Veterinary internships in the USA
Thinking about doing an internship in the USA? This article delivers a brief guide to the advantages and disadvantages as to what one can offer.
Issue number 32.3 Other Scientific
Published 12/04/2023
Also available in Français , Deutsch , Italiano and Español
Sepsis is a medical emergency in the dog; rapid identification and proactive intervention are essential for a positive outcome.

The presence of sepsis must be considered a medical emergency, and a rapid diagnosis is essential for a good outcome.
The transition from sepsis to multiple organ failure and septic shock can be prevented with fast, adequate resuscitation.
Antibiotic therapy is essential in critical patients with suspected sepsis and signs of shock.
Evaluate, re-evaluate, and re-evaluate again – sepsis is a situation that constantly progresses, requiring intensive monitoring of the response to treatment.
Sepsis is a medical emergency that has been recognized since the time of Hippocrates, around 400 years BC, when it was associated with any condition causing meat putrefaction, and the Persian physician Avicenna went on to describe sepsis as blood and tissue putrefaction with fever some 1,400 years later 1. Today we know that this condition is a systemic disease caused by an uncontrolled response of the immune system to an infectious condition. This can lead to dysfunction of one or more organs, which is the most common cause of mortality in sepsis patients, so early diagnosis and an effective treatment plan are extremely important.
The first definitions of sepsis in modern medicine were established in 1991 (known as Sepsis – 1), and although they have changed over the years (Sepsis – 2 and – 3), the primary concepts are still those most commonly used for practical purposes in veterinary medicine 2. The initial idea behind the definition was to standardize the criteria and establish the guidelines that aid recognition and treatment of patients with sepsis in the fastest and most effective way possible. These concepts include:
Table 1. Criteria for identifying a dog with SIRS.
| Temperature (°C/F) | ≤ 37.2/99.0 or ≥ 39.2/102.5 |
| Heart rate (beats/min) | ≥ 140 |
| Respiratory rate (breaths/min) | > 20 |
| White blood cells (x 103/µL) | ≥ 16 or ≤ 6 |
| Bands (%) | ≥ 3 |
Understanding the concept of SIRS is the key point. SIRS is the systemic response to any disorder causing inflammation, which can initially be either widespread, as in a patient with sunstroke, or localized, as in a case of acute pancreatitis. The body is unable to maintain the inflammatory response at the original focus, so that the inflammatory condition becomes generalized, and starts to affect normal tissues in other body areas that are unrelated to the original cause. Therefore, regardless of the initial cause, all body systems can show signs of inflammation. If the initial condition is of infectious origin, the patient is septic by definition; in other words, all septic patients show systemic inflammation (i.e., SIRS), but not all patients with SIRS are septic (e.g., injuries, sunstroke, pancreatitis).
Table 2. Definitions of organ dysfunction in the dog.
| Organ system | Criterion |
|---|---|
| Renal |
Urine output < 0.5 mL/kg/h for ≥ 12 hours or serum creatinine increase ≥ 0.3 mg/dL with no evidence of pre- or post-renal azotemia
|
| Cardiovascular |
Hypotension requiring the use of vasopressors
|
| Respiratory |
Need for administration of oxygen or assisted ventilation, based on clinical assessment, arterial gases (alveolar – arterial gradient > 10 mmHg and/or pulse oximetry (SpO2 < 95%)
|
| Liver |
Total serum or plasma bilirubin > 0.5 mg/dL
|
| Coagulation |
Prothrombin time or activated partial thromboplastin time > 25% of the upper reference limit and/or platelet count ≤ 100,000/µL
|
Based on the above concept, the first step is to establish if a patient is experiencing a systemic inflammatory response. The criteria used for this include changes in the respiratory rate (tachypnea), heart rate (bradycardia or tachycardia) and rectal temperature (hypothermia or fever), as well abnormal blood parameters (leukopenia or leukocytosis with or without left shift) (Table 1). The presence of two or more of these criteria in a dog is sufficient to confirm SIRS 3. The diagnostic value of these criteria lies in the simplicity; there is no need for more sophisticated tests, and it allows for rapid identification of an affected patient. The biggest problem is that the criteria are not specific, and most in-patients at a veterinary hospital will meet one or more of the SIRS criteria. However, as a general rule, the more criteria demonstrated by a patient, the higher the probability of SIRS. A dog with tachycardia, tachypnea, fever and leukocytosis is much more likely to suffer SIRS than one with tachycardia and tachypnea where, for instance, the stress and anxiety of being at the clinic might cause these changes.
The next step is to establish if the origin of the systemic inflammatory condition is infectious (i.e., sepsis) or not. For this, the clinical history and physical examination are necessary (Figure 1). In some cases – such as detection of a skin abscess following a bite – it is easy to establish the initial cause; in other cases, it may be more difficult, requiring a more comprehensive investigation. It is always advisable to perform at least one complete blood test (hematology and biochemistry) and a urine examination (including culture) whenever sepsis is suspected. Results of blood tests may show, for instance, the presence of leukocytosis or leukopenia, thrombocytopenia, hypoalbuminemia, hyperbilirubinemia, hypocalcemia, azotemia, and hyper- or hypoglycemia, which are the most common disorders in septic patients. These initial tests can be complemented with other diagnostics to search for the focus of infection, including radiography of the chest, abdomen and/or spinal column (Figure 2); abdominal and/or thoracic ultrasound and other imaging modalities if available (Figure 3); cerebrospinal fluid and/or joint punctures; serology and/or PCR for infectious diseases based on the clinical presentation and geographical location. As a general rule, any abnormal fluid accumulation must be examined by cytology, as the presence of intracellular bacteria will confirm infection, with a sample retained for subsequent culture and antibiogram testing as necessary (Figure 4). If a patient has suspected septic peritonitis but no intracellular bacteria can be found in the peritoneal fluid, blood glucose levels can be compared to the glucose concentration in the peritoneal fluid; a difference greater than 20 mg/dL (blood > peritoneal fluid) is consistent with the diagnosis of septic peritonitis 4. However, although an infectious cause may be suspected on initial assessment, it is not always possible to confirm this, so the clinician must consider other possibilities, or monitor the treatment response by frequent re-evaluation of the patient.

Figure 1. Congested oral mucous membranes in a dog with SIRS.
© Rocio Redondo

Figure 2. A lateral thoracic radiograph of a dog with aspiration pneumonia. Note the presence of alveolar bronchograms in right and left cranial lung lobes and the right middle lobe; there is also a generalized esophageal dilation.
© Rafael Obrador de Aguilar
The clinical presentation of a dog with sepsis can be highly variable, and whereas some animals will require intensive treatment to survive, others may be treated on an outpatient basis. Important points for stabilization of these patients are resuscitation, controlling the infection source, antibiotic administration, and the use of vasopressors (if septic shock is present).
 |
| a |
 |
| b |
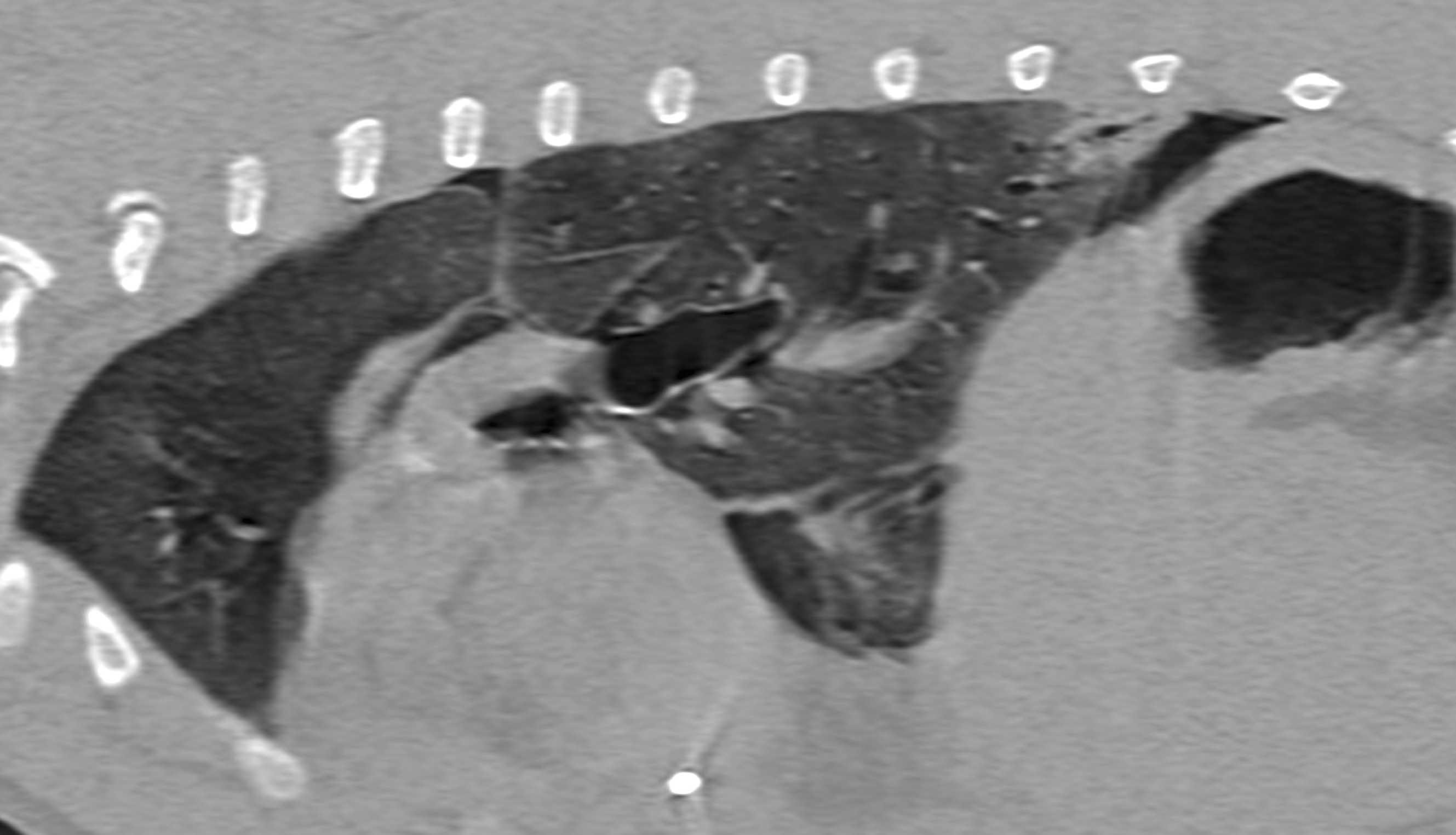
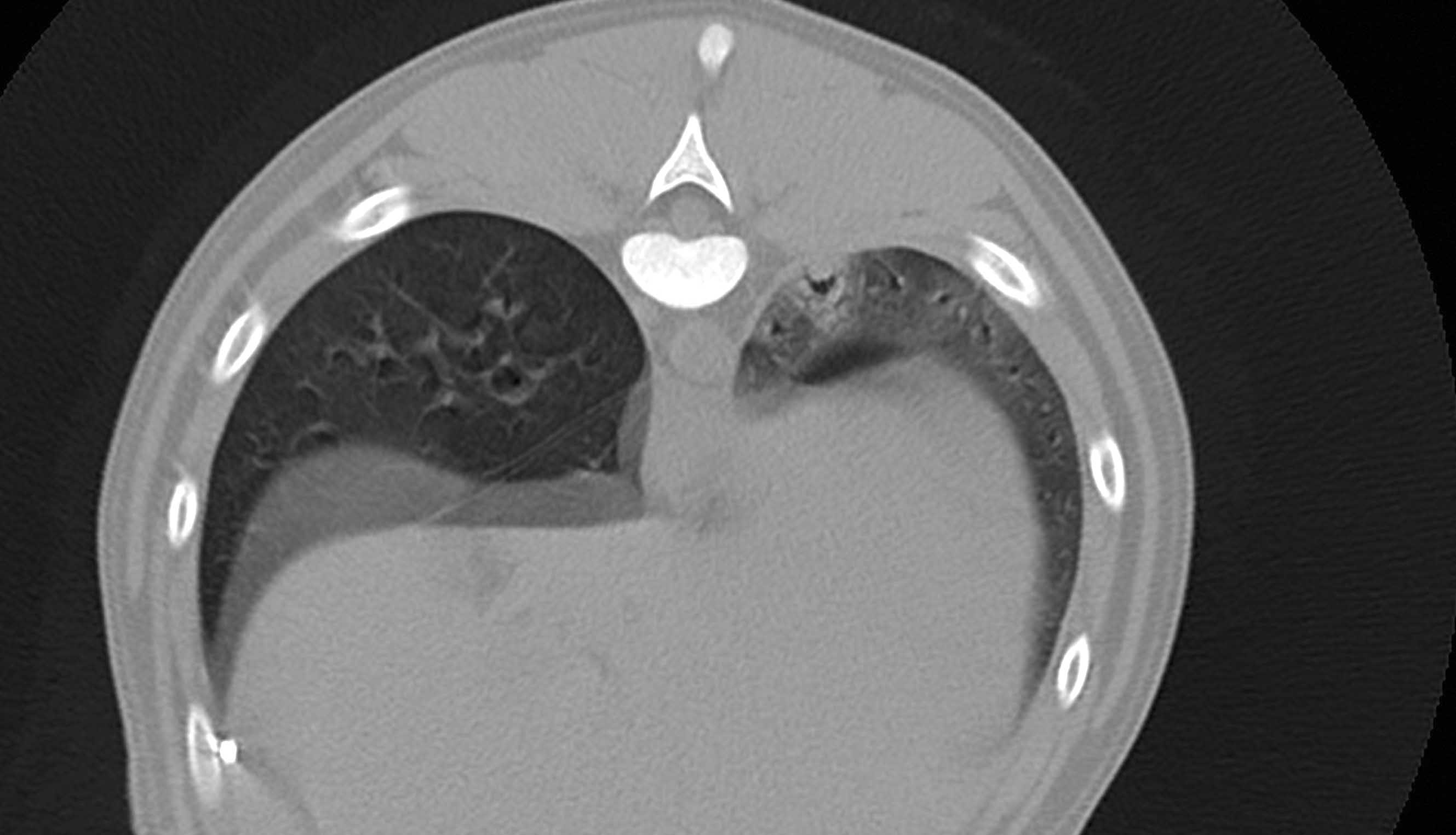
Figure 3. Sagittal (a) and transverse (b) images obtained by computerized tomography of a sepsis patient with pyothorax secondary to migration of a plant spike. The left caudal lung lobe is pushed away from the thoracic wall and shows a focal alveolar pattern; a small air bronchogram is also visible.
© Rafael Obrador de Aguilar
Resuscitation in a dog with sepsis differs from that in patients with other forms of shock. In sepsis, the cause of hemodynamic failure is the combination of absolute hypovolemia (which does benefit from fluid administration) and relative hypovolemia as a consequence of the increased vascular permeability that causes fluid loss, loss of vascular tone and (in some patients) myocardial dysfunction. The degree to which each of these factors can affect a given individual is highly variable and dynamic. It is logical to assume that a standardized treatment approach will not benefit all patients equally, and it is therefore much better to individualize the fluid therapy protocol for each animal based on its needs.
 |
 |
| a | b |
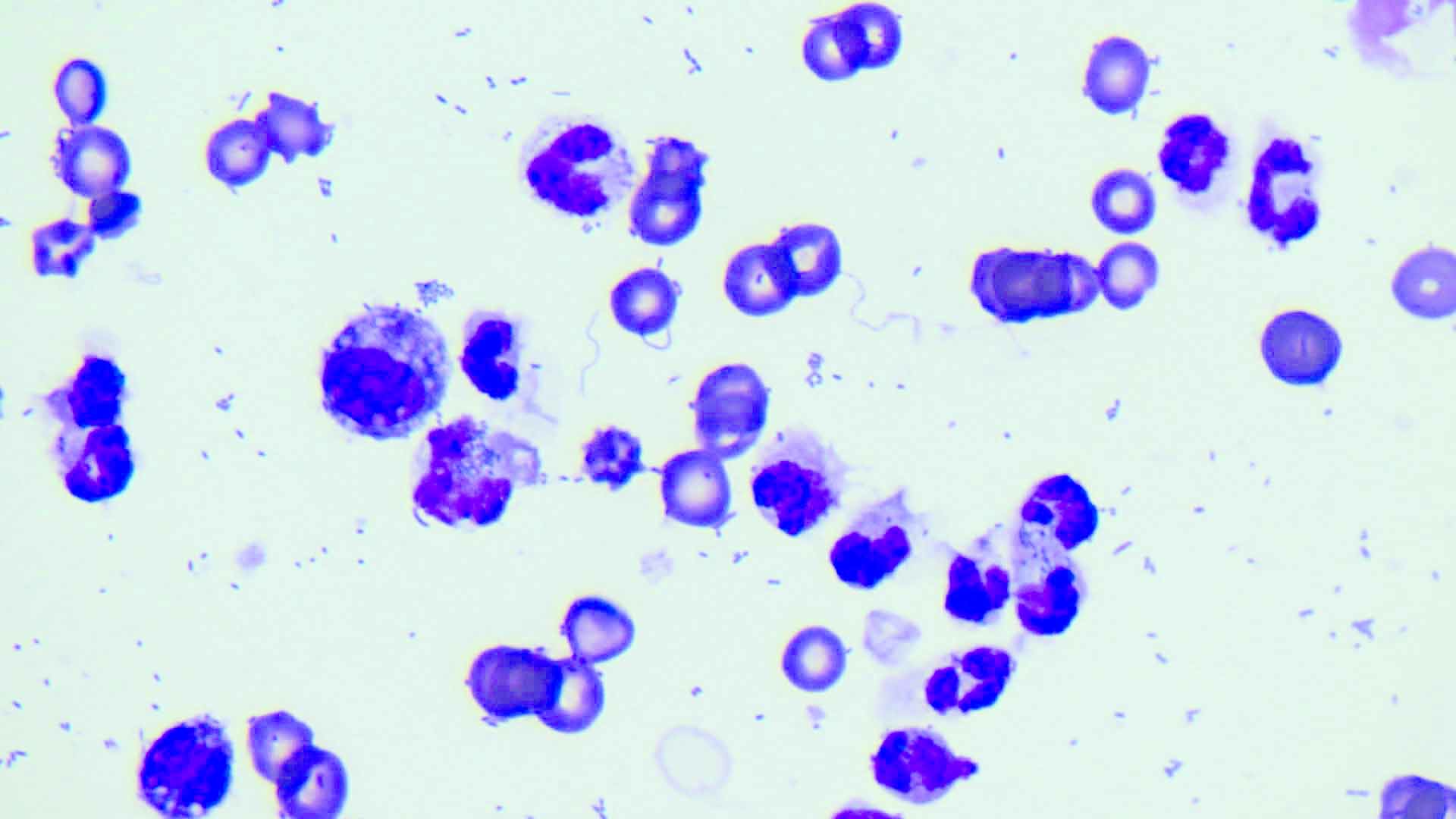
Figure 4. (a) Sample obtained by abdominocentesis in a patient with septic abdomen. (b) Cytology of the fluid shows the presence of intracellular bacteria (magnification x100).
© Rafael Obrador de Aguilar
Currently, although no strategy for fluid administration (type, volume and frequency) has been shown to be superior for treating septic patients, the time to resuscitation once hemodynamic disorders have been detected is very important; ideally this should be within 20-30 minutes wherever possible. In fact, early administration of fluids can modulate the inflammatory condition and even reduce the need for vasopressors (Figure 5), whereas delayed or inadequate administration is associated with a worsening of the hemodynamic condition, microcirculation blood flow, and organ dysfunction 5,6,7. It is therefore recommended to start resuscitation with a balanced isotonic crystalloid (e.g., Ringers Lactate) 8. An intravenous bolus (10-20 mL/kg over about 15 minutes) is generally administered while perfusion parameters are monitored (i.e., mental status, heartrate, color of mucosal membranes, capillary refill time, temperature of the extremities, pulse quality) for the purpose of improving or even returning these parameters to normal. If the patient’s response is not favorable (i.e., the perfusion parameters are not improving), the fluid bolus can be repeated two or three times, whilst checking that there are no signs which might indicate possible volume overload (i.e., tremor, nausea, wheezing, chemosis, serous nasal discharge, increased respiratory rate and/or effort, subcutaneous edema, crackles on pulmonary auscultation); where this occurs, further fluid administration will be harmful and is not indicated.
Once the patient is resuscitated, the fluid plan must be adjusted – as with any inpatient – depending on the animal’s dehydration status, daily physiological requirements and possible ongoing fluid losses. However, about 15% of patients with sepsis progress to septic shock, characterized by circulatory failure, multiple organ dysfunction and increased mortality. At this point even adequate fluid administration cannot reverse the consequent systemic hypotension, and the addition of vasopressors (see below) are necessary.

Figure 5. Fluid administration to a dog in septic shock should be started as soon as possible, using a balanced isotonic crystalloid.
© Shutterstock
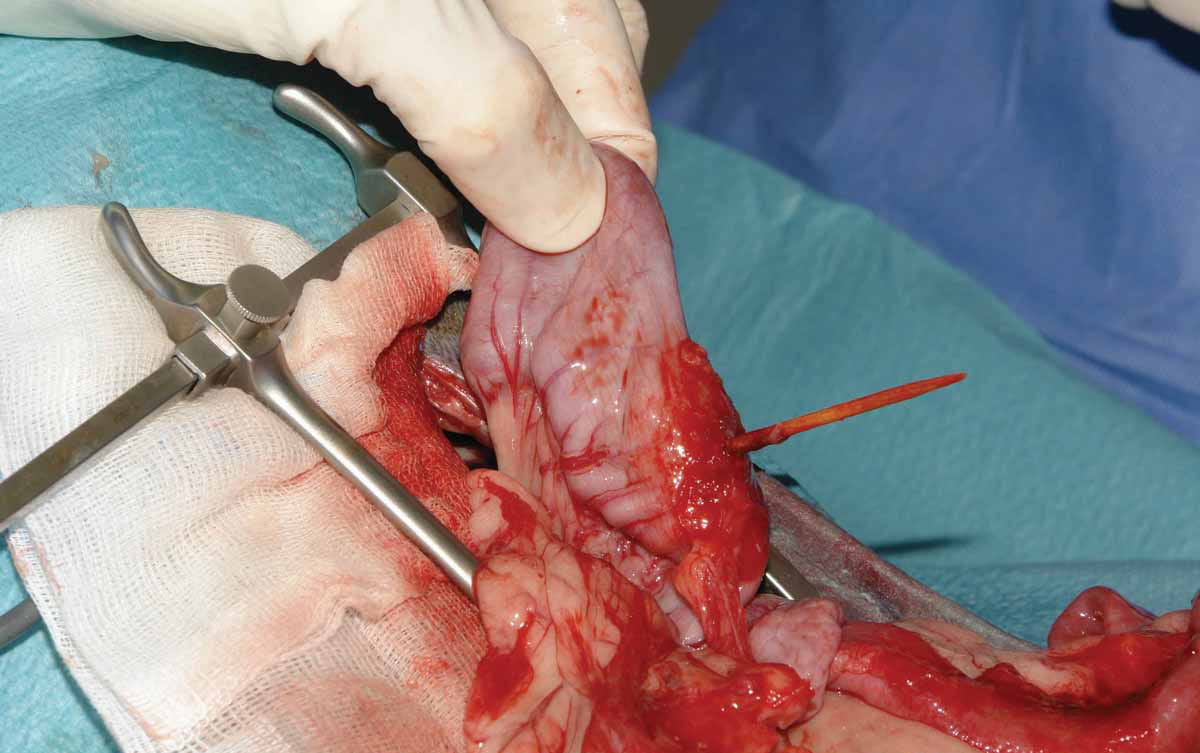
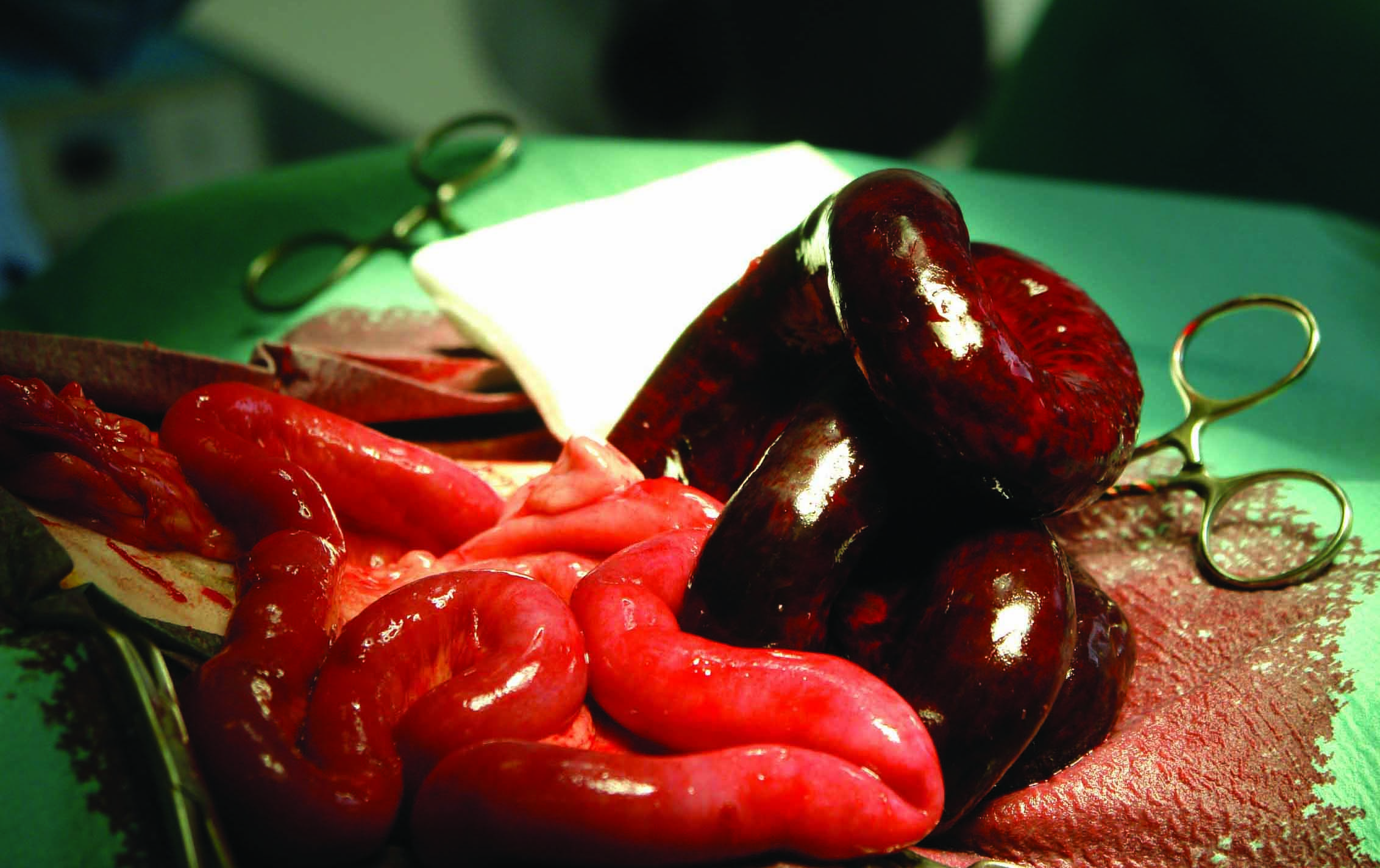
Controlling the focus of infection allows removal of the original cause and prevents possible progression to organ dysfunction in patients with sepsis. Therefore, once resuscitation is achieved, it is essential to drain or surgically remove the infected tissue as soon as possible, as it is known that adequate early removal of the infective focus has a positive impact on the patient’s prognosis (Figure 6) 9. This step also allows microbiological samples to be obtained for analysis.
 |
| a |
 |
| b |
 |
| c |
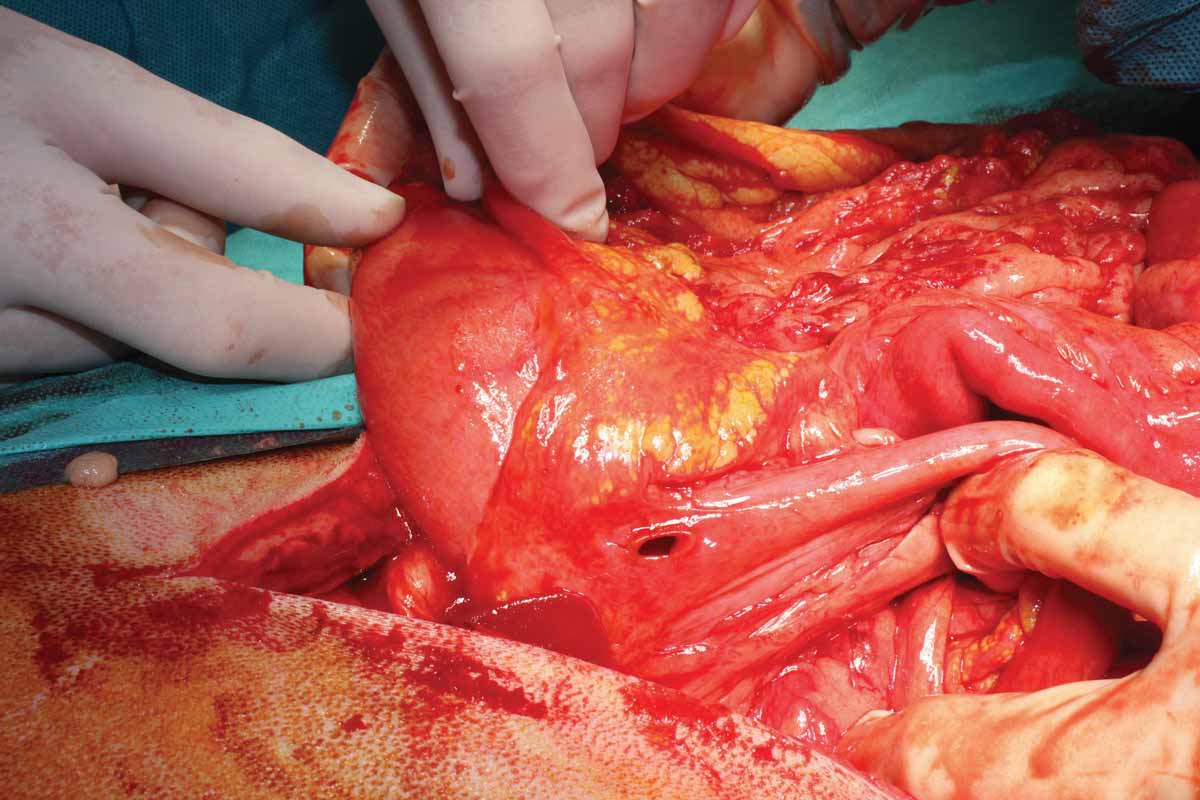
Figure 6. Examples of a septic abdomen: (a) duodenal perforation secondary to nonsteroidal anti-inflammatory drugs; (b) intestinal perforation from a foreign body; and (c) mesenteric torsion.
© Nuria Vizcaíno
Appropriate early antibiotic therapy is one of the main pillars for treating sepsis, and in particular for the most critical patients, such as those with organ dysfunction or septic shock. Wherever possible, and as long as it does not delay antibiotic administration, it is advisable to obtain samples for culture and susceptibility testing. Until sample results are obtained, empirical therapy is necessary, selecting a broad-spectrum antimicrobial which is likely to be effective against organisms that are potentially causing the problem. Other factors to be considered include the patient’s condition (i.e., does the preferred antibiotic require oral or intravenous administration) and the possibility for multi-resistant infections (from previous exposure to antibiotics, recent hospitalizations or community-acquired infection). The dose chosen must be appropriate for maximizing pathogen eradication whilst minimizing the chance of allowing antimicrobial resistance to develop, and avoiding adverse reactions. Some examples of combination therapy for patients with sepsis that require hospitalization and have not received antibiotics in the weeks prior to admission include 10:
The chosen empirical combination must be subsequently adjusted based on the patient’s progress and the results of the culture/susceptibility testing, a process called de-escalation. This involves adjusting the spectrum and number of antibiotics to the minimum essential to achieve adequate control of the infection once the organism has been identified. This demonstrates rational antibiotic usage, minimizing the chances of side effects and helping prevent bacterial resistance.
If systemic hypotension (systolic blood pressure < 90 mmHg) is detected in a septic patient with no response to fluid resuscitation (septic shock), treatment with vasopressors must be started as soon as possible 11. Early introduction of vasopressors in patients with septic shock is associated with lower volumes of resuscitation fluids, less accumulation of fluid within tissues, and a shortened duration of hypotension, which could reduce patient mortality 12. The aim is to try to increase systemic vascular resistance by improving blood pressure and thus maintain perfusion of vital organs. Systolic pressure must be maintained above 90-100 mmHg if the patient previously had normal blood pressure.
The two vasopressor drugs most commonly used in veterinary medicine are noradrenaline (norepinephrine) and dopamine (Table 3). Noradrenaline is a catecholamine that increases blood pressure mainly due to its vasoconstrictive properties, with a minimum effect on heart rate (marked α-1 effect and moderate β-1 effect). In addition, with adequate volume resuscitation, renal blood flow improves and lactate levels decrease. Dopamine is the natural precursor of noradrenaline in the body, although its effects are slightly different and are dose-dependent. In general, compared to noradrenaline, its effects at intermediate doses (3-10 µg/kg/min) are more marked on the heart (β-1) and weaker at a vascular level (α-1), so there is greater risk of tachycardia and/or arrhythmias, and the drug is less commonly used than noradrenaline. However, the choice ultimately depends on the veterinarian’s personal preference and familiarity.
Table 3. Vasopressors in the dog: mechanism of action and recommended doses.
| Vasopressor | α | β1 | β2 |
Contractibility | Heart rate | Vascular tone | Blood pressure | Dose |
|---|---|---|---|---|---|---|---|---|
| Dobutamine | + | + | ++ | ↑↑ | ↑ | ↓ | Variable | 5-20 µg/kg/min |
| Dopamine | ++ | + | ++ | ↑↑ | ↑↑ | ↑↑ | ↑↑ | 5-20 µg/kg/min |
| Adrenaline | +++ | +++ | +++ | ↑↑↑ | ↑↑↑ | ↑↑↑ | ↑↑↑ | 0.05-1.0 µg/kg/min |
| Noradrenaline | +++ | + | 0 | ↑ | Variable | ↑↑↑ | ↑↑↑ | 0.1-2.0 µg/kg/min |
| Vasopressin | 0 | 0 | 0 | 0 | ↓ | ↑↑ | ↑↑ |
0.5-5.0 mU/kg/min
|
Both drugs are administered by intravenous continuous rate infusion (CRI), initially at the lower end of the recommended dose before gradually increasing until the patient can be stabilized hemodynamically (systolic pressure > 90-100 mmHg). However, in some dogs with septic shock, hypotension may not improve despite fluids administration and high doses of vasopressors, a situation known as refractory shock 13. In these cases, the patient must be re-evaluated, and possible causes of refractory shock investigated (Table 4).
Table 4. Causes of refractory shock.
|
The administration of glucocorticoids is not recommended in septic patients if hemodynamic stability can be maintained with fluid therapy and/or vasopressors. However, if a dog with septic shock has not responded to volume expansion and hypotension persists despite high doses of catecholamines, corticosteroid administration must be considered 14,15. This class of drug increases the vascular α-adrenergic response and reduces vasodilation derived from inflammation. Recommended options include administration of an intravenous bolus of either hydrocortisone (1 mg/kg followed by a CRI at 0.08 mg/kg/h 16) or dexamethasone (using a CRI of 0.01-0.02 mg/kg/h).
The digestive tract is considered the “shock organ” in the canine species and is therefore susceptible to hypoperfusion problems, as blood flow is diverted to more important organs when needed. This effect, in combination with the presence of anorexia, can significantly affect permeability of the gastrointestinal barrier, with the possibility of bacterial (or endotoxin) translocation. In addition, septic patients that are anorexic are particularly susceptible to malnutrition, hence the importance of starting nutrition as soon as possible, but the patient must first be hydrated and hemodynamically stable, with no significant electrolyte disorders. Nutrition can be voluntary or through the use of feeding tubes if the patient is anorexic. The first objective is to provide nutrients to the enterocytes to maintain gastrointestinal barrier function, and therefore may initially be below the dog’s resting energy requirements (RER) (calculated at 70 x (weight in kg)0.75).
Changes in blood glucose regulation are frequent seen in septic patients, with hyperglycemia in the very early stages, followed by hypoglycemia as the severity of the disease worsens. It is recommended to maintain glucose levels between 70-140 mg/dL. For hypoglycemic patients an intravenous glucose bolus must be administered (0.5-1.0 mL/kg of dilute 50% glucose). If the response to the bolus is transient, maintenance fluids can be supplemented with glucose (2.5-5%). Where blood glucose levels continuously exceed 150 mg/dL, regular insulin given by CRI (0.05-0.1 IU/kg/h) can be started, with intensive monitoring of blood glucose levels.
Rafael Obrador de Aguilar
The prognosis of septic patients depends on the severity of the condition, the initial underlying cause, and the presence or absence of organ dysfunction. In dogs with uncomplicated sepsis, the prognosis is good, with survival rates around 84%. However, once an animal starts to experience organ dysfunction, or septic shock is established, the prognosis worsens markedly. In fact, mortality is clearly associated with organ dysfunction, and increases with the number of organ systems affected 17. However, tissue damage need not to be irreversible, and it can subside in part or completely if the underlying cause of the inflammation is successfully treated.
Sepsis is a medical emergency where the prognosis depends on how fast some procedures are undertaken for both the diagnosis and therapeutic management of the patient. Evaluation and re-evaluation should be performed as often as necessary, as it is a constantly progressing condition that requires intensive monitoring of treatment response.
Funk DJ, Parrillo JE, Kumar A. Sepsis and septic shock: a history. Crit. Care Clin. 2009;125(1):83-101.
Bone RC, Balk RA, Cerra FB, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis: the ACCP/SCCM consensus conference committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 1992;101(6): 1644-1655.
Greiner M, Wolf G, Hartmann K. A retrospective study of the clinical presentation of 140 dogs and 39 cats with bacteraemia. J. Small Anim. Pract. 2008;49(8):378-383.
Bonczynski JJ, Ludwig LL, Barton LJ, et al. Comparison of peritoneal fluid and peripheral blood pH, bicarbonate, glucose, and lactate concentration as a diagnostic tool for septic peritonitis in dogs and cats. Vet. Surg. 2003;32(2):161-166.
Leisman DE, Doerfler ME, Schneider SM, et al. Predictors, prevalence, and outcomes of early crystalloid responsiveness among initially hypotensive patients with sepsis and septic shock. Crit. Care Med. 2018;46(2):189-198.
Morley PT. Early fluid management in sepsis: yes. Crit. Care Med. 2018;46(2):327-328.
Ospina-Tascon G, Neves AP, Occhipinti G, et al. Effects of fluids on microvascular perfusion in patients with severe sepsis. Intens. Care Med. 2010;36(6):949-955.
Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intens. Care Med. 2017;43:304-377.
Rausei S, Pappalardo V, Ruspi L, et al. Early versus delayed source control in open abdomen management for severe intra-abdominal infections: a retrospective analysis on 111 cases. World J. Surg. 2018;42(3):707-712.
Hardie, E. Therapeutic management of sepsis. In; Bonagura J (ed): Kirk´s Current Veterinary Therapy: XIII Small Animal Practice Philadelphia: WB Saunders, 2000;272-275.
Cecconi M, De Backer D, Antonelli M, et al. Consensus on circulatory shock and hemodynamic monitoring. Task Force of the European Society of Intensive Care Medicine. Intens. Care Med. 2014;40(12):1795-1815.
Ospina-Tascón G, Hernandez G, Alvarez I, et al. Effects of very early start of norepinephrine in patients with septic shock: a propensity score-based analysis. Crit. Care 2020;24(1):52.
Kimmoun A, Ducrocq N, Levy B. Mechanisms of vascular hyporesponsiveness in septic shock. Curr. Vasc. Pharmacol. 2013;11:139-149.
Annane D, Pastores SM, Rochwerg B, et al. Guidelines for the diagnosis and management of critical illness-related corticosteroid insufficiency (CIRCI) in critically ill patients (part I): Society of Critical Care Medicine (SCCM) and European Society of Intensive Care Medicine (ESICM) 2017. Intens. Care Med. 2017;43:1751-1763.
Pastores SM, Annane D, Rochwerg B. Corticosteroid Guideline Task Force of SCCM and ESICM. Guidelines for the diagnosis and management of critical illness-related corticosteroid insufficiency (CIRCI) in critically ill patients (part II): Society of Critical Care Medicine (SCCM) and European Society of Intensive Care Medicine (ESICM) 2017. Crit. Care Med. 2018;46:146-148.
Summers AM, Culler C, Yaxley PE, et al. Retrospective evaluation of the use of hydrocortisone for treatment of suspected critical illness-related corticosteroid insufficiency (CIRCI) in dogs with septic shock (2010-2017): 47 cases. J. Vet. Emerg. Crit. Care 2021;31:371-379.
Kenney EM, Rozanski EA, Rush JE, et al. Association between outcome and organ system dysfunction in dogs with sepsis: 114 cases (2003-2007). J. Am. Vet. Med. Assoc. 2010;236(1):83-87.
Rafael Obrador de Aguilar
Dr. Obrador obtained his veterinary science degree from the University of Zaragoza Read more
Thinking about doing an internship in the USA? This article delivers a brief guide to the advantages and disadvantages as to what one can offer.
Prescribing IV fluids for cats is not as straightforward as it may first appear; this paper offers a comprehensive overview of current knowledge.
Acute heart failure in dogs carries a serious risk of death, and optimizing diagnosis and treatment is paramount, as discussed by Luca Ferasin.
What do you do when the critical diabetic patient arrives at the emergency clinic? This paper offers a step-by-step approach for optimal results.
