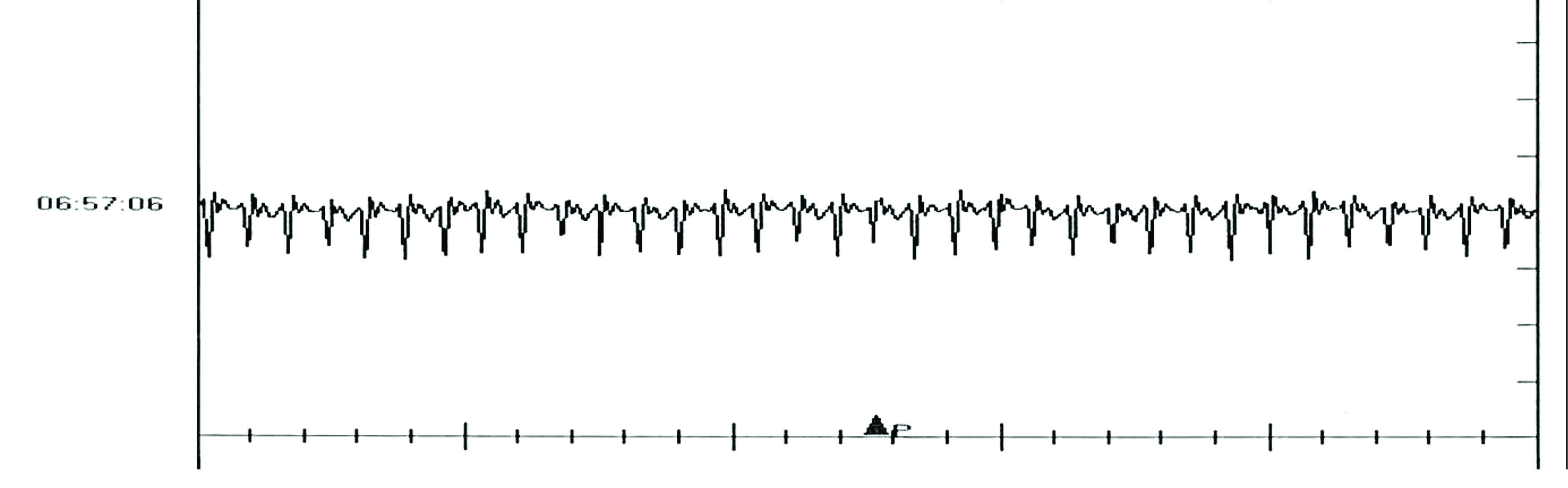
Figure 3a. Eleven-year-old female neutered Domestic Shorthair cat, initially referred for neurological work-up following onset of daily episodes of tremors, facial twitching and fainting. Prolonged cardiac pauses were detected during clinical examination, which prompted an immediate cardiac workup; echocardiography, thoracic radiography and resting ECG were unremarkable, but 24-hour Holter monitoring showed predominant sinus rhythm alternating with periods of atrio-ventricular (AV) dissociation, with an average daily rate of 93 bpm. The falling and twitching episodes coincided with periods of complete AV block with ventricular standstill, the longest period lasting approximately 8 seconds.
© Luca Ferasin

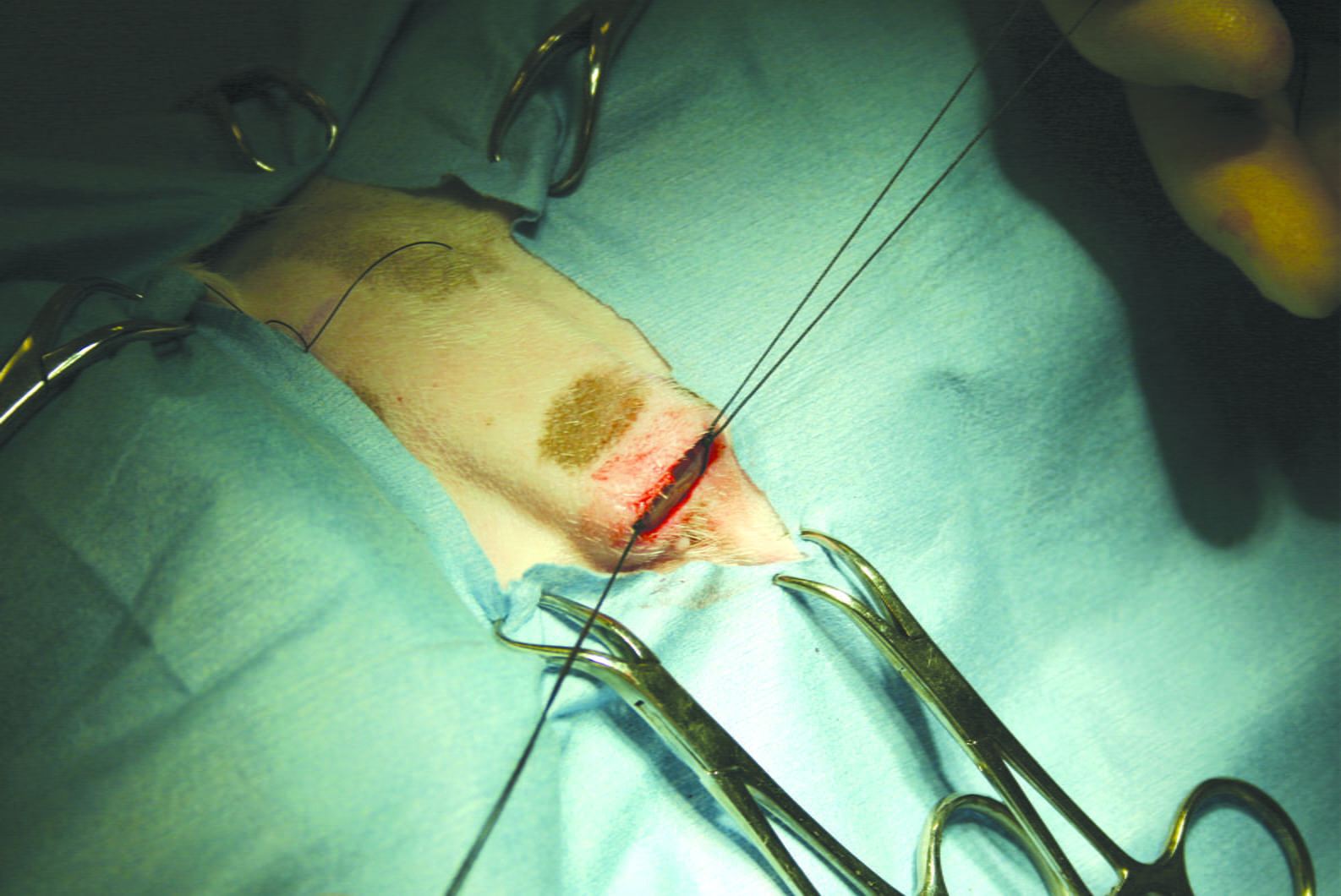
Figure 3b. A permanent pacemaker was surgically implanted, with a lead sutured to the left ventricular epicardium.
© Luca Ferasin

Figure 3c. The epicardial lead was tunneled under the lateral thoracic wall muscles and the pulse generator was inserted in a subcutaneous pocket created over the left cranioventral abdominal wall.
© Luca Ferasin

Figure 3d. Syncopal episodes did not represent following the procedure and device interrogation, performed six months later, revealed that the pacemaker was intervening 24% of the time to avoid cardiac pauses and associated fainting episodes.
© Luca Ferasin
The pathophysiological mechanism of syncope secondary to outflow obstruction is rather intuitive, since any clinical condition that significantly reduces SV will cause a drop in CO. Congenital abnormalities, such as aortic and pulmonic stenosis, can potentially cause severe left and right outflow obstruction respectively, often accompanied by exercise intolerance and syncopal events. However, the most common form of outflow obstructions in cats is represented by dynamic left ventricular outflow tract obstruction secondary to systolic anterior motion (SAM) of the mitral valve, which is frequently observed with hypertrophic cardiomyopathy (HCM) 13, although this abnormality can also be observed in the absence of left ventricular hypertrophy (LVH) 14. However, syncope secondary to SAM is seldom observed in cats, unlike in humans where fainting episodes are commonly observed in severe SAM, with or without concomitant LVH 15,16. Dynamic right ventricular outflow tract obstruction (DRVOTO) is another common finding in feline patients, although this condition appears to be completely benign in this species 17,18 and not associated with fainting. Syncope secondary to outflow obstruction in cats has also been observed in cases of heartworm disease 9 and severe aortic stenosis (Figure 4).
 |
| a |
 |
| b |
 |
| c |
 |
| d |
 |
| e |
 |
| f |
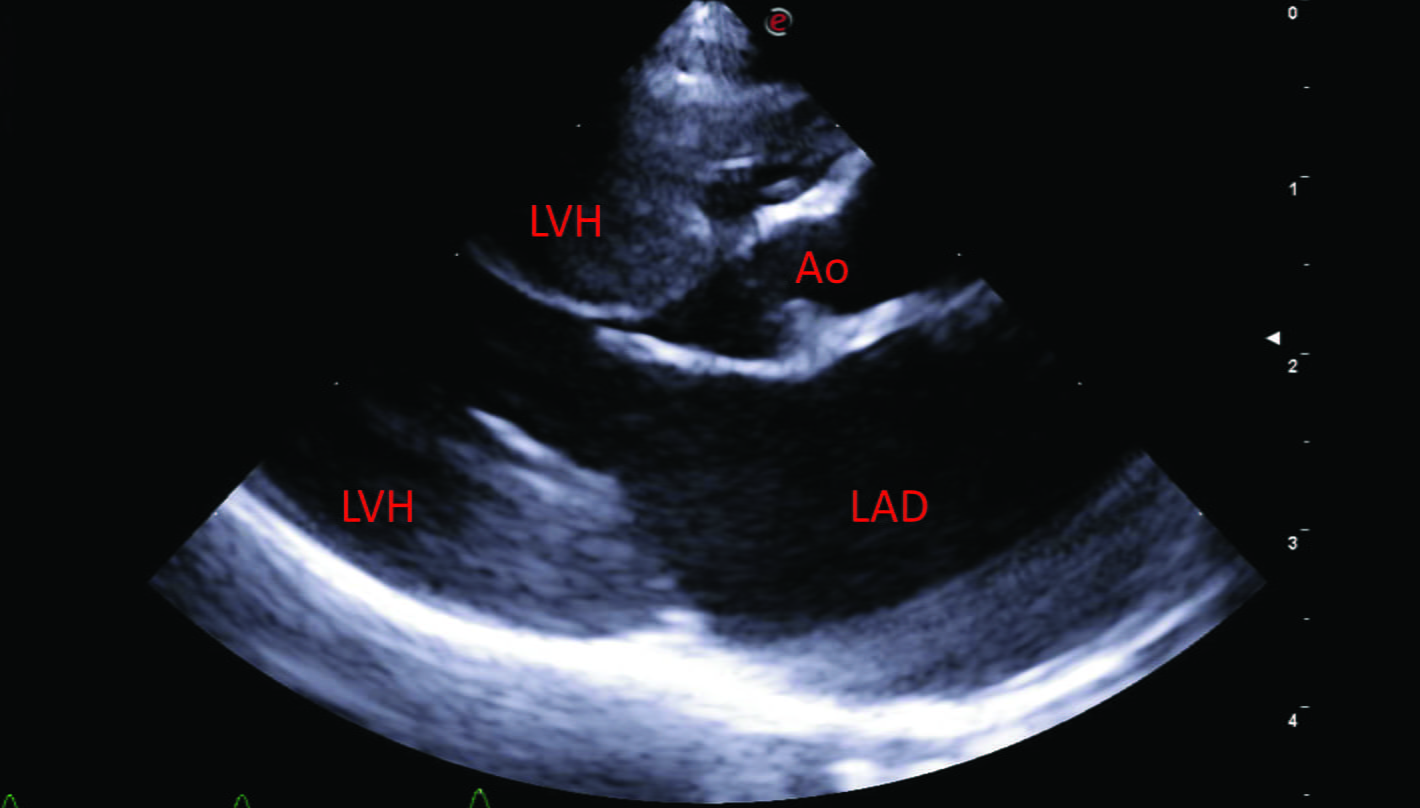
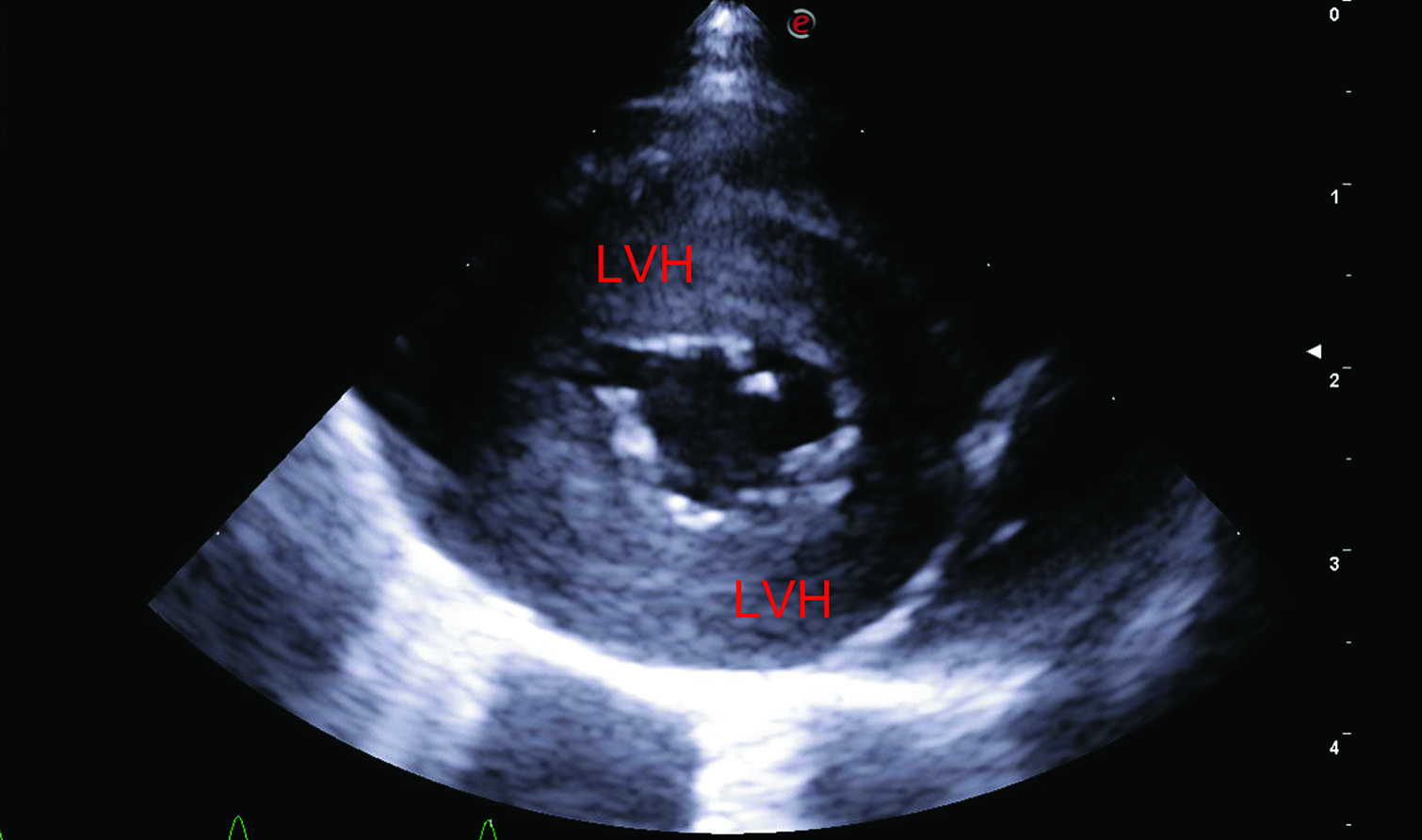
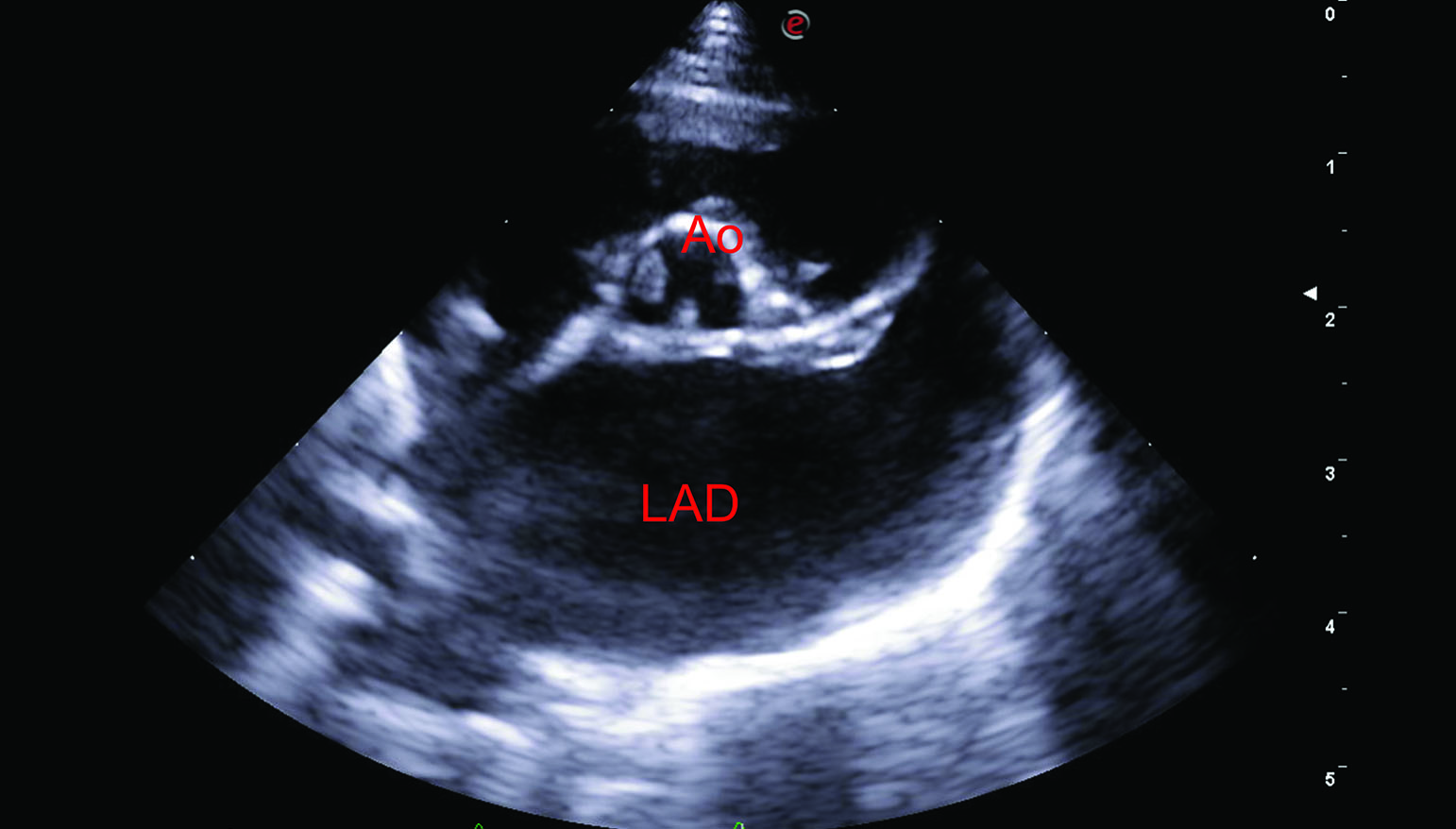
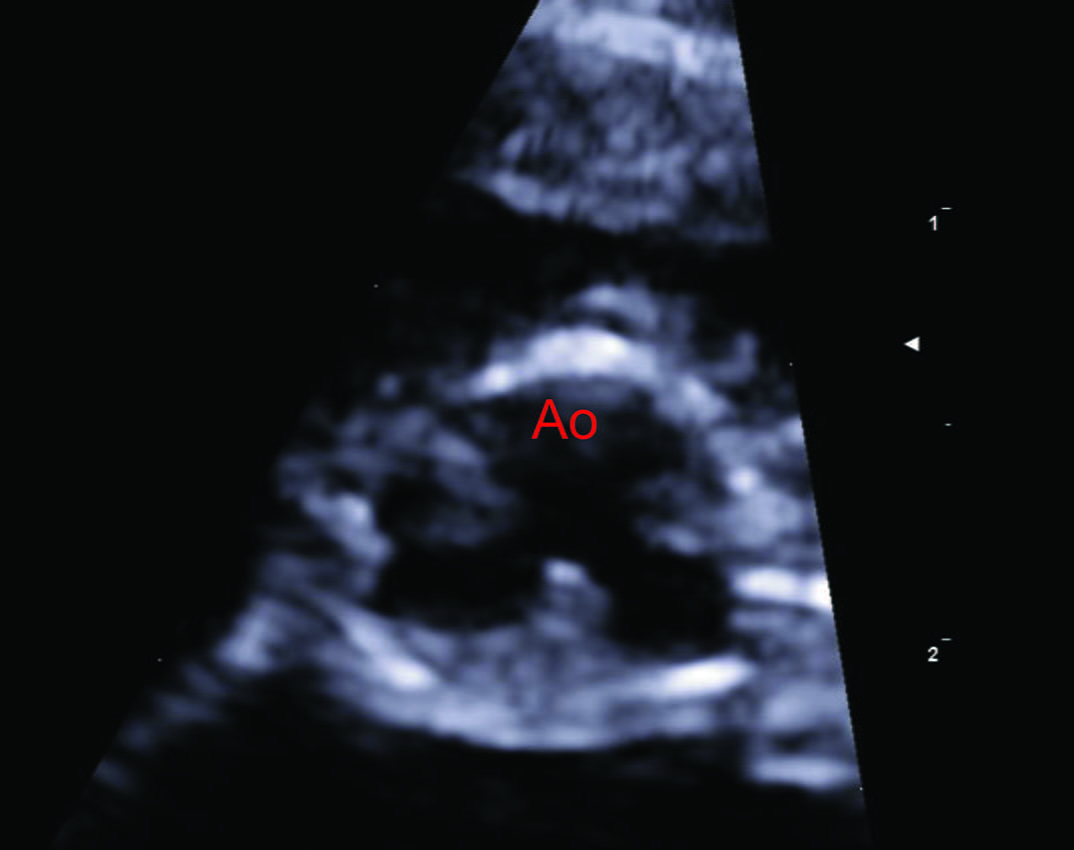
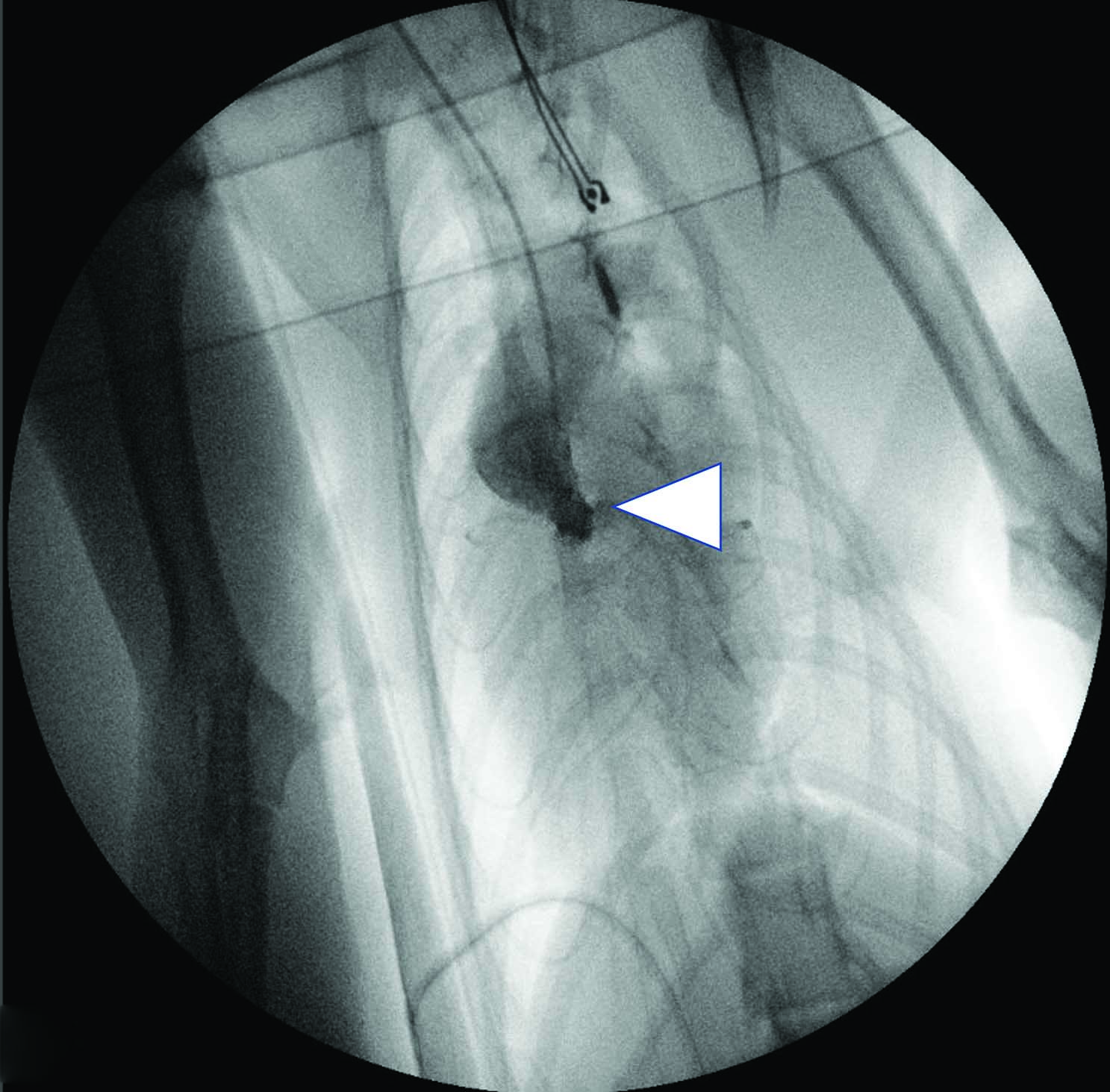
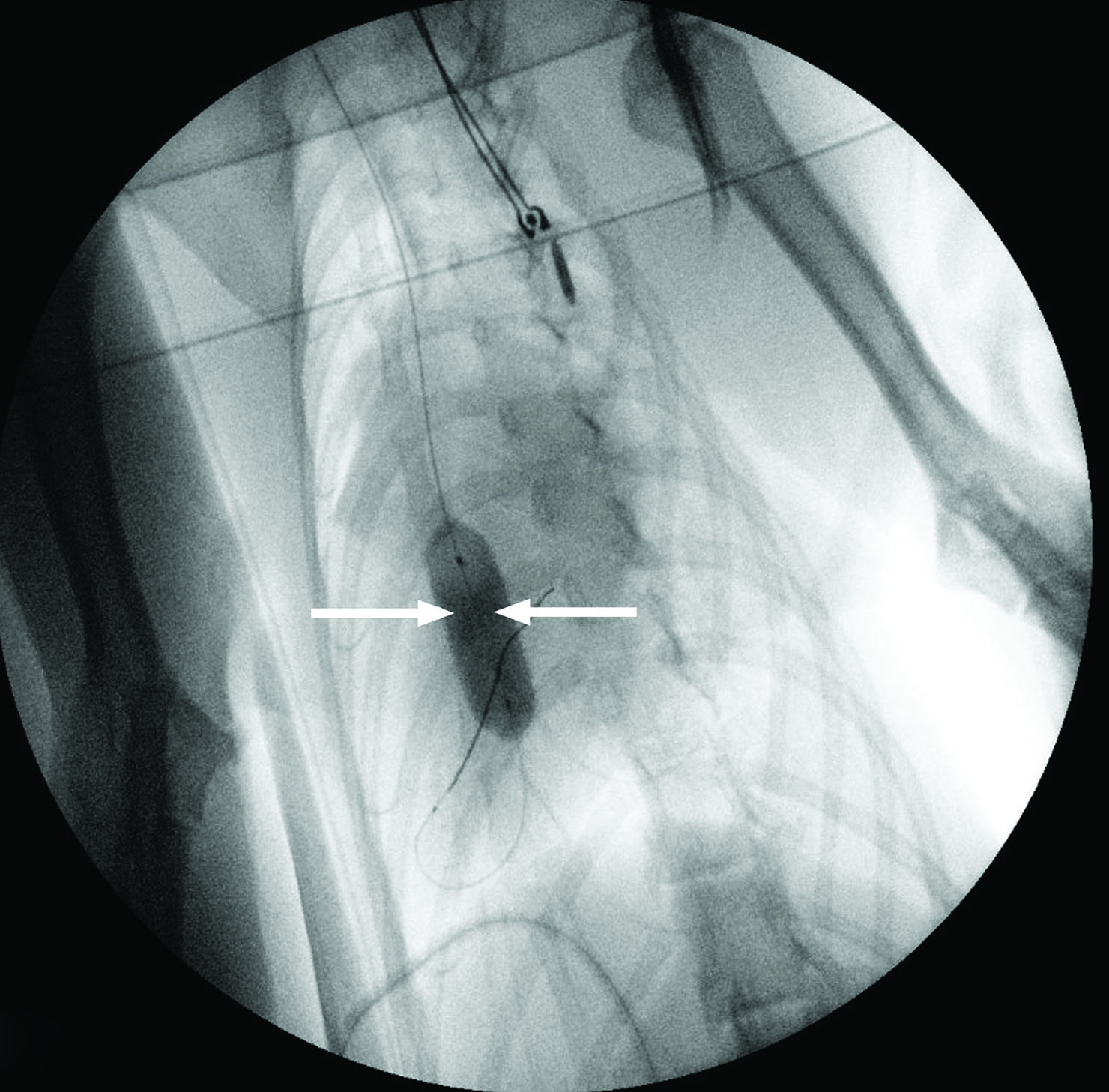
Figure 4. Three-year-old female Bengal cat with a history of syncopal episodes following excitement during playing. The cat was diagnosed with a severe form of valvular aortic stenosis (pressure gradient 118 mmHg) causing left ventricular concentric hypertrophy and left-sided congestive heart failure. Echocardiographic images were obtained from right parasternal views in long axis view 5-chamber (a), short axis at the level of the papillary muscles (b), and short axis at the level of the heart base (c and d). This showed significant left ventricular concentric hypertrophy (LVH), left atrial dilation (LAD) and thickened and partially fused aortic valve cusps (Ao), which are responsible for the stenotic lesion. Images E and F were obtained on fluoroscopic guidance during balloon valvuloplasty to palliate the magnitude of the aortic stenosis. The stenosis is highlighted during selective angiography (e, white arrowhead), while the inflated balloon (f, white arrows) indicates the successful dilation of the stenotic valve.
© Luca Ferasin
Myocardial disease, such as HCM and arrhythmogenic right ventricular cardiomyopathy (ARVC), can cause cardiac syncope both in people and cats 1,13,19. Although cardiac arrhythmias may play a pivotal role in the onset of syncope in myocardial disease, myocardial failure should also be taken into consideration. In HCM for example, the diminished left ventricular filling volume and impaired contractility secondary to myocardial hypertrophy, disorganization of the myocyte fibers, myocardial scarring, and fibrosis can severely affect CO, resulting in diminished blood flow to intramural coronary vessels (and the rest of the body), causing intramural myocardial infarctions. In ARVC, the presence of fibro-fatty replacement of the right ventricle can cause right ventricular dilation and ventricular aneurysm, with significant systolic dysfunction and consequent drop in CO. Similarly, all end-stage forms of cardiac disease will eventually be accompanied by myocardial dysfunction, reduced cardiac contractility and ultimately a decline in CO.
This is the most common type of syncope in people, comprising approximately 45% of cases 20, but it is relatively uncommon in cats. It can be vasovagal, situational, or secondary to carotid sinus hypersensitivity. The pathophysiology of neurally mediated syncope is rather complex, and originates from an increased sympathetic tone, which can sensitize and facilitate the activation of cardiac mechanoreceptors that occurs with very vigorous ventricular contraction or by C-fibers activation secondary to myocardial ischemia and reperfusion. The activation of these receptors triggers an abrupt centrally mediated reduction in sympathetic activity, with concomitant parasympathetic activation, resulting in vasodilation and bradycardia and, ultimately, a rapid and profound hypotension 21. An example of vasovagal syncope in a cat is shown in Figure 5.
Situational syncope has been reported in a cat following defecation 10. The mechanism is likely associated with the increased intrathoracic and intrabdominal pressure during defecation, which tends to reduce venous return and subsequently SV and CO. This cat had a history of straining prior to fainting, which may have activated tension receptors in the intestinal wall, resulting in sudden hypotension and bradycardia. In people, situational syncope is also observed during or immediately after urination, defecation, coughing or swallowing, with a mechanism similar to the one explained above.
 |
| a |
 |
| b |
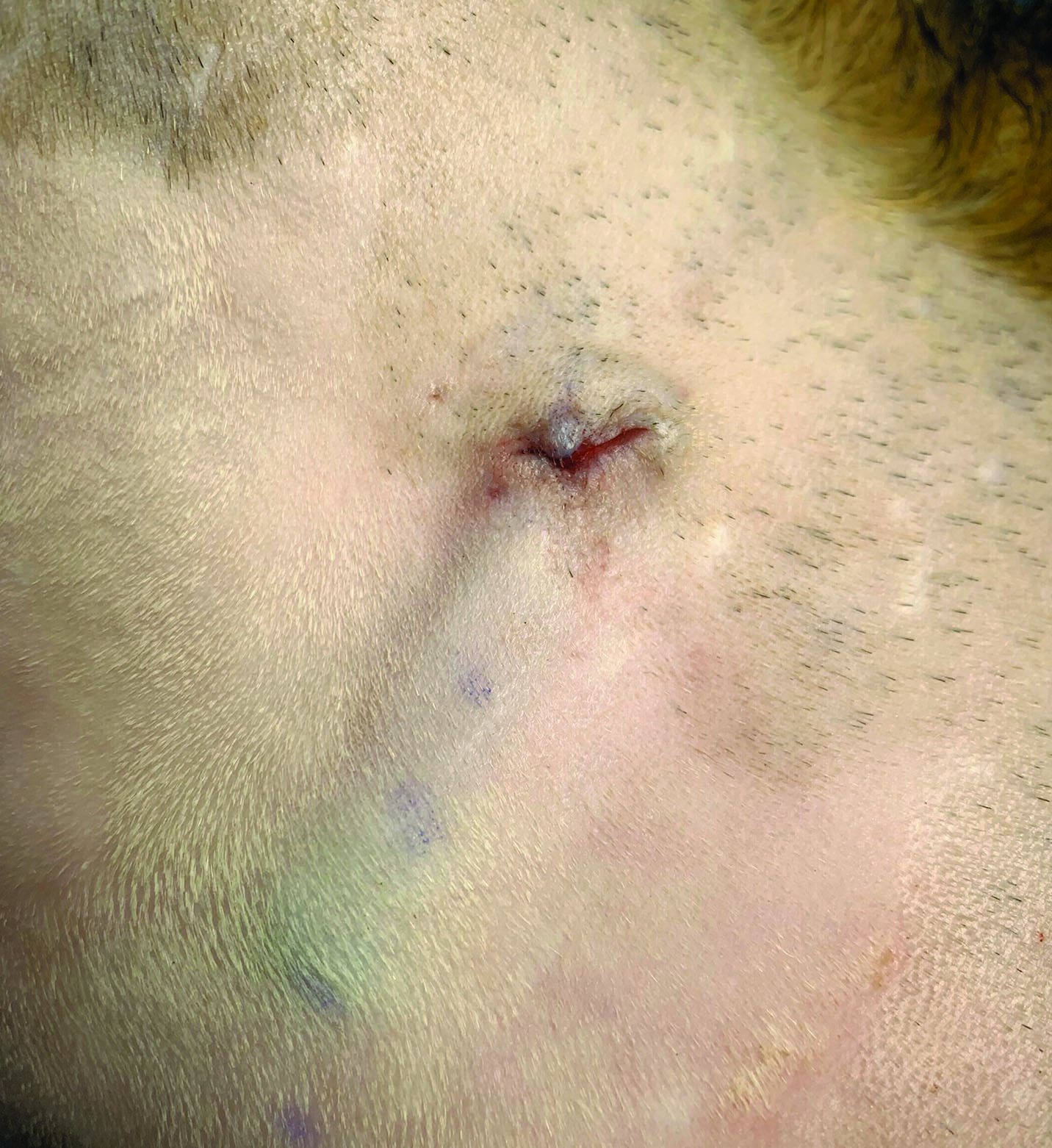
Figures 5a and b. Five-year-old male neutered Domestic Shorthair cat with syncopal episodes observed following periods of stress or excitement, such as fighting with other cats in the garden. The cat was experiencing episodes approximately every 3-4 weeks and was initially referred for a neurological assessment, which was unremarkable. A cardiology workup was therefore requested. Echocardiography was unremarkable, with the exception of a significant dynamic left ventricular outflow obstruction (LVOTO) caused by systolic anterior motion (SAM) of the mitral valve. Standard ECG recording showed normal sinus rhythm with a heart rate of 180 bpm, and 24-hour Holter recording failed to identify any abnormalities, revealing only normal sinus rhythm with an average daily rate of 166 bpm (range 88-244 bpm). However, no syncopal events occurred during the recording period. Empiric treatment with atenolol (6.25 mg PO SID) showed no significant response, so the dose was increased to 6.25mg PO BID, which resulted in an even more frequent occurrence of fainting episodes. An implantable loop recorder (ILR) was then surgically inserted subcutaneously on the left side of the chest to allow continuous ECG monitoring.
© Luca Ferasin

Figure 5c. The device functionality was tested during the procedure using a pacemaker programmer and dedicated patient activator device.
© Luca Ferasin

Figure 5d. ILR interrogation following another fainting episode showed a period of tachycardia during a cat fight, followed by a sudden drop-in heart rate, from approximately 300 bpm (“Cat Fight”) to 100 bpm (“Fainting”). The carers heard the fight and, finding their cat unconscious on the ground, immediately activated the patient activator device (“Detected”), which saved the ECG recording throughout the event. The tachogram (RR interval time series) from the ECG is compatible with a vasovagal (neurocardiogenic) syncope. The carer was advised to discontinue atenolol and to avoid stressful events, such as interaction with other cats, which resulted in complete disappearance of the syncopal episodes.
© Luca Ferasin
The transitory nature of episodes and spontaneous resolution of clinical signs represent a real diagnostic challenge. In humans with no abnormalities reported on the clinical history or physical examination, diagnosis may not be achieved in around 50% of cases 22, and similar figures are reported in small animal practice. An excellent understanding of the efficacy and utility of the investigational tools for syncope is therefore required in order to institute appropriate management promptly and efficiently. In fact, a wrong choice of sequential tests may result in poor diagnostic yield and unnecessary costs. Furthermore, a delayed diagnosis may result in sudden death 23. Collecting the patient’s history in a thorough and consistent manner is fundamental, and appropriate questions should be formulated to understand whether or not the patient is experiencing syncopal events. Indeed, cat carers will often only report a vague description of “collapsing” events, so the clinician should determine if the reported episodes are accompanied by transient loss of consciousness. Syncopal events should also be characterized by rapid onset, short duration (typically no longer than 20 seconds) and followed by a spontaneous and complete recovery, although transient disorientation may be observed for several minutes in some cases. Information obtained from carers should include any history of cardiac disease or medications that may induce arrhythmias or hypotension, the number and frequency of the episodes (in full detail), identification of precipitating or trigger factors, including a detailed description of physical activity, time of the day, environmental conditions, degree of excitement, etc., prior to or at the time of the episode. It is important to accurately quantify the type and duration of the event and obtain information (when available) about the color of mucous membranes and heartbeat during the episodes. Lethargy, disorientation or behavioral changes following a collapse, frothing at the mouth, unconsciousness lasting for more than a few minutes, or an absence of pale or cyanotic mucosa would suggest seizures or a neurological etiology rather than syncope. Nevertheless, patients experiencing prolonged syncopal events may also lose sphincter control, with subsequent involuntary defecation and/or urination, and can also display tonic-clonic activity and opisthotonos, mimicking seizures 24. For this reason, many cats experiencing syncope are misdiagnosed with epilepsy and undergo expensive and often unnecessary diagnostic investigations, sometimes involving general anesthesia. Therefore, it is important to understand what happens before, during and after a falling event in order to distinguish, as much as possible, between these two similar presentations. In such cases an owner’s detailed diary of events and video recording of falling episodes, even using a mobile phone camera, can be extremely helpful.
A full physical examination, with a particular emphasis on the assessment of the neurological and cardiovascular systems, is probably the most important component of the evaluation of a patient with suspected syncope 25. Physical examination should be conducted in a thorough and consistent manner, paying particular attention to assessment of the cardiovascular system (including blood pressure measurement), level of hydration, and presence of concomitant neurological or musculoskeletal abnormalities. Any cardiac arrhythmia, murmur, or gallop sounds would be highly suggestive of an underlying heart abnormality, and should justify a complete cardiac evaluation.
Laboratory tests (basic hematology and serum biochemistry) have a low diagnostic yield, although they are important to rule out severe anemia, dehydration, electrolyte imbalances and musculoskeletal lesions. Measurement of cardiac troponin I may reveal elevations consistent with an underlying myocardial insult, including ischemia and myocarditis. Elevated thyroxine (T4) would suggest hyperthyroidism, which can potentially cause cardiotoxicity.
Echocardiography should be considered if there are abnormal cardiac findings during the physical examination, or where there is a familial history/predisposition of cardiac disease. However, the majority of fainting cats have unremarkable echocardiographic findings. Resting electrocardiography (ECG) identifies the reason for fainting in only in a small percentage of patients. Indeed, sporadic syncope may reflect the episodic nature of an underlying ECG abnormality, which is often missed even on a five-minute continuous recording, therefore prolonged ECG monitoring can represent a more successful diagnostic tool. Nevertheless, even 24/48-hour Holter recording has a relatively low diagnostic value, especially when the falling episodes occur occasionally and can be missed during the recording. The only exception for justifying Holter monitoring is for fainting episodes occurring several times a week 19. Alternatively, 7/14 day monitoring, which is available only with a limited number of recorders, has a higher chance of achieving a conclusive diagnosis 25, but diagnostic yield increases dramatically if an implantable loop recorder (ILR) is used. Reliability and accuracy of ILRs has been reported in the veterinary literature with excellent results 7,23; for cats with unexplained syncopal episodes, the manually activated ILR can provide invaluable diagnostic and prognostic information by either confirming or disproving the association between syncope and arrhythmias or conduction abnormalities. If a syncopal event is observed and the tests listed above (including ILR) are inconclusive or not suggestive of a cardiac etiology, then other investigations should be considered, including abdominal ultrasound (to rule out internal bleeding), advanced imaging (MRI, CT), electromyography (EM), electroencephalography (EEG), and cerebrospinal fluid (CSF) collection.
Luca Ferasin
Treatment of a cat with syncope has three main goals: avoiding sudden death and prolonging survival, avoiding traumatic injuries sustained during a falling event, and preventing further syncopal events. The therapeutic approach depends largely on the cause and mechanism of the syncope. If a definitive diagnosis has not yet been achieved, carers should be instructed to keep their cat indoors to avoid potential trauma that may occur if the pet is left unsupervised. However, if a diagnosis has been obtained, therapy should be aimed at controlling the underlying primary condition responsible for the transient loss of consciousness. For example, treatment of sustained or paroxysmal rapid ventricular and supraventricular tachycardia can be attempted with anti-arrhythmic medications such as oral atenolol and sotalol. Diltiazem has no effect on ventricular arrhythmias but can be considered for supraventricular arrhythmias. However, all these drugs can also significantly decrease resting heart rate and affect stroke volume and cardiac output, potentially precipitating congestive heart failure in patients with underlying myocardial disorders. Therefore, it is important to consider 24-hour Holter recording a few days after starting anti-arrhythmic therapy in order to monitor the appropriate response to treatment and possible residual presence of paroxysmal tachycardias.
The appropriate treatment for most cats with syncope associated with bradycardia would be a permanent pacemaker implant. Although transvenous pacing lead placement into the right ventricular apex is the most common method of pacemaker implantation in dogs, cats appear predisposed to complications, such as cranial vena cava syndrome with chylothorax, right ventricular outflow tract obstruction, and intracardiac thrombosis 4,26. Therefore, surgical epicardial lead placement via thoracotomy is currently the most commonly adopted technique for pacemaker implantation in cats, and is generally successful with few minor complications.
Fixed outflow obstructions, such as aortic and pulmonic stenosis, can be palliated with balloon valvuloplasty, a minimally invasive intervention using a catheter with a balloon on its tip that can be inflated at the level of the stenotic valve to stretch and open the partially fused cusps. Dynamic outflow obstructions are very rarely associated with syncope in cats, but if a cause-effect relationship can be documented, therapy with beta blockers (e.g., atenolol) can be attempted to reduce the magnitude of the obstruction.
Therapy for neurally mediated syncope is seldom necessary. A careful history collection with attention focused on identifying precipitating factors should reveal the triggering events (e.g., stress, excitement), and avoidance of these circumstances is usually sufficient to drastically reduce the frequency and severity of fainting episodes. Situational syncope can be successfully controlled by addressing and eliminating, when possible, the underlying condition (e.g., coughing or straining during micturition and defecation). Pacemaker implant represents a controversial solution for neurally mediated syncope, but may be considered if the fainting event is caused by very long cardiac pauses. The major limitation here is that the pacemaker cannot control the systemic hypotension associated with the sudden vagally induced vasodilation, so it should be programmed with specialized pacing algorithms such as heart rate hysteresis; this allows the pacemaker to start ventricular pacing only if the spontaneous heart rate falls below a critical level, whereby pacing continues at a selected higher rate unless intrinsic ventricular activity is sensed. The intention is that when an event occurs, the higher rate of artificial pacing may compensate for the hypotension caused by the sudden reduction in total peripheral resistance.
A cat which presents with episodes of falling/fainting can be a diagnostic challenge, and a logical approach to history-taking, examination and diagnostic testing is essential. The majority of cats with syncope will have a primary cardiac etiology, although other causes can and do exist. Ultimately therapy should be aimed at prolonging survival (and hence averting sudden death), avoiding injuries from falling, and preventing further syncopal events, but the therapeutic approach depends largely on the cause and mechanism of the syncope. It is, however, vital not to misdiagnose syncope for seizures, which requires a completely different approach and may carry a different prognosis.
Acknowledgement
The author would like to thank Dr. Heidi Ferasin for her assistance with management of some of the reported clinical cases and for her invaluable assistance in writing this manuscript. A special thank you also goes to Dr. Daniella McCready who performed the surgical pacemaker implant described in Figure 3.
Payne JR, Borgeat K, Brodbelt DC, et al. Risk factors associated with sudden death vs. congestive heart failure or arterial thromboembolism in cats with hypertrophic cardiomyopathy. J. Vet. Cardiol. 2015;17 Suppl 1:S318-328. DOI: 10.1016/j.jvc.2015.09.008. PubMed PMID: 26776589.
Brignole M. Distinguishing syncopal from non-syncopal causes of fall in older people. Age Ageing 2006;35 Suppl 2:ii46-ii50. DOI: 10.1093/ageing/afl086. PubMed PMID: 16926204.
Meurs KM, Miller MW, Mackie JR, et al. Syncope associated with cardiac lymphoma in a cat. J. Am. Anim. Hosp. Assoc. 1994;30:583-585.
Ferasin L, van de Stad M, Rudorf H, et al. Syncope associated with paroxysmal atrioventricular block and ventricular standstill in a cat. J. Small Anim. Pract. 2002;43(3):124-128. DOI: 10.1111/j.1748-5827.2002.tb00042.x. PubMed PMID: 11916056.
Harvey AM, Battersby IA, Faena M, et al. Arrhythmogenic right ventricular cardiomyopathy in two cats. J. Small Anim. Pract. 2005;46(3):151-156. DOI: 10.1111/j.1748-5827.2005.tb00306.x. PubMed PMID: 15789811.
Lee S, Kittleson MD. Naturally occurring torsades de pointes and QT interval prolongation in a domestic cat. J. Vet. Cardiol. 2021;35:42-47. Epub 20210303. DOI: 10.1016/j.jvc.2021.02.006. PubMed PMID: 33812132.
Ferasin L. Recurrent syncope associated with paroxysmal supraventricular tachycardia in a Devon Rex cat diagnosed by implantable loop recorder. J. Feline Med. Surg. 2009;11(2):149-152. Epub 20080703. DOI: 10.1016/j.jfms.2008.04.006. PubMed PMID: 18602324.
Harwood ML. Paroxysmal atrial tachycardia and left superior bundle branch blockage in a cat. Vet. Med. Small Anim. Clin. 1970;65(9):862-866. PubMed PMID: 5201514.
Malik R, Church DB, Eade IG. Syncope in a cat. Aust. Vet. J. 1998;76(7):465, 70-71. DOI: 10.1111/j.1751-0813.1998.tb10180.x. PubMed PMID: 9700397.
Whitley NT, Stepien RL. Defaecation syncope and pulmonary thromboembolism in a cat. Aust. Vet. J. 2001;79(6):403-405. DOI: 10.1111/j.1751-0813.2001.tb12982.x. PubMed PMID: 11491217.
Gouni V, Papageorgiou S, Debeaupuits J, et al. Aortic dissecting aneurysm associated with systemic arterial hypertension in a cat. Schweiz Arch. Tierheilkd. 2018;160(5):320-324. DOI: 10.17236/sat00162. PubMed PMID: 29717986.
Hsueh T, Yang CC, Lin SL, et al. Symptomatic partial anomalous pulmonary venous connection in a kitten. J. Vet. Intern. Med. 2020;34(6):2677-2681. Epub 20201016. DOI: 10.1111/jvim.15934. PubMed PMID: 33063892; PubMed Central PMCID: PMC7694797.
Ferasin L. Feline myocardial disease. 1: Classification, pathophysiology and clinical presentation. J. Feline Med. Surg. 2009;11(1):3-13. DOI: 10.1016/j.jfms.2008.11.008. PubMed PMID: 19154970.
Ferasin L, Kilkenny E, Ferasin H. Evaluation of N-terminal prohormone of brain natriuretic peptide and cardiac troponin-I levels in cats with systolic anterior motion of the mitral valve in the absence of left ventricular hypertrophy. J. Vet. Cardiol. 2020;30:23-31. Epub 20200518. DOI: 10.1016/j.jvc.2020.05.001. PubMed PMID: 32645686.
Hodges K, Rivas CG, Aguilera J, et al. Surgical management of left ventricular outflow tract obstruction in a specialized hypertrophic obstructive cardiomyopathy center. J. Thorac. Cardiovasc. Surg. 2019;157(6):2289-2299. Epub 20181229. DOI: 10.1016/j.jtcvs.2018.11.148. PubMed PMID: 30782406.
Sabzwari SRA, Kimber JR, Ayele H, et al. The disappearing murmur: systolic anterior motion of the mitral valve leaflet in a non-hypertrophic cardiomyopathy patient. Cureus. 2018;10(6):e2855. Epub 20180621. DOI: 10.7759/cureus.2855. PubMed PMID: 30148008; PubMed Central PMCID: PMC6104906.
Ferasin L, Ferasin H, Kilkenny E. Heart murmurs in apparently healthy cats caused by iatrogenic dynamic right ventricular outflow tract obstruction. J. Vet. Intern. Med. 2020;34(3):1102-1107. Epub 20200428. DOI: 10.1111/jvim.15774. PubMed PMID: 32343450; PubMed Central PMCID: PMC7255668.
Rishniw M, Thomas WP. Dynamic right ventricular outflow obstruction: a new cause of systolic murmurs in cats. J. Vet. Intern. Med. 2002;16(5):547-552. DOI: 10.1892/0891-6640(2002)016<0547:drvooa>2.3.co;2. PubMed PMID: 12322704.
Ferasin L, Ferasin H, Borgeat K. Twenty-four-hour ambulatory (Holter) electrocardiographic findings in 13 cats with non-hypertrophic cardiomyopathy. Vet. J. 2020;264:105537. Epub 20200906. DOI: 10.1016/j.tvjl.2020.105537. PubMed PMID: 33012440.
Runser LA, Gauer RL, Houser A. Syncope: evaluation and differential diagnosis. Am. Fam. Physician 2017;95(5):303-312. PubMed PMID: 28290647.
Grubb BP. Pathophysiology and differential diagnosis of neurocardiogenic syncope. Am. J. Cardiol. 1999;84(8A):3Q-9Q. DOI: 10.1016/s0002-9149(99)00691-8. PubMed PMID: 10568555.
Gould PA, Krahn AD, Klein GJ, et al. Investigating syncope: a review. Curr. Opin. Cardiol. 2006;21(1):34-41. DOI: 10.1097/01.hco.0000198986.16626.86. PubMed PMID: 16355027.
Willis R, McLeod K, Cusack J, et al. Use of an implantable loop recorder to investigate syncope in a cat. J. Small Anim. Pract. 2003;44(4):181-183. DOI: 10.1111/j.1748-5827.2003.tb00142.x. PubMed PMID: 12703871.
Schriefl S, Steinberg TA, Matiasek K, et al. Etiologic classification of seizures, signalment, clinical signs, and outcome in cats with seizure disorders: 91 cases (2000-2004). J. Am. Vet. Med. Assoc. 2008;233(10):1591-1597. DOI: 10.2460/javma.233.10.1591. PubMed PMID: 19014293.
Bright JM, Cali JV. Clinical usefulness of cardiac event recording in dogs and cats examined because of syncope, episodic collapse, or intermittent weakness: 60 cases (1997-1999). J. Am. Vet. Med. Assoc. 2000;216(7):1110-1114. DOI: 10.2460/javma.2000.216.1110. PubMed PMID: 10754673.
Frantz EW, Tjostheim SS, Palumbo A, et al. A retrospective evaluation of the indications, complications, and outcomes associated with epicardial pacemakers in 20 cats from a single institution. J. Vet. Cardiol. 2021;36:89-98. Epub 20210513. DOI: 10.1016/j.jvc.2021.05.001. PubMed PMID: 34118563.
Luca Ferasin
Dr. Ferasin graduated with honors in 1992 from the University of Bologna and spent three years doing endocrinology research at the BBSRC Institute in Cambridge, gaining his PhD in 1996 Read more
Lungworm and heartworm are an underestimated but potentially serious threat for many cats, as this article reveals.
Diaphragmatic hernia is a common consequence of traumatic injury in cats and may be fatal if not promptly diagnosed and treated; this article reviews the pathophysiology and treatment of the condition.
Chylothorax is a differential for any cat with pleural effusion; here Elizabeth Rozanski describes her preferred approach to such cases.
Asthma is a common disease in cats, but it can mimic many other pathologies, and the diagnosis and treatment of the condition can be challenging, as this paper explains.
