Veterinary internships in the USA
Thinking about doing an internship in the USA? This article delivers a brief guide to the advantages and disadvantages as to what one can offer.
The cardinal sign of acute heart failure in dogs is tachypnea and/or dyspnea, although various other abnormalities may also be present.
Three immediate steps – namely minimizing stress, oxygen supplementation, and sedation – should be considered whenever a dog presents with suspected acute heart failure.
Successful diuretic therapy should provide rapid and complete relief from the effects of pulmonary edema, and must then be continued to prevent it from recurring.
Respiratory rate, respiratory effort and general vitality are the most important parameters to monitor when treating a dog with acute heart failure.
Heart failure (HF) is one of the most common causes of morbidity and mortality in dogs and, as an acute presentation (acute heart failure; AHF), it represents a serious risk of death, requiring intensive care hospitalization. There are several definitions for HF, but ultimately it should be considered as a complex clinical syndrome that can result from any structural or functional cardiac disorder that impairs the heart’s capacity to fill with or eject blood, with a consequent inability to fulfil the metabolic requirement of the body tissues 1. Clinical signs associated with AHF depend mainly on the underlying cardiac pathology, which can affect the endocardium (i.e., valvular disease), myocardium (i.e., cardiomyopathies), or great vessels (e.g., systemic or pulmonary hypertension, arterial embolisms). Dogs in AHF usually present with clinical signs relating to a left-sided cardiac impairment, namely left ventricle and atrium, but right-sided or biventricular failure is also relatively common. In most dogs with heart disease, abnormalities of systolic and diastolic dysfunction coexist, so the traditional differentiation between systolic and diastolic failure does not have any real clinical importance.
The cardinal manifestations of AHF are tachypnea and/or dyspnea, although dogs with right-sided HF may present primarily with abdominal enlargement secondary to ascites. Less specific signs can include lethargy, exercise impairment, inappetence, anxiety, and hypothermia, although exercise intolerance is frequently unnoticed in small/toy breeds due to their sedentary lifestyle.
Tachypnea/dyspnea is mainly the consequence of fluid retention (congestive heart failure or CHF) characterized by pulmonary edema and/or pleural effusion and/or ascites, although ascites needs to be particularly severe to cause respiratory abnormalities. Therefore, the terms heart failure (HF) and congestive heart failure (CHF) are often used interchangeably in canine cardiology.
Approximately 50% of dogs with mitral valve disease present with coughing, often referred to as “cardiac cough” and its mechanism is frequently explained in different ways. The most common erroneous explanation is the presence of pulmonary edema, as often reported in textbooks and historically inappropriately reinforced in veterinary education. However, a cough reflex cannot be evoked in the deeper respiratory tract (respiratory bronchioles or alveolar space) where pulmonary edema occurs, because there are no cough receptors at this level. Instead, the presence of fluid in these areas will almost inevitably cause tachypnea and/or dyspnea. Another explanation is the mechanical stimulus of bronchial cough receptors caused by the enlarged heart (and in particular the enlarged left atrium, which lies just beneath the main stem bronchi). However, puppies with significant congenital cardiac defects and severe cardiomegaly rarely present with coughing. Since airway disease is more commonly observed in older dogs, cardiomegaly may be a more likely cause of cough in patients with pre-existing airway disease, due to a summative stimulation of coughing receptors caused by the two co-morbidities, compared with young individuals that have a healthy respiratory system. Indeed, left atrial enlargement is associated with an increased risk of cough in dogs with chronic degenerative mitral valve disease, and there is a ten-fold increased risk of coughing if left atrial enlargement and airway disease coexist, even when pulmonary edema is absent. Therefore, the term “cardiac cough” should be considered a misnomer, since coughing originates from the airways, even in cardiac patients. The only exception might be a cough secondary to fulminant pulmonary edema in peracute CHF, mainly observed in larger breeds, due to flooding of airways and subsequent stimulation of cough receptors. However, even here the main clinical signs will be tachypnea/dyspnea, so a cough without tachypnea/dyspnea cannot be indicative of CHF. For this reason, in cardiac patients presenting with a cough, differential diagnosis should always include other comorbidities, such as infectious tracheobronchitis, tracheal collapse, bronchomalacia, gastro-esophageal reflux disease (GORD), post-nasal drip syndrome (PNDS), neoplasia, or bronchopneumonia 2,3.
Physical findings that can support the presence of HF in a dog presenting with acute onset tachypnea/dyspnea include the detection of a heart murmur on auscultation (usually loud, although it can be softer in large breeds), pale mucous membranes, cyanosis, weak femoral pulses, tachycardia, arrhythmias, and hypothermia. Heart murmurs are certainly an important finding to identify an underlying cardiac abnormality that may have led to the onset of AHF. Loud murmurs in small-breed dogs with myxomatous mitral valve disease (MMVD) are not necessarily proof of a disease sufficiently severe to cause AHF, but the probability of advanced cardiac remodeling associated with HF increases with increasing murmur intensity. Conversely, soft murmurs are strongly suggestive of mild disease in these dogs 4. However, in large and giant breed dogs, a loud heart murmur is not necessarily heard in the presence of a severe cardiac disease associated with valvular regurgitation, valvular stenosis and even shunts. The reason for these “silent” murmurs in large breeds is not fully understood, but it could be associated with the chest size and conformation, causing attenuation of sounds travelling from the origin of the blood flow turbulence to the surface of the chest. Furthermore, these dogs often pant incessantly during physical examination, making cardiac auscultation challenging.
Although crackles have historically been reported as indicative of alveolar pulmonary edema, this is not entirely correct. They are mostly generated by sudden airway closing during expiration and sudden airway reopening during inspiration, which is more common in pneumonia, interstitial lung disease and other parenchymal disorders. Furthermore, rhonchi associated with bronchial disease are often mistakenly described as crackles, potentially leading to a wrong diagnosis 5.
Chest percussion is seldom performed nowadays in small animal practice due to the widespread availability of point-of-care ultrasound (POCUS) examination, but this simple assessment is always recommended to identify the horizontal line of dullness associated with pleural effusion.
Another important physical finding to consider before making a diagnosis of AHF is the presence or absence of sinus arrhythmia. This is a physiological variation in heart rate (HR), usually associated with the respiratory cycle (i.e., increased HR during inspiration and reduced HR during expiration), which tends to disappear in HF due to the increased sympathetic tone and concomitant withdrawal of the parasympathetic (vagal) tone 6. Therefore, the presence of sinus arrhythmia in a dog presenting with acute onset tachypnea/dyspnea should make a diagnosis of AHF very unlikely.
Based on the above considerations, AHF diagnosis is not always straightforward, and it should be based on various factors, including an acute onset of tachypnea/dyspnea, specific findings on physical examination (e.g., a heart murmur, arrhythmia or gallop sounds), and recognition of pulmonary edema, pleural effusion and ascites on radiography and/or ultrasonography. Cardiac biomarkers can be useful to detect myocardial damage (i.e., cardiac troponin-I) or myocardial stretch (NT-proBNP), although they cannot provide definitive confirmation of CHF if not evaluated alongside the entire clinical picture. Therefore, a single diagnostic test for AHF does not exist, and its diagnosis remains largely a clinical judgment based on a careful history collection and thorough clinical examination 1.
Dogs presenting with acute signs of CHF are vulnerable and any additional stress or anxiety can potentially exacerbate the respiratory signs, so it is advisable to sedate these patients before attempting any diagnostic test or therapeutic procedure. This makes the dog more comfortable and compliant, and can reduce its peripheral metabolic requirements which cannot otherwise be satisfied by the reduced cardiac output that accompanies all cases of AHF. The dog should be maintained in a comfortable sternal recumbency in order to facilitate its natural respiratory movements. If dullness is detected on chest percussion, this should raise suspicion of pleural effusion, which can easily be confirmed if ultrasound facilities and expertise are available. A rapid point-of-care ultrasound examination (POCUS) can be performed before thoracic radiography, which allows signs of significant underlying cardiac disease, such as left atrial enlargement, systolic dysfunction, ascites, pleural and pericardial effusion, to be identified 7. Ideally, POCUS should be performed with the dog standing or in sternal recumbency, after sedation and with simultaneous oxygen supplementation. Learning how to perform thoracic ultrasound is relatively simple and intuitive, and general practitioners can achieve proficiency in identifying left atrial enlargement, pleural and pericardial effusions 8. Left atrial size (LA) measured in B-mode from the right parasternal four-chamber long axis view is readily achievable even by untrained practitioners. A LA size indexed to the aortic diameter (LA/Ao) greater than 1.6 in a right parasternal short-axis view at the level of the heart base suggests left atrial enlargement, although in dogs with AHF the LA/Ao index is usually significantly higher than this 9. Conversely, if the LA size is normal in a dog with acute onset of tachypnea/dyspnea, non-cardiac causes of dyspnea should initially be sought. The right atrial (RA) size can be subjectively assessed with thoracic POCUS, using the right parasternal long axis four-chamber view or a left parasternal apical view. Therefore, if the RA appears bigger than the LA, this would suggest RA enlargement, which could explain the presence of right-sided failure (pleural effusion and/or ascites).
POCUS can also be used to identify artefacts associated with pulmonary edema. In the case of acute pulmonary edema, a vertical, hyperechoic artefact can be noted, arising from the pleural line, extending to the bottom of the screen without fading, and moving synchronously with lung sliding. These “B lines” consist of a trail of dense echoes that resemble a distally oriented comet-tail, generated by ultrasound reverberations from fluid accumulation in the alveolar spaces. B-lines can be detected using either a phased array or a curvilinear probe, and a diagnosis of pulmonary edema generally requires identification of 3 or more B lines. The number of B lines should correspond to the severity of the underlying pulmonary edema, and may also correlate with the degree of improvement after therapy 10,11,12. However, B-line artefacts are not pathognomonic of pulmonary edema, and can be observed in a number of other conditions, including dyspneic patients after lung contusions, with pneumonia, acute respiratory distress syndrome (ARDS) and pulmonary fibrosis and, occasionally, even in healthy individuals 12 (Figure 1).
 |
 |
| a | b |
 |
 |
| c | d |
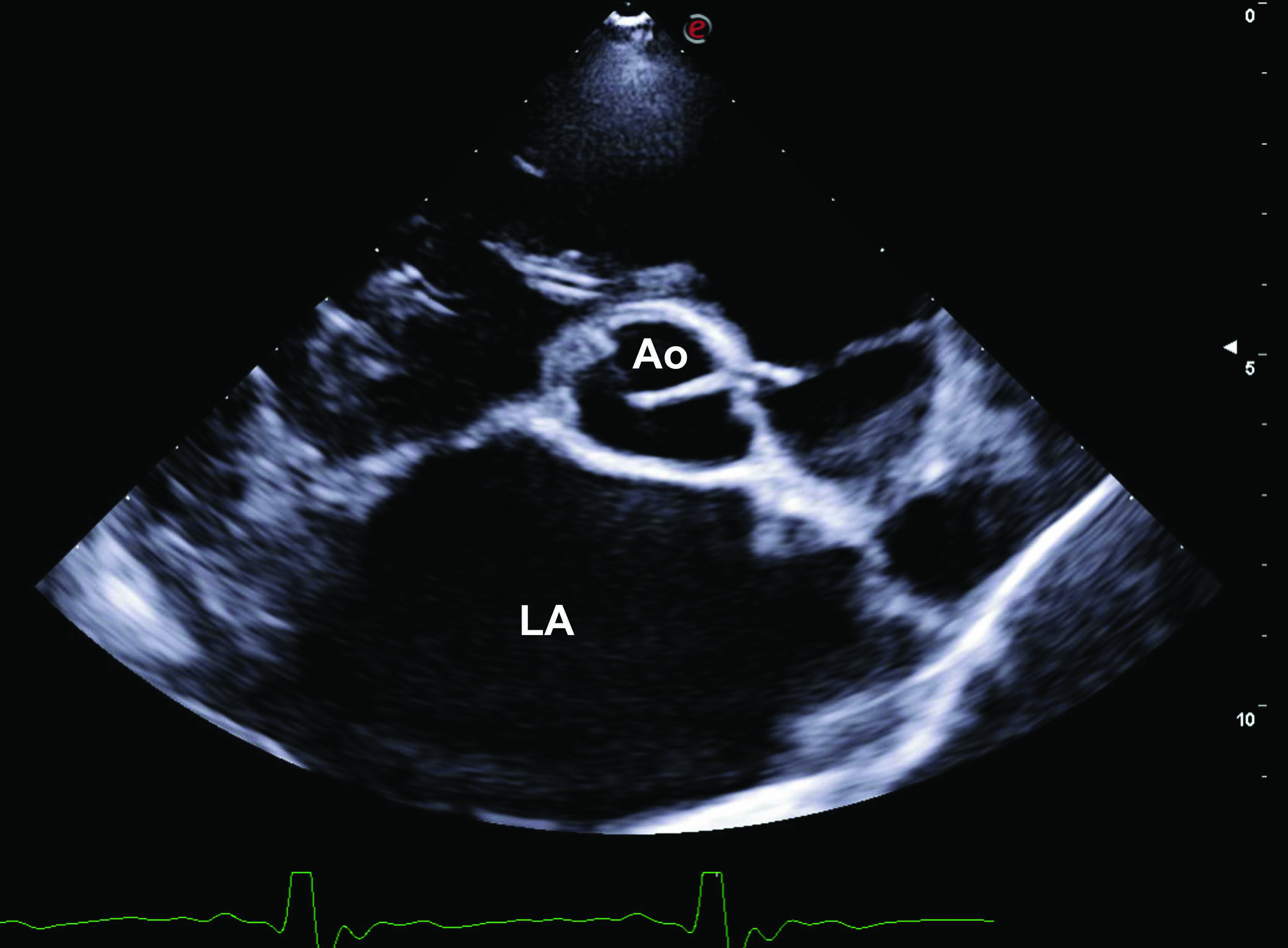
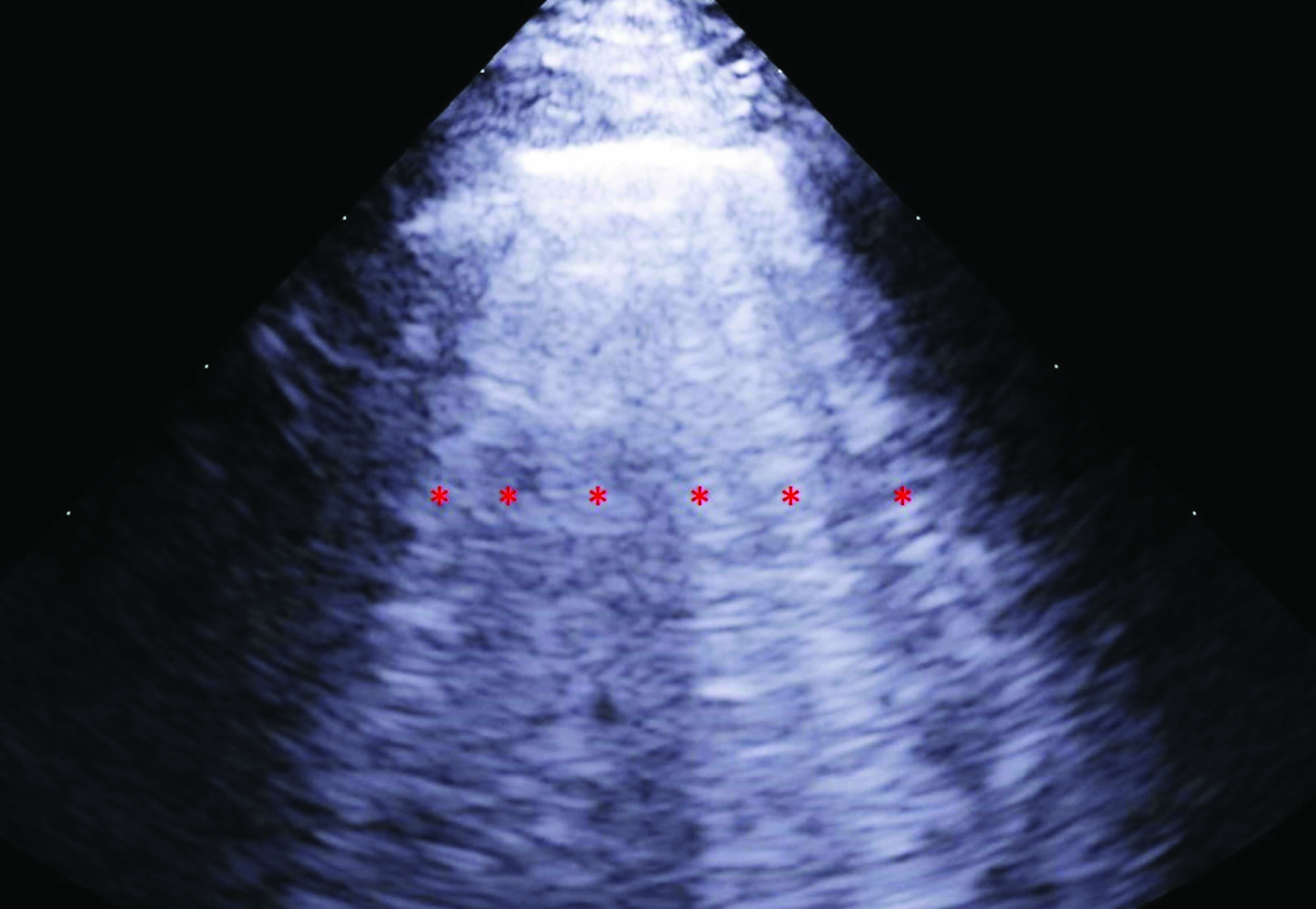
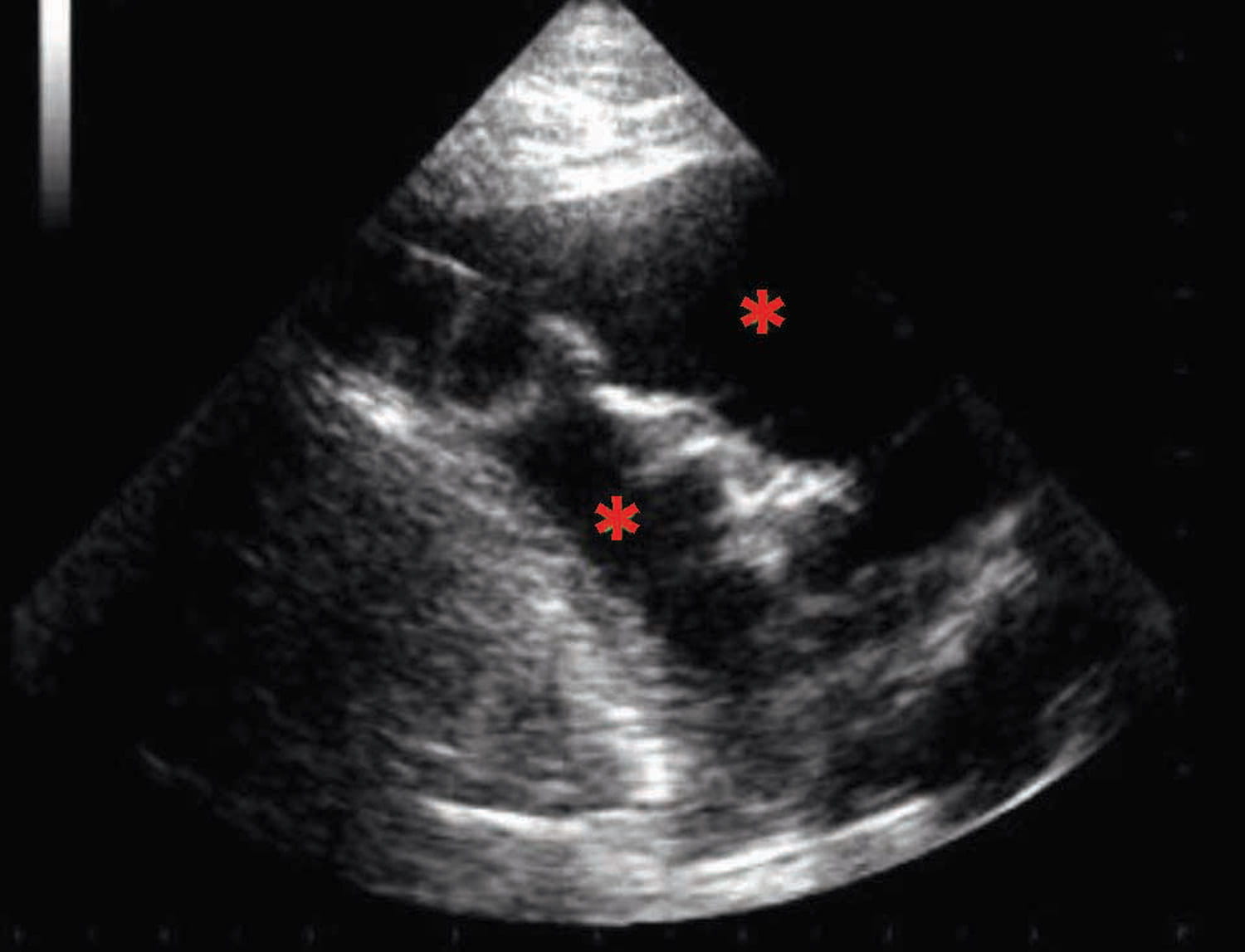
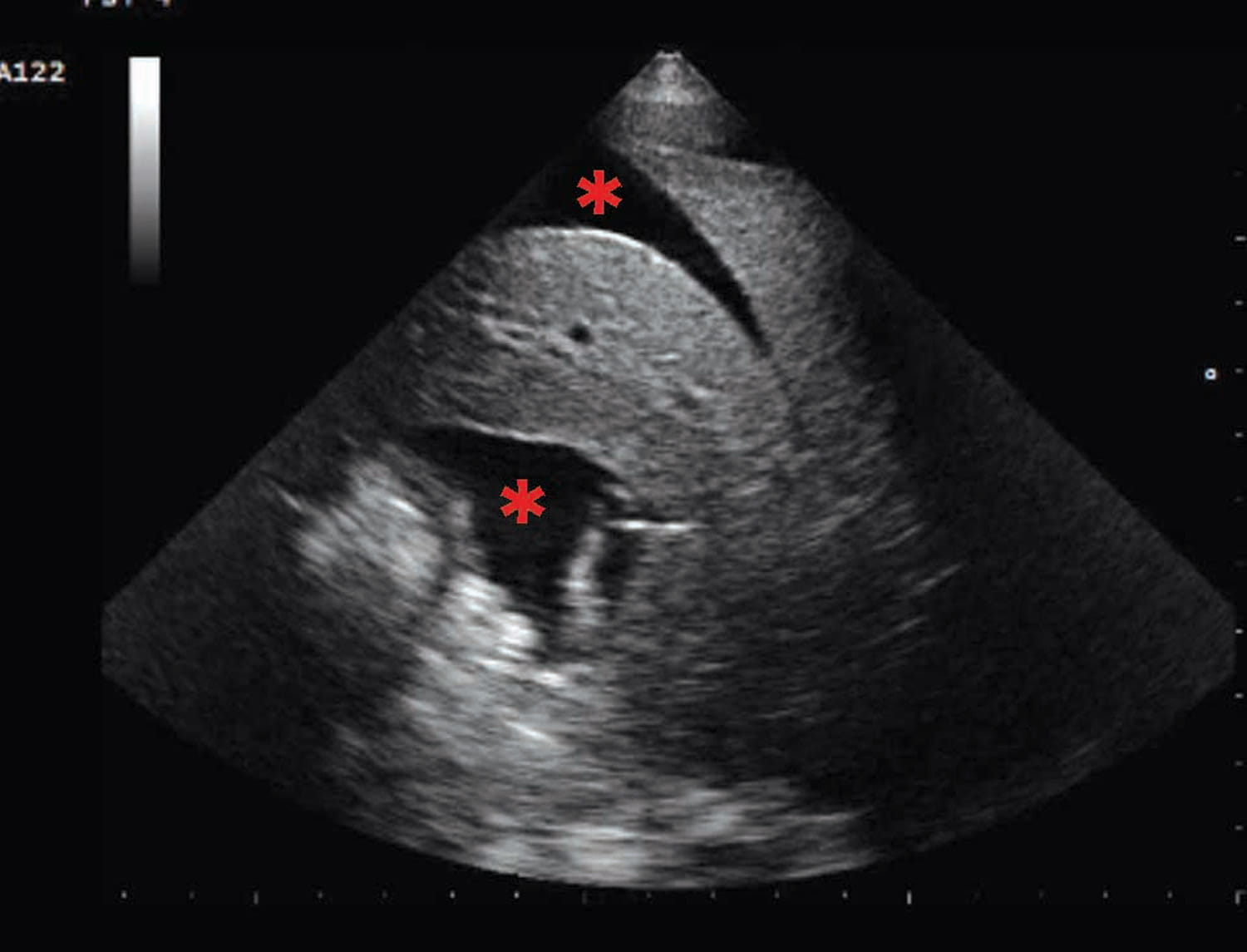
Figure 1. Point-of-care ultrasound (POCUS) images of dogs in acute heart failure. (a) right parasternal short axis view at the level of the heart base showing a significantly increased left atrium (LA) when compared to the aortic section (Ao); (b) comet tails or B-lines artefact (marked as red *) compatible with the presence of pulmonary edema; (c) pleural effusion characterized by hypoechoeic areas in the chest cavity (marked as red *); (d) ascites revealed by the presence of a hypoechoeic area in the abdominal cavity (marked as red *).
© Luca Ferasin
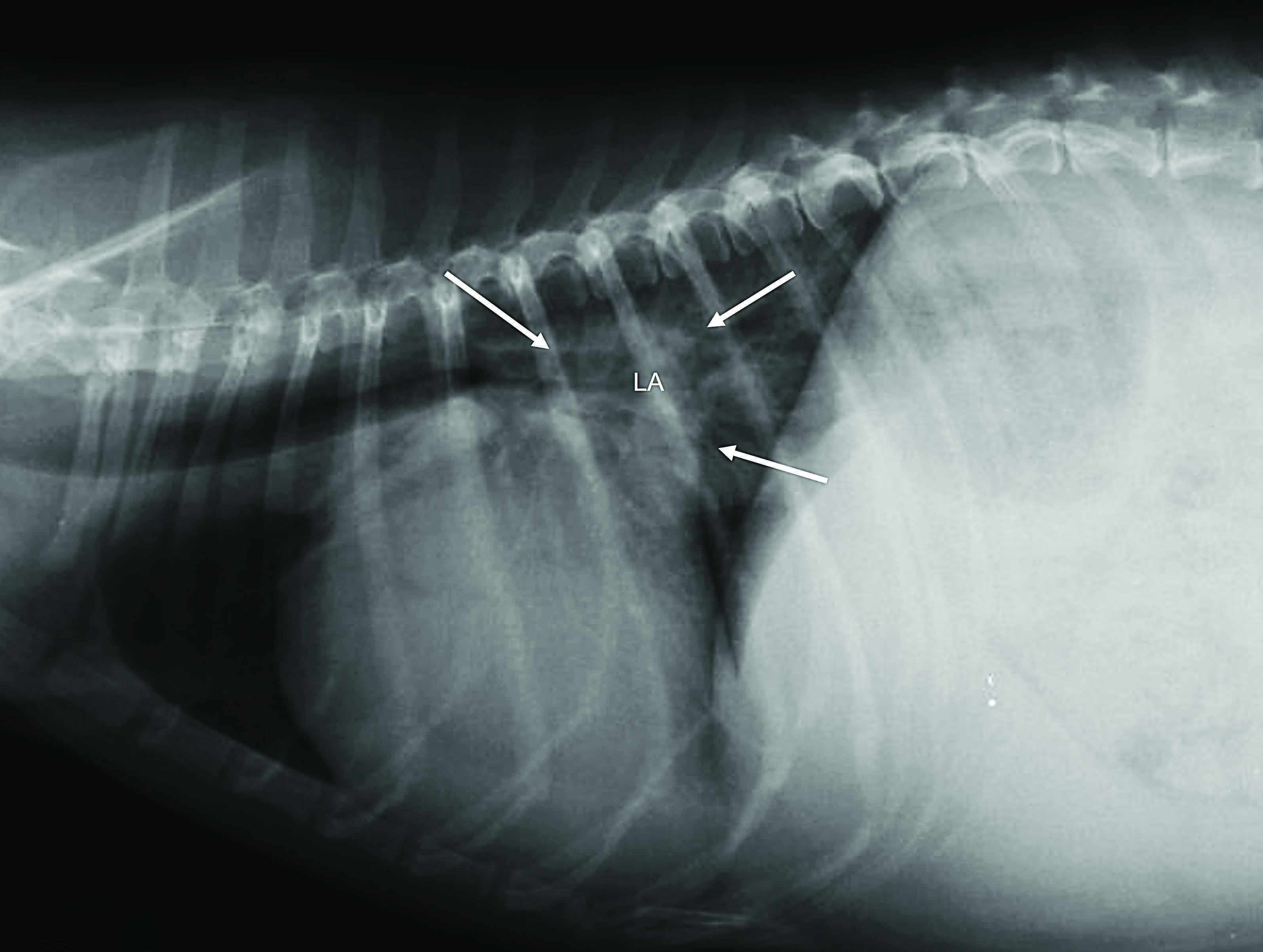
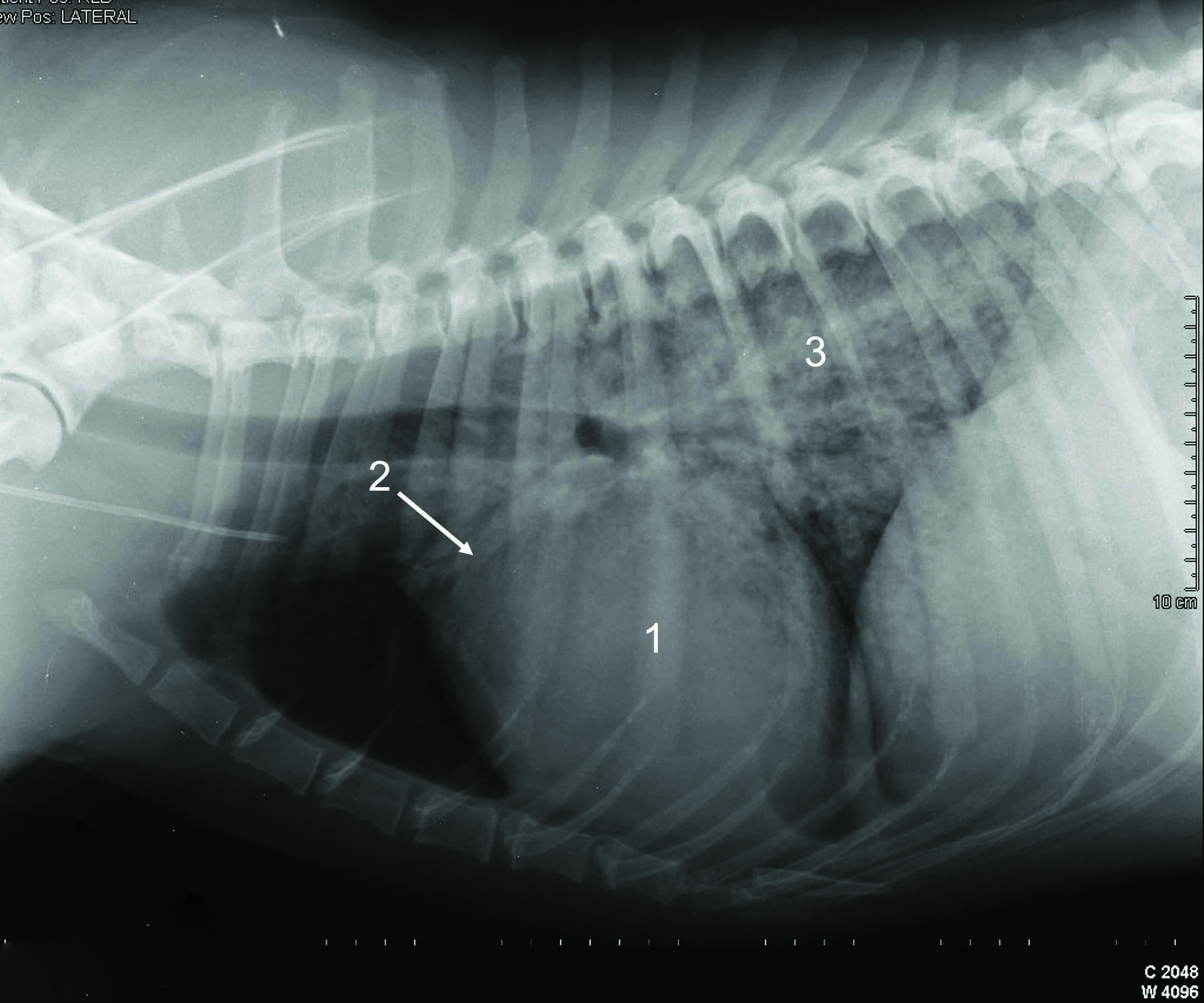
In life-threatening cases of dyspnea and where ultrasound is not readily available, parenteral furosemide administration may be justified before radiographic confirmation of pulmonary edema, should the full clinical presentation suggest acute onset of CHF 13. Thoracic radiography is still considered by many cardiologists as the “gold standard” for confirming cardiogenic pulmonary edema and signs compatible with right-sided HF. Most dogs do not require any physical restraint for radiography, which minimizes the risk of additional stress, and in fact (as noted above) AHF dogs should be sedated before any diagnostic test, which tends to significantly increase their compliance. Correct positioning can be facilitated with the use of sandbags and foam positioners. If pleural effusion has been detected on chest percussion and POCUS, complete thoracocentesis should be performed prior to radiography; this minimizes additional stress, improves respiratory function, and aids visualization of the lung fields otherwise obscured by the fluid within the chest cavity. Radiographic diagnosis of cardiogenic pulmonary edema requires identification of a triad of changes, namely cardiomegaly, the presence of pulmonary venous congestion, and a diffuse alveolar/interstitial pattern, which is particularly pronounced in the caudal lung fields. Cardiomegaly can be confirmed by subjective assessment, or by measuring the vertebral heart score (VHS), which should be higher than 10.5 14. A method to assess the left atrial size on thoracic radiographs has also been recently described 15. Particular attention should be paid when interpreting a pronounced left atrial enlargement, which is sometimes misinterpreted as “peri-hilar edema” by less experienced clinicians (Figure 2).
 |
| a |
 |
| b |
Figure 2. Thoracic radiographs of dogs with advanced cardiac disease. (a) Severe cardiomegaly particularly pronounced at the level of the left atrium (LA) as indicated by arrows. This sign is often misinterpreted by inexperienced clinicians as “peri-hilar edema”. Although this dog has clear signs of cardiomegaly, the pulmonary veins do not appear particularly congested and there are no signs referable to an interstitial-alveolar pattern that could suggest the presence of pulmonary edema; (b). A radiograph from a dog presenting with acute onset dyspnea showing the typical triad of radiographic changes, namely 1 cardiomegaly, 2 presence of pulmonary venous congestion, and 3 a diffuse alveolar/interstitial lung pattern, which is particularly pronounced in the caudal lobes.
© Luca Ferasin
Although general therapeutic guidelines have been proposed by cardiology experts, it should be highlighted that no prospective clinical trials have been published to date in dogs with AHF 7,10. The following recommendations therefore reflect current state-of-the-art clinical medicine, rather than conclusive evidence-based medicine, and come from the author’s anecdotal experiences and evidence extrapolated from human clinical trials, guidelines and consensus statements 1.
Before discussing drug therapy, three major actions should be immediately considered whenever a dog presents with a history and clinical signs compatible with AHF, namely minimizing stress, sedation, and oxygen supplementation, and all three are often necessary before making a definitive diagnosis of heart failure.
 |
 |
| a | b |
 |
 |
| c | d |
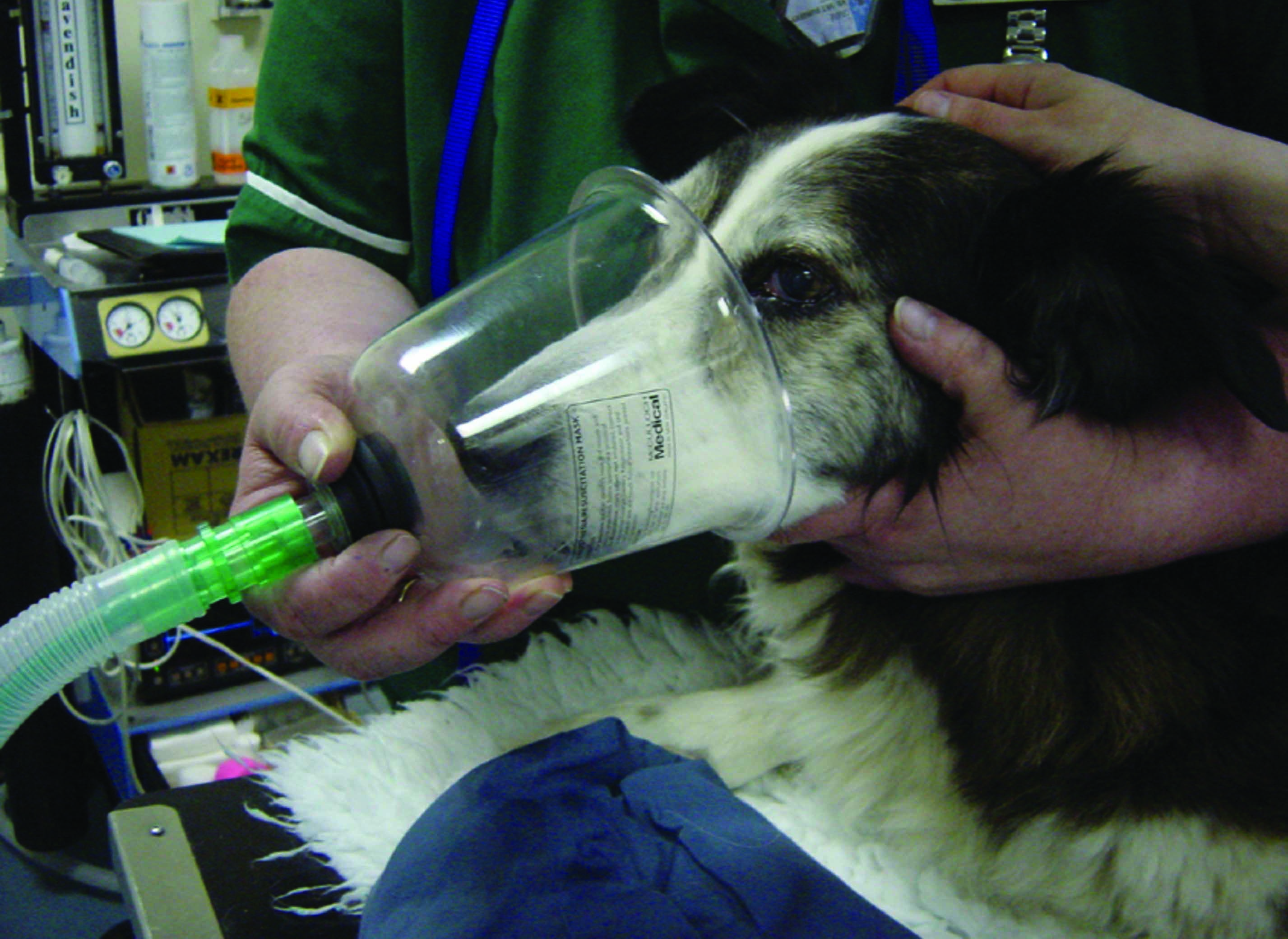
Figure 3. Examples of different techniques for oxygen supplementation in dogs with acute onset heart failure. (a) Oxygen mask; although this is commonly used in small animal emergencies, some dogs do not tolerate the mask, and a flow-by administration may be preferred instead. (b) Oxygen prongs; these can provide a better oxygen delivery compared to a flow-by method, and are generally well tolerated, especially after sedation. (c) Oxygen cages and tents; these are one of the methods most accepted by dyspneic dogs. (d) The “baggie” method; this should be avoided because of the unpredictable oxygen enrichment, potential risk of overheating and inadequate CO2 removal, with subsequent accumulation which could compromise the clinical stability of a dyspneic dog.
© Luca Ferasin
Diuretics promote increased urine production and reduce signs of congestion, particularly pulmonary edema, although they do not directly improve cardiac output and can potentially lead to diminished renal perfusion, electrolyte abnormalities and further activation of the renin-angiotensin-aldosterone system (RAAS) 20. Successful diuretic therapy should provide a rapid and complete decongestion in AHF and, once resolved, medication should be continued to prevent it from recurring 21.
In general, diuretics lead to a decreased circulating plasma volume that causes movement of water from the interstitial and alveolar spaces in the lungs into the intravascular space, resulting in improved breathing effort and a more comfortable patient. Loop diuretics (e.g., furosemide and torasemide) are most commonly used, but by themselves they are rather ineffective in controlling large volumes of pleural effusion and/or ascites (which should be alleviated by mechanical removal) and they are contraindicated in most cases of pericardial effusion. Injectable furosemide is always used for the acute onset of CHF, with the dose tailored to the individual patient, since an excess can lead to deleterious effects on renal perfusion (azotemia) and electrolytes depletion (hypokalemia, hyponatremia, hypochloremia, hypomagnesemia, and hypocalcemia), especially in older dogs 22. On the other hand, underdosing furosemide can lead to therapeutic failure, prolonged hospitalization, and potential euthanasia because of refractory heart failure. If possible, furosemide should be administered intravenously for rapid onset of action and more predictable bioavailability; however intramuscular injections may represent a reasonable alternative in some uncooperative dogs, although the therapeutic effect may be delayed. The author prefers furosemide 4 mg/kg IV for severely decompensated patients and 2 mg/kg for those with mild-moderate congestion, with an initial clinical improvement expected approximately 30 minutes after administration. Depending on the severity of the underlying pulmonary edema, repeated intravenous boluses (IVB) or initiation of continuous rate infusion (CRI, 0.66 mg/kg/h over 8 hours) of furosemide may be indicated 23. CRI have previously been recommended to increase the diuretic effect while reducing the risk of renal ischemia caused by sudden changes in vascular volume. However, a recent study did not seem to support the suggestion that adding CRI to an IVB loading dose of furosemide might improve short term outcomes in dogs in AHF and, although either IVB or CRI are efficient in relieving respiratory distress to a similar extent, CRI might carry a higher risk of dehydration and azotemia, while IVB administration may be associated with longer hospitalization and might require higher doses. Therefore, furosemide therapy for canine AHF should take into consideration the presence of co-morbidities, such as concomitant renal disease, available resources, total dose of furosemide, etc. 24.
Monitoring the respiratory rate and effort every 30-60 minutes will provide guidance on further furosemide administration. If the dose and frequency is effective, a gradual decrease in respiratory rate and effort should be apparent within a few hours and, based on the response to the first dose, subsequent furosemide doses and frequency of injections can be estimated. An empirical approach is to consider a cumulative maximal dose of 12 mg/kg/day 7.
Luca Ferasin
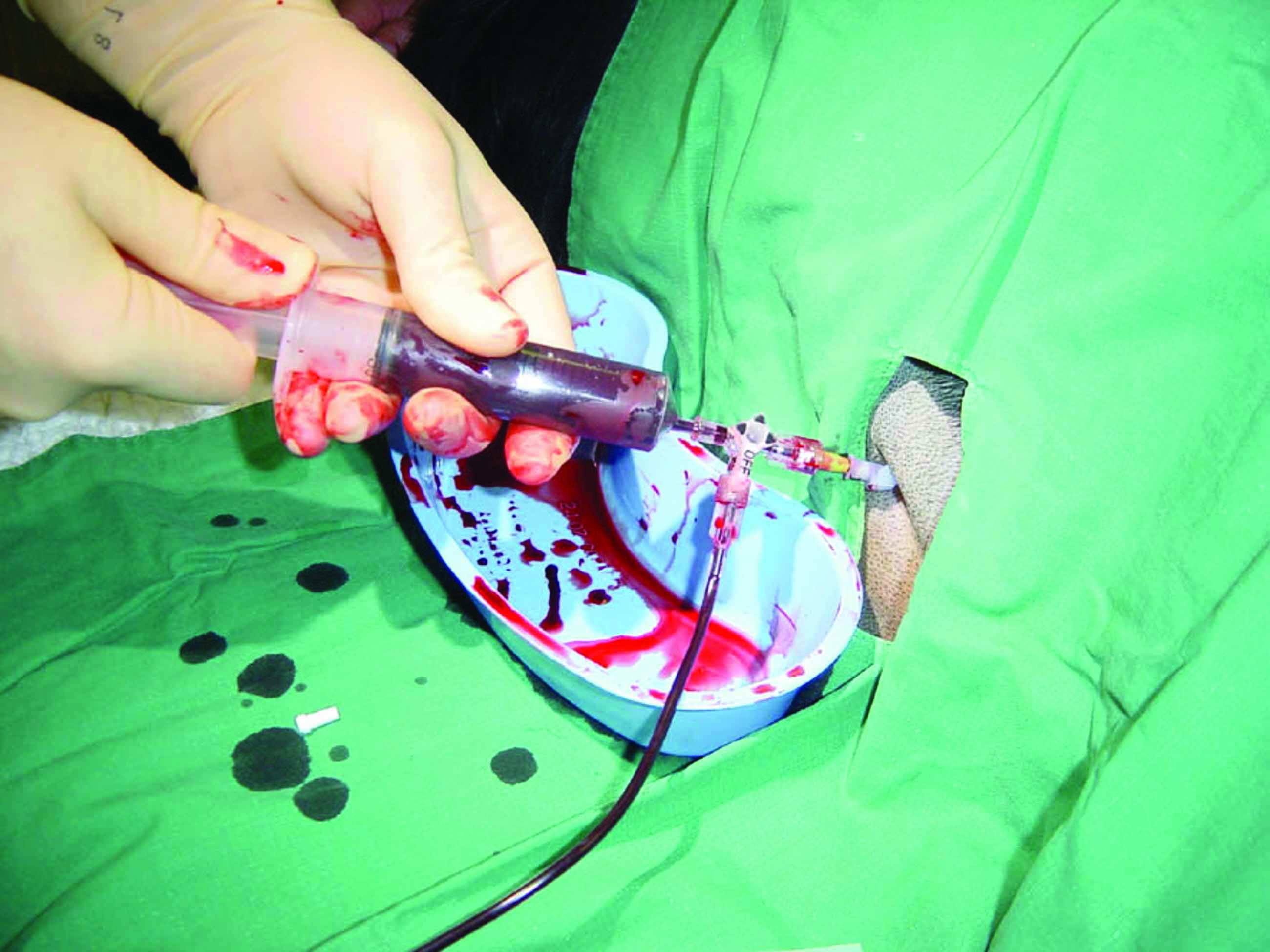
The decision to perform mechanical drainage in a dog with pleural effusion, pericardial effusion or ascites should consider the risk/benefit ratio of the procedure. For example, mild ascites or a small amount of pleural or pericardial effusion, often associated with concurrent pulmonary edema, may respond to diuresis alone. However, when the volume of fluid accumulation is severe enough to contribute to the presenting clinical signs, centesis is always recommended (Figure 4), and the most common techniques (thoracocentesis, abdominocentesis, pericardiocentesis) are relatively safe and simple; iatrogenic complications are rarely observed. Needle insertion sites for the first and third methods can be aided by physical exam or thoracic radiographs, although ultrasound guidance is currently the preferred option for most clinicians.
 |
 |
| a | b |
 |
 |
| c | d |
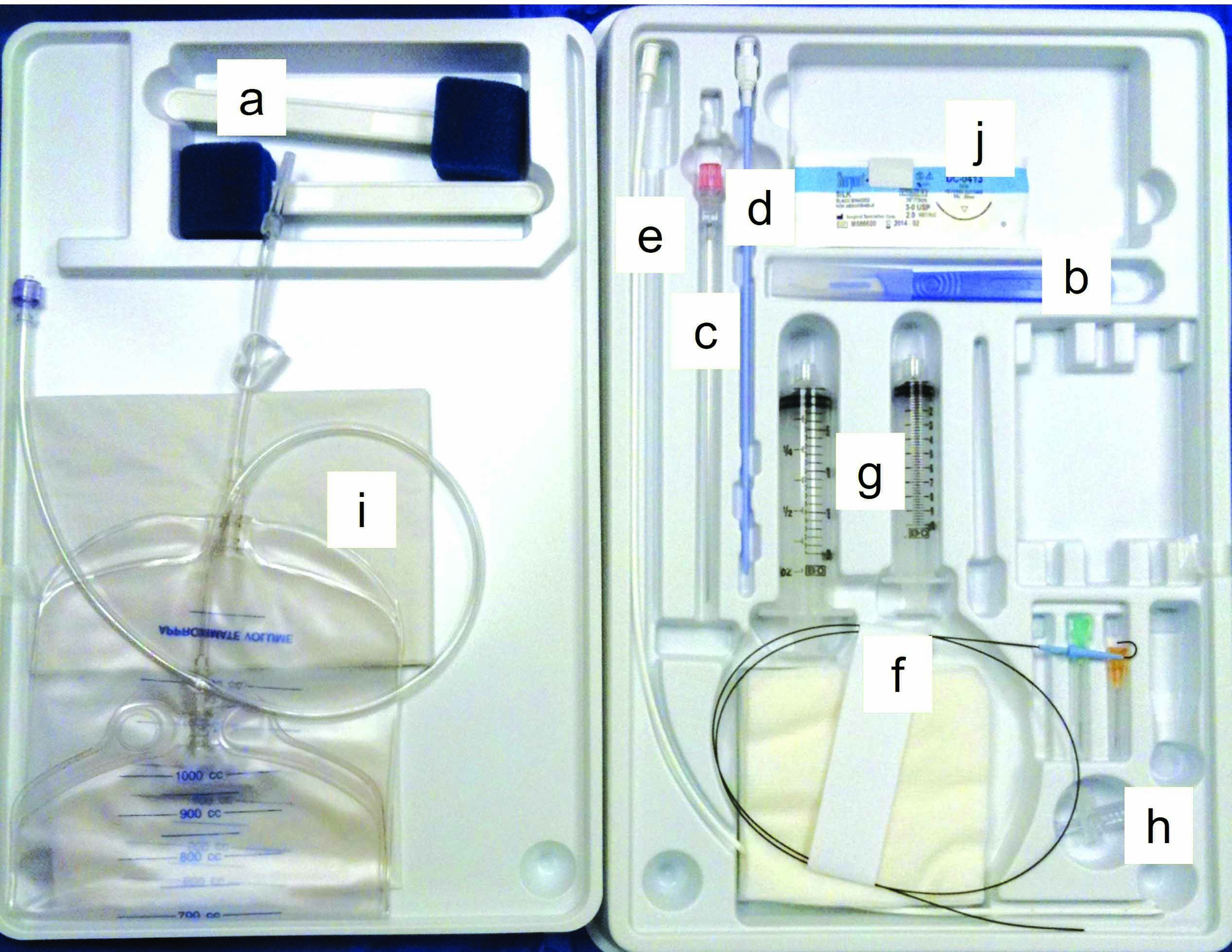
Figure 4. Centesis equipment and techniques. (a) centesis set based on Seldinger wire technique; a: skin scrubbing pads; b: scalpel blade; c: needle; d: dilator; e: catheter; f: guidewire; g: Luer lock syringes; h: three-way stopcock; i: collection bag; j: suture material. (b) thoracocentesis performed in a dog with pleural effusion using a butterfly catheter; (c) insertion of a pre-fenestrated catheter in the abdomen to alleviate severe ascites (abdominocentesis); (d) pericardiocentesis performed using a pre-fenestrated soft catheter; most pericardial effusions are characterized by serosanguineous fluid accumulation.
© Luca Ferasin
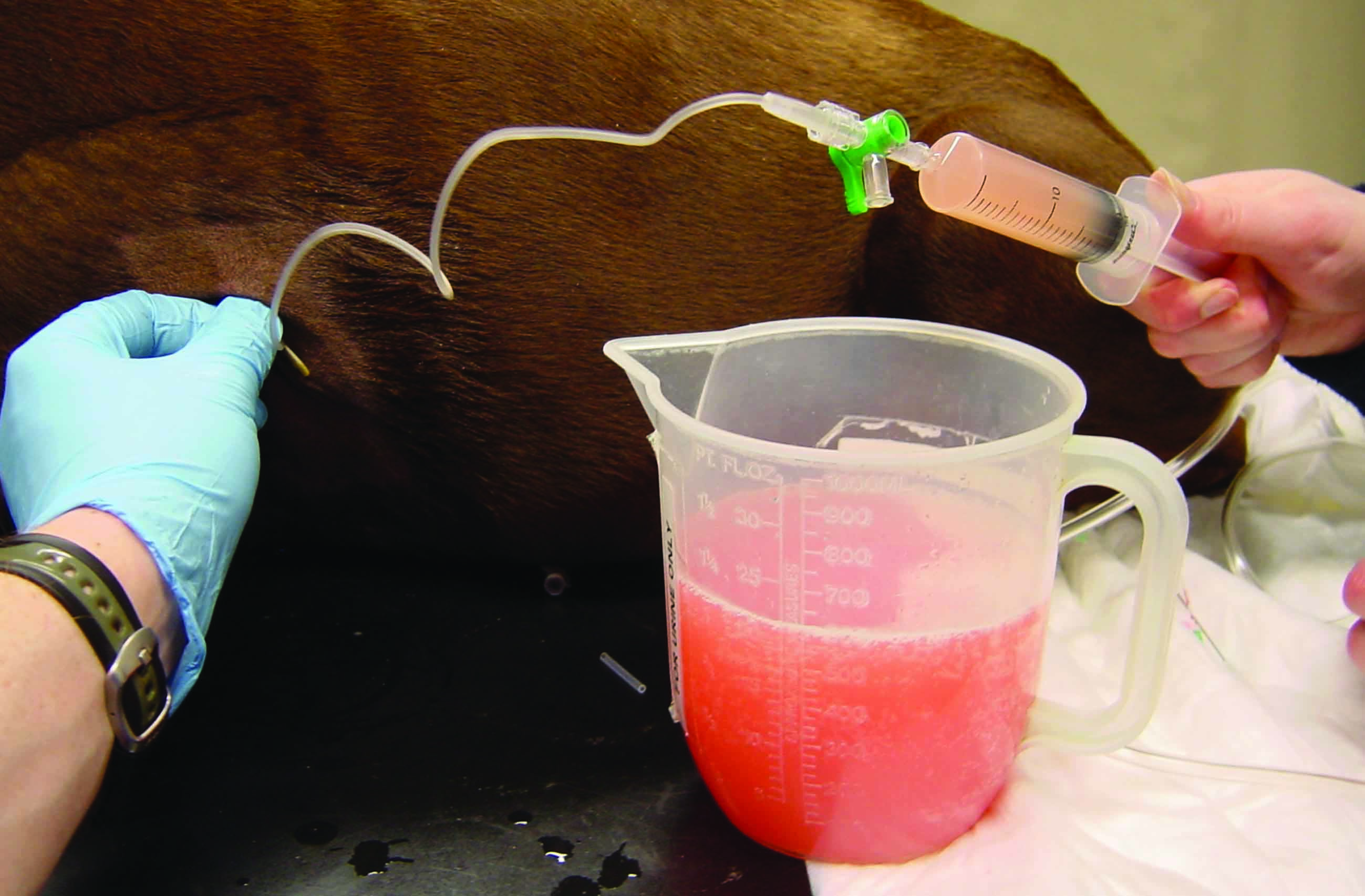
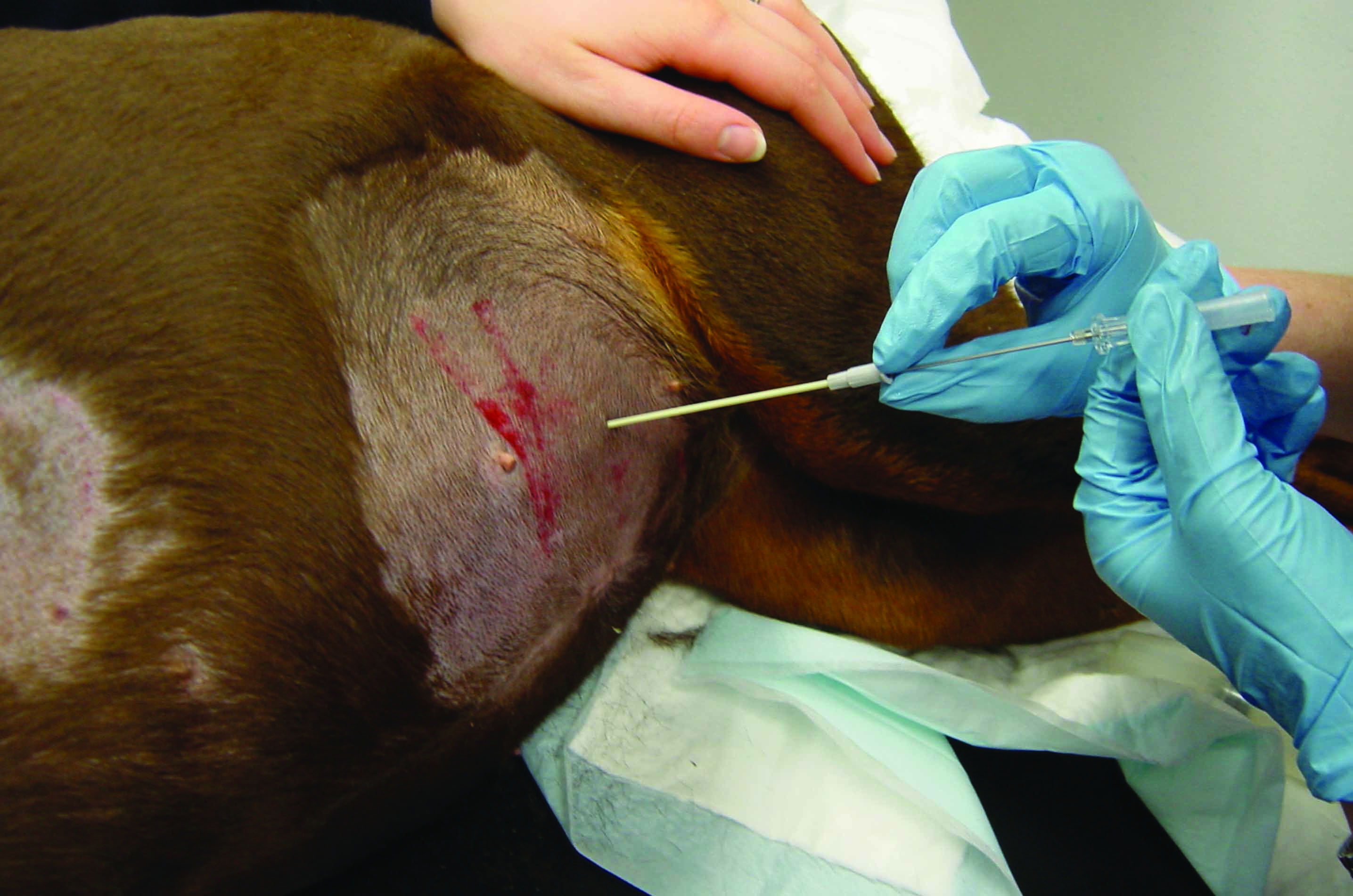
Thoracocentesis is performed by inserting a needle into the chest cavity at the level of the 7-8th intercostal space, lower and middle third, entering the pleural space in front of the rib to avoid vessels and nerves running along the caudal aspect of the ribs. Most effusions can be drained just on one side due to the presence of a large mediastinal fenestration, and bilateral centesis is usually unnecessary. The procedure should be performed with the dog in sternal recumbency with adequate manual restraint, while providing supplemental oxygen if necessary. Either a dedicated centesis set, a butterfly needle, or an over-the-needle IV catheter attached to extension tubing can be used, each with its advantages and disadvantages. A 3-way tap or one-way centesis valve will expedite the procedure and minimize movement. The butterfly needle, for example, works well with small dogs, but the needle is not long enough to be safely introduced and positioned in bigger dogs, where over-the-needle catheters (16G or 18G) potentially allow more complete and safer removal of pleural fluid once in the chest cavity. For this technique, the author prefers to use a small dose of lidocaine to minimize patient discomfort associated with the needle insertion. The disadvantages of the over-the-needle catheter system relate to the longer preparation time, especially if fenestrations are made, the need for a releasing incision and local block in the skin, and difficulties in threading the catheter into the chest. There is also a risk of catheter fragmentation, resulting in a small piece of the catheter remaining in the chest cavity. The clinician should pay particular attention while pulling the needle stylet back as the catheter is advanced in the chest, since this can result in kinking or loss of positioning in the chest cavity 13. Dedicated centesis sets provide a soft guidewire that can be inserted through a sharp hollow needle and, when correctly positioned, the guidewire is left in place and exchanged with a pre-fenestrated soft catheter which is then advanced within the chest cavity (Seldinger technique). All these techniques work well in experienced hands, and the decision is ultimately personal preference. After thoracocentesis, respiratory rate and effort should be monitored carefully, and a significant improvement should be expected within a few minutes, but continuous monitoring in the following few hours is advisable to ensure that an iatrogenic pneumothorax has not developed.
Abdominocentesis should be considered when excessive ascites interferes with respiration and causes excessive patient discomfort. Again, the process can be performed successfully using a butterfly needle (in small dogs) or an over-the-needle fenestrated catheter. The author favors a hybrid technique using fistula cannulas (also called fistula needles), which are specifically designed for hemodialysis cannulation. These are similar to butterfly cannulas but tend to be of larger gauge and the needle features a side hole (“back eye”) to maximize flow from the access point, which again depends on clinician’s preference, although ultrasound-guidance will allow identification of the largest pocket of fluid in order to maximize drainage and avoid puncture of abdominal organs and major vessels.
Pericardial effusion in dogs is commonly caused by cardiac tumors, although many cases are of unknown etiology (idiopathic effusion), and pericardiocentesis is needed when the amount of fluid within the pericardial sac is severe enough to cause signs of cardiac tamponade, usually accompanied by right atrial wall collapse. For this procedure the dog should be placed in sternal or left lateral recumbency depending on its demeanor, degree of sedation, and the clinician’s preference. The optimal site for pericardiocentesis is typically on the right side at the 4th or 5th intercostal space, just at or above the costochondral junction, with ultrasound commonly used to identify the ideal point of needle insertion. Again, various methods are available, although the author favors the use of dedicated pericardiocentesis sets with pre-fenestrated soft catheters inserted via Seldinger technique following local anesthesia. A continuous ECG is used to detect arrhythmias induced by the needle or catheter. When performed properly, the complication rate of pericardiocentesis is low, but the risks should be discussed with the owner before attempting the procedure. The use of diuretics in pericardial effusion is often discouraged because they may reduce an already compromised cardiac preload. However, pericardial effusion can also be secondary to right-sided heart failure, but here it is usually low volume, and will often resolve with diuretics alone.
Luca Ferasin
In humans with acute pulmonary edema, positive inotropes may aid resolution of AHF regardless of the etiology because of the ability of these agents to improve myocardial function. Pimobendan has been used with increased frequency in the management of canine AHF due to its dual mechanism of action, namely calcium sensitization (which improves contractility with minimal effects on myocardial oxygen consumption) and phosphodiesterase inhibition (which promotes peripheral vasodilation). An injectable form is available in many countries which can be administered to dogs with AHF, especially in the presence of left ventricular systolic dysfunction, significant pleural effusion, renal insufficiency, or severe refractory pulmonary edema. Dobutamine (an adrenergic positive inotrope with primarily beta-1 receptor effects) can also be considered in dogs with acutely decompensated heart failure characterized by low cardiac output. Intravenous administration should be considered, especially for the first-time heart failure patient with systolic dysfunction. The dose ranges from 1-10 mcg/kg/min CRI, starting with a low dose and titrating upward based on blood pressure and ECG monitoring, since hypertension and tachyarrhythmias represent the main adverse effects 7.
Drugs such as nitrates (e.g., nitroglycerin, sodium nitroprusside) that promote arterial and venous dilation can be considered to reduce afterload and improve cardiac output in failing hearts because the afterload is often elevated in heart failure due to the upregulation of the sympathetic nervous system and activation of the renin-angiotensin-aldosterone system (RAAS). Nitroglycerin potently dilates large arteries (including coronary arteries) and veins but has less effect on smaller arterioles; it is available in different formulations, but 2% ointment is most commonly used in small animals, generally applied topically in conjunction with diuretics in the acute management of severe heart failure to further reduce preload. Sodium nitroprusside is a potent venous and arterial dilator which dilates both the systemic and pulmonary vasculature, but has the potential to cause marked hypotension. An initial low dose (0.1-0.2 mcg/kg/min) is recommended, with upward titration based on blood pressure, targeting a mean blood pressure of 70 mmHg or systolic blood pressure 90-100 mmHg 8. Continuous arterial blood pressure monitoring is recommended to reduce the risk of severe hypotension. In the absence of systemic hypotension, the use of nitrates appears to be safe and effective, and seems associated with improved symptoms and reduced mortality in dogs with AHF.
These agents should be used with caution in dogs with heart failure. In most cases, efficient control of the congestion and improvement of the cardiac output will improve the arrhythmia indirectly, so low grade arrhythmias are generally not treated. Nevertheless, if hemodynamically significant supraventricular or ventricular tachyarrhythmias are present, then specific antiarrhythmic treatment is recommended. Although the rapid rate compromises cardiac output, another major concern is tachycardia-induced myocardial damage (tachycardiomyopathy) which can result from sustained rapid rates for prolonged periods of time, and is known to occur in patients with supraventricular or ventricular tachyarrhythmias.
To improve cardiac output and reduce the risk of tachycardiomyopathy in rapid atrial fibrillation (e.g., ventricular response rate higher than 180 bpm) the author recommends a rate-control therapy with combined administration of digoxin + diltiazem. Digoxin should be given at 0.003-0.005 [maximum] mg/kg PO BID), the goal being to use the lowest effective dose, not the highest without side effects (as commonly advocated in the past). It is extremely important to determine the serum digoxin level 3-5 days after initiation of treatment and always 6-8 hours after pilling (trough level). The modern target therapeutic serum level is 0.5-1.2 ng/mL (0.65-1.53 nmol/L), which is probably lower than that often reported in the literature. Diltiazem can be given at 2-5 mg/kg q12h in a sustained release form in combination with digoxin. The efficacy of treatment of atrial fibrillation should be periodically monitored, ideally after 5-7 days and by ambulatory 24h Holter recording (target average daily rate 100-120 bpm) 25. However, if unavailable, standard ECG monitoring (target rate approximately 130-150 bpm) is recommended, performed 5-7 days after starting anti-arrhythmic therapy and then every 3-6 months thereafter.
If rapid and sustained ventricular tachycardia is diagnosed, administration of lidocaine (2 mg/kg IV bolus over 1 minute) followed by CRI (0.025-0.1 mg/kg/min) should be considered, while monitoring for potential side effects (depression, seizures, muscle fasciculations, vomiting, bradycardia and hypotension). If reactions are severe, it is advisable to decrease or discontinue administration.
Other anti-arrhythmic interventions should be carefully assessed. For example, starting beta-blockade (e.g., propranolol, atenolol, sotalol) in dogs with AHF could be deleterious and should be discouraged.
Some severe cases of AHF (characterized by respiratory muscle fatigue refractory to medical therapy) may require intubation and mechanical ventilation as a lifesaving intervention. Respiratory muscle fatigue is generally diagnosed by a simultaneous presence of decreased respiratory rate, hypercapnia and decreased consciousness. Mechanical ventilation in these dogs will support the respiratory system and increase the safety of further diagnostic testing, but is expensive and requires extensive nursing care, ventilator equipment and expertise. Therefore any decision to offer mechanical ventilation needs to consider both the prognosis for a meaningful recovery of the patient, hospital equipment and expertise, as well as owner’s financial availability and expectations 13.
Respiratory rate, respiratory effort and general vitality are the most important parameters when monitoring dogs with AHF. A dog with severe cardiogenic pulmonary edema would be expected to improve over the course of a few hours to a day following diuretic therapy. If no improvement is noted in respiratory rate and effort after an initial furosemide dose within a couple of hours, then a repeated dose is recommended. Titration of the dose and frequency of furosemide administration should be instituted once the breathing rate has normalized, and oxygen supplementation has been discontinued. If the dog remains clinically stable after reducing the dose of injectable furosemide, oral furosemide (or torasemide) should be introduced before discharge. Blood pressure monitoring every 4-6 hours during hospitalization is also recommended, especially if using adjunctive vasodilator therapy, and measurement of renal and electrolyte parameters should ideally be performed before starting treatment and repeated a few days later. In general, dogs respond successfully to treatment after their first episode of acutely decompensated heart failure, with an estimated survival rate to discharge approaching 80% 26.
Finally, an important and often overlooked part of the successful emergency management of heart failure is open communication with the dog’s owner regarding the emotional, practical, and financial ability to deal with the long-term management of the animal’s condition. It is also good practice to contact the owners for follow-up a few days after discharge and arrange a clinical recheck after approximately 1-2 weeks, depending on individual circumstances.
|
Acknowledgement
The author would like to thank Dr. Heidi Ferasin for her contribution to the management of some of the reported clinical cases and for her invaluable assistance in writing this manuscript.
|
Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guidelines for the Management of Heart Failure: Executive Summary. J. Am. Coll. Cardiol. 2022;79(17):1757-1780. Epub 20220401. DOI: 10.1016/j.jacc.2021.12.011. PubMed PMID: 35379504.
Ferasin L, Crews L, Biller DS, et al. Risk factors for coughing in dogs with naturally acquired myxomatous mitral valve disease. J. Vet. Intern. Med. 2013;27(2):286-292. Epub 20130209. DOI: 10.1111/jvim.12039. PubMed PMID: 23398050.
Ferasin L, Linney C. Coughing in dogs: what is the evidence for and against a cardiac cough? J. Small Anim. Pract. 2019;60(3):139-145. Epub 20190116. DOI: 10.1111/jsap.12976. PubMed PMID: 30652329.
Ljungvall I, Rishniw M, Porciello F, et al. Murmur intensity in small-breed dogs with myxomatous mitral valve disease reflects disease severity. J. Small Anim. Pract. 2014;55(11):545-550. Epub 20140912. DOI: 10.1111/jsap.12265. PubMed PMID: 25213440.
Ferasin L. Coughing. In; Textbook of Veterinary Internal Medicine. Ettinger SJ, Feldman EC, (eds.) 8th ed. St. Louis, MI: Saunders; 2016;107-110.
Chan SHH. Reinstatement of respiratory sinus arrhythmia as a therapeutic target of cardiac pacing for the management of heart failure. J. Physiol. 2020;598(3):433-434. Epub 20200115. DOI: 10.1113/JP279261. PubMed PMID: 31805609.
DeFrancesco TC. Management of cardiac emergencies in small animals. Vet. Clin. North Am. Small Anim. Pract. 2013;43(4):817-842. DOI: 10.1016/j.cvsm.2013.03.012. PubMed PMID: 23747262.
Tse YC, Rush JE, Cunningham SM, et al. Evaluation of a training course in focused echocardiography for noncardiology house officers. J. Vet. Emerg. Crit Care (San Antonio). 2013;23(3):268-273. Epub 20130506. DOI: 10.1111/vec.12056. PubMed PMID: 23647602.
Borgarelli M, Ferasin L, Lamb K, et al. DELay of Appearance of sYmptoms of Canine Degenerative Mitral Valve Disease Treated with Spironolactone and Benazepril: the DELAY Study. J. Vet. Cardiol. 2020;27:34-53. Epub 20200110. DOI: 10.1016/j.jvc.2019.12.002. PubMed PMID: 32032923.
DeFrancesco TC, Ward JL. Focused canine cardiac ultrasound. Vet. Clin. North Am. Small Anim. Pract. 2021;51(6):1203-1216. Epub 20210909. DOI: 10.1016/j.cvsm.2021.07.005. PubMed PMID: 34511294.
Taylor A, Anjum F, O’Rourke MC. Thoracic and lung ultrasound. StatPearls. https://www.ncbi.nlm.nih.gov/books/NBK500013/ accessed 15/09/22
Dietrich CF, Mathis G, Blaivas M, et al. Lung B-line artefacts and their use. J. Thorac. Dis. 2016;8(6):1356-1365. DOI: 10.21037/jtd.2016.04.55. PubMed PMID: 27293860; PubMed Central PMCID: PMC4885976.
Ferasin L, DeFrancesco T. Management of acute heart failure in cats. J. Vet. Cardiol. 2015;17 Suppl 1:S173-189. DOI: 10.1016/j.jvc.2015.09.007. PubMed PMID: 26776576.
Boswood A, Haggstrom J, Gordon SG, et al. Effect of pimobendan in dogs with preclinical myxomatous mitral valve disease and cardiomegaly: the EPIC Study – a randomized clinical trial. J. Vet. Intern. Med. 2016;30(6):1765-1779. Epub 20160928. DOI: 10.1111/jvim.14586. PubMed PMID: 27678080; PubMed Central PMCID: PMC5115200.
Lee D, Yun T, Koo Y, et al. Change of vertebral left atrial size in dogs with preclinical myxomatous mitral valve disease prior to the onset of congestive heart failure. J. Vet. Cardiol. 2022;42:23-33. Epub 20220511. DOI: 10.1016/j.jvc.2022.05.003. PubMed PMID: 35675727.
Crane SD, Elliott MW, Gilligan P, et al. Randomised controlled comparison of continuous positive airways pressure, bilevel non-invasive ventilation, and standard treatment in emergency department patients with acute cardiogenic pulmonary oedema. Emerg. Med. J. 2004;21(2):155-161. DOI: 10.1136/emj.2003.005413. PubMed PMID: 14988338; PubMed Central PMCID: PMC1726258.
Briganti A, Melanie P, Portela D, et al. Continuous positive airway pressure administered via face mask in tranquilized dogs. J. Vet. Emerg. Crit. Care (San Antonio). 2010;20(5):503-508. Epub 20100929. DOI: 10.1111/j.1476-4431.2010.00579.x. PubMed PMID: 20955301.
Staffieri F, Crovace A, De Monte V, et al. Noninvasive continuous positive airway pressure delivered using a pediatric helmet in dogs recovering from general anesthesia. J. Vet. Emerg. Crit. Care (San Antonio). 2014;24(5):578-585. Epub 20140814. DOI: 10.1111/vec.12210. PubMed PMID: 25123478.
Pouzot-Nevoret C, Hocine L, Negre J, et al. Prospective pilot study for evaluation of high-flow oxygen therapy in dyspnoeic dogs: the HOT-DOG study. J. Small Anim. Pract. 2019;60(11):656-662. Epub 20190716. DOI: 10.1111/jsap.13058. PubMed PMID: 31313305.
Felker GM, O’Connor CM, Braunwald E. Heart Failure Clinical Research Network I. Loop diuretics in acute decompensated heart failure: necessary? Evil? A necessary evil? Circ. Heart Fail. 2009;2(1):56-62. DOI: 10.1161/CIRCHEARTFAILURE.108.821785. PubMed PMID: 19750134; PubMed Central PMCID: PMC2742422.
Martens P, Mullens W. How to tackle congestion in acute heart failure. Korean J. Intern. Med. 2018;33(3):462-473. Epub 20180411. DOI: 10.3904/kjim.2017.355. PubMed PMID: 29627971; PubMed Central PMCID: PMC5943665.
Boswood A, Murphy A. The effect of heart disease, heart failure and diuresis on selected laboratory and electrocardiographic parameters in dogs. J. Vet. Cardiol. 2006;8(1):1-9. Epub 20060427. DOI: 10.1016/j.jvc.2005.12.001. PubMed PMID: 19083331.
Adin DB, Taylor AW, Hill RC, et al. Intermittent bolus injection versus continuous infusion of furosemide in normal adult greyhound dogs. J. Vet. Intern. Med. 2003;17(5):632-636. DOI: 10.1111/j.1939-1676.2003.tb02493.x. PubMed PMID: 14529128.
Ohad DG, Segev Y, Kelmer E, et al. Constant rate infusion vs. intermittent bolus administration of IV furosemide in 100 pets with acute left-sided congestive heart failure: A retrospective study. Vet. J. 2018;238:70-75. Epub 20180729. DOI: 10.1016/j.tvjl.2018.07.001. PubMed PMID: 30103918.
Gelzer AR, Kraus MS, Rishniw M, et al. Combination therapy with digoxin and diltiazem controls ventricular rate in chronic atrial fibrillation in dogs better than digoxin or diltiazem monotherapy: a randomized crossover study in 18 dogs. J. Vet. Intern. Med. 2009;23(3):499-508. DOI: 10.1111/j.1939-1676.2009.0301.x. PubMed PMID: 19645836.
Goutal CM, Keir I, Kenney S, et al. Evaluation of acute congestive heart failure in dogs and cats: 145 cases (2007-2008). J. Vet. Emerg. Crit. Care (San Antonio). 2010;20(3):330-337. DOI: 10.1111/j.1476-4431.2010.00524.x. PubMed PMID: 20636986.
Luca Ferasin
Dr. Ferasin graduated with honors in 1992 from the University of Bologna and spent three years doing endocrinology research at the BBSRC Institute in Cambridge, gaining his PhD in 1996 Read more
Thinking about doing an internship in the USA? This article delivers a brief guide to the advantages and disadvantages as to what one can offer.
Prescribing IV fluids for cats is not as straightforward as it may first appear; this paper offers a comprehensive overview of current knowledge.
Sepsis is a medical emergency in the dog; rapid identification and proactive intervention are essential for a positive outcome.
What do you do when the critical diabetic patient arrives at the emergency clinic? This paper offers a step-by-step approach for optimal results.
