Petfood: how to prevent insect infestation
Insect contamination of petfoods is a potential problem in tropical countries; this paper offers an overview of the situation and how the risks can be minimized
This article contains sensitive photo that may be harmful for young children.
Hepatic lipidosis, the most common feline liver disease, is associated with significant metabolic derangements, liver injury and failure, and potentially life-threatening complications.
Regardless of the instigating cause and whether it has resolved, once it develops, nausea follows, and a vicious cycle of anorexia and hepatic lipid accumulation ensues.
Nutritional support constitutes the cornerstone of treatment for hepatic lipidosis, and will commonly entail the use of a feeding tube.
While hepatic lipidosis is associated with significant morbidity, requires intensive treatment and entails considerable expense, treatment is often successful, and recurrence seems rare.
First described in 1977, hepatic lipidosis (HL) has since emerged as the most commonly diagnosed hepatobiliary disease in cats 1,2. Accumulation of triglycerides within hepatocytes engenders hepatocyte swelling and intrahepatic cholestasis, oxidative damage, secondary inflammation, and ultimately hepatic dysfunction 1,2. Metabolic complications and systemic ramifications of HL are diverse, and include electrolyte imbalances, insulin resistance, pancreatitis, hyperammonemia, hepatic encephalopathy and coagulopathies. Therefore, feline hepatic lipidosis (FHL) is associated with significant morbidity and often requires hospitalization for initial stabilization, with the possibility of several weeks of intensive treatment for complete recovery, and it carries a guarded-to-favorable prognosis overall.
Feline HL is defined by abnormal accumulation of lipid-filled vacuoles within hepatocytes. In severe cases, fat accounts for over 30% of the total liver weight, in striking contrast to normal cats where hepatic fat content rarely exceeds 1-5% 3,4. The predominant fat type in FHL is adipose tissue-derived triglycerides, from enhanced lipolysis during catabolic states of starvation, rather than de novo synthesis of triglycerides in the liver 2,3. Furthermore, impaired oxidation and utilization of fat within hepatocytes, diminished redistribution of fat from the liver to peripheral tissues, and altered lipoprotein metabolism all contribute to the development of FHL 4,5. Whether an individual propensity to develop FHL exists is debatable; a female predisposition is infrequently reported 1,6,7, but there is no obvious gender or breed predilection. The most salient risk factor for FHL is probably obesity, owing to pre-existing abnormalities in lipid and carbohydrate metabolism and higher fat reserves 2. Nonetheless, HL has been shown to develop irrespective of the body condition score 8, and FHL should always be suspected when compatible history, clinical findings and laboratory abnormalities exist.
Experimentally, HL develops after several weeks of restricted caloric intake, but in the clinical setting periods of anorexia as short as two days have been reported to precede the diagnosis 1,6,7,9. These reports, however, are subjective, relying on owner perception of food intake by their cat, and must therefore be cautiously interpreted. Maintenance energy requirements should be reduced by more than 50% for HL to develop, but the composition of the diet, rather than its caloric density alone, is also important 2. A diet deficient in essential amino acids (AA) might predispose to hepatic lipid accumulation, while L-carnitine supplementation can protect against FHL even in the face of severe caloric restriction 2,10.
Cats are obligate carnivores, whose capacity to endogenously synthetize many essential fatty acids and AA has been greatly diminished or lost over time. Furthermore, feline carbohydrate metabolism is different from omnivorous species, with cats having a lower overall requirement for carbohydrates, a propensity to utilize fats and proteins over carbohydrates for energy, and a tendency to favor AA-dependent pathways of gluconeogenesis to maintain normoglycemia 1,2. These peculiarities are reflected in their diet, which is primarily protein and fat-based, and might also account for their proclivity to accumulate fat in the liver in states of food deprivation. For example, compared to dogs and humans, endogenous production of several long-chain polyunsaturated fatty acids is reduced in cats. These fatty acids protect against HL by promoting triglyceride oxidation and glycogen synthesis rather than lipogenesis, but these protective properties are lost when dietary intake is reduced, which may contribute to the development of FHL 1. Cats also have limited capacity to synthesize enough arginine, methionine, cysteine and taurine. Depletion of these essential AA interferes with beta-oxidation of non-esterified fatty acids (NEFA), diminishes the production of very low density lipoprotein particles which transport triglycerides from the liver to the rest of the body, and reduces endogenous L-carnitine production, thereby preventing transportation of NEFA to mitochondria 1,2. Lastly, impaired insulin secretion and responsiveness in peripheral tissues are common in obese cats and in cats with FHL 4, and this promotes lipolysis and mobilization of NEFA from peripheral tissues. Collectively, these dietary deficiencies, metabolic derangements and hormonal changes culminate in the development of FHL.
Protracted periods of hyporexia/anorexia always precede the development of FHL 6,7,9, which in turn may result from inadvertent food deprivation, decreased palatability of new diets, or stressful environmental situations (e.g., boarding, change of ownership, moving to a new house). Secondary FHL implies the presence of an identifiable underlying disease and occurs in 50-95% of cats 1,6,7,9. Therefore, a thorough questioning and diagnostic work-up is warranted to identify and address any possible underlying cause (Table 1).
| Primary FHL (5-51%) | |
|---|---|
| Decreased food intake |
|
| Secondary FHL (49-95%) | |
| Oral diseases |
|
| Decreased food intake (pathological) |
|
| Endocrinopathies |
|
| Hepatobiliary diseases |
|
| Pancreatic diseases |
|
| Urogenital diseases |
|
| Gastrointestinal |
|
| Neurological diseases |
|
| Miscellaneous |
|
* FIP: Feline infectious peritonitis
** FeLV: Feline leukemia virus
Weight loss is invariably present, as the result of decreased caloric intake. Even in cats with an ostensibly normal bodyweight, a more careful physical examination often reveals a decreased body condition score and sarcopenia, especially in the hindlimbs and epaxial muscles. Lethargy, weakness, clinical signs referable to the gastrointestinal tract (e.g., vomiting, diarrhea, constipation), dehydration and hepatomegaly are frequent complaints and findings. Jaundice is common, and is best appreciated in the sclera or pinnae, in addition to mucosal surfaces 6,7,9 (Figure 1). Ptyalism, albeit a non-specific sign, might be the only clinical manifestation of hepatic encephalopathy (HE) in FHL, and occurs in a minority of cases. Furthermore, ptyalism at presentation has been associated with a worse outcome 9. Other etiologies of ptyalism include oral diseases, nausea, and idiosyncratic reactions to orally administered medications.
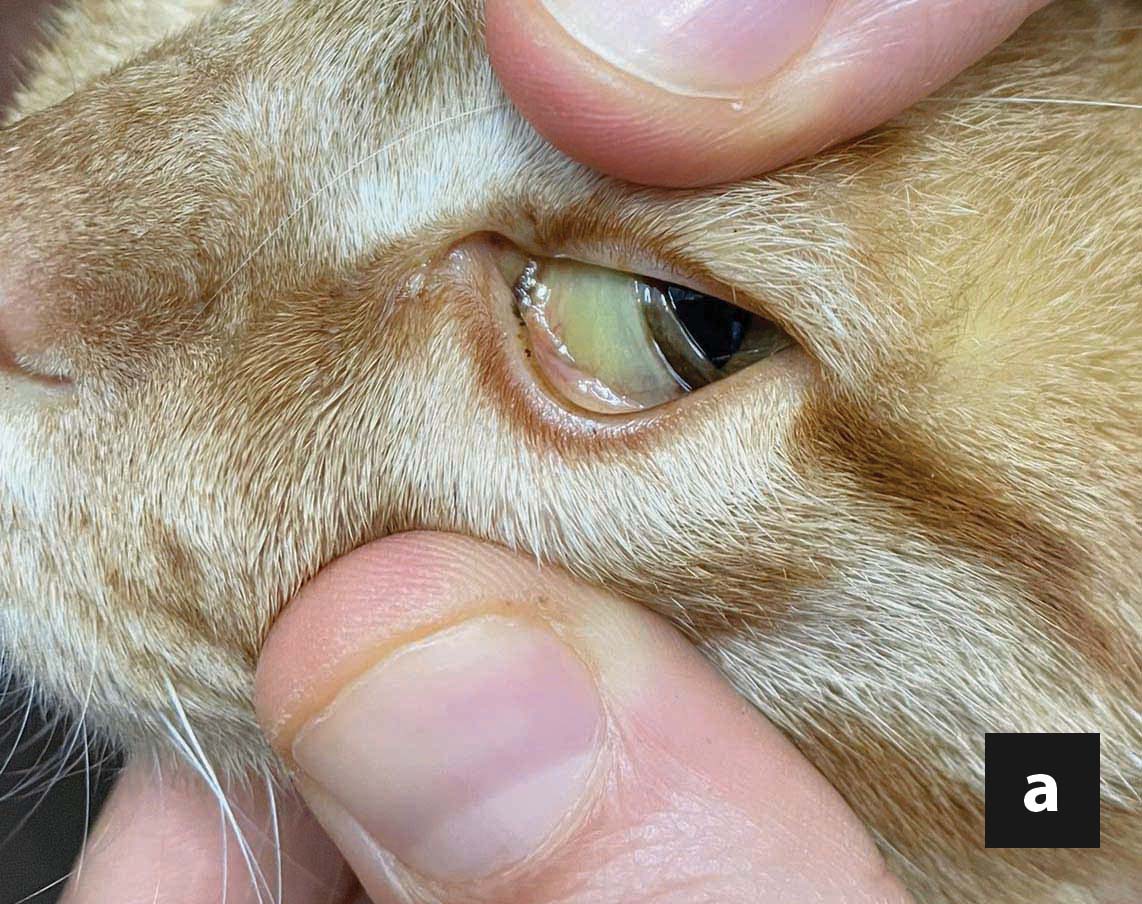
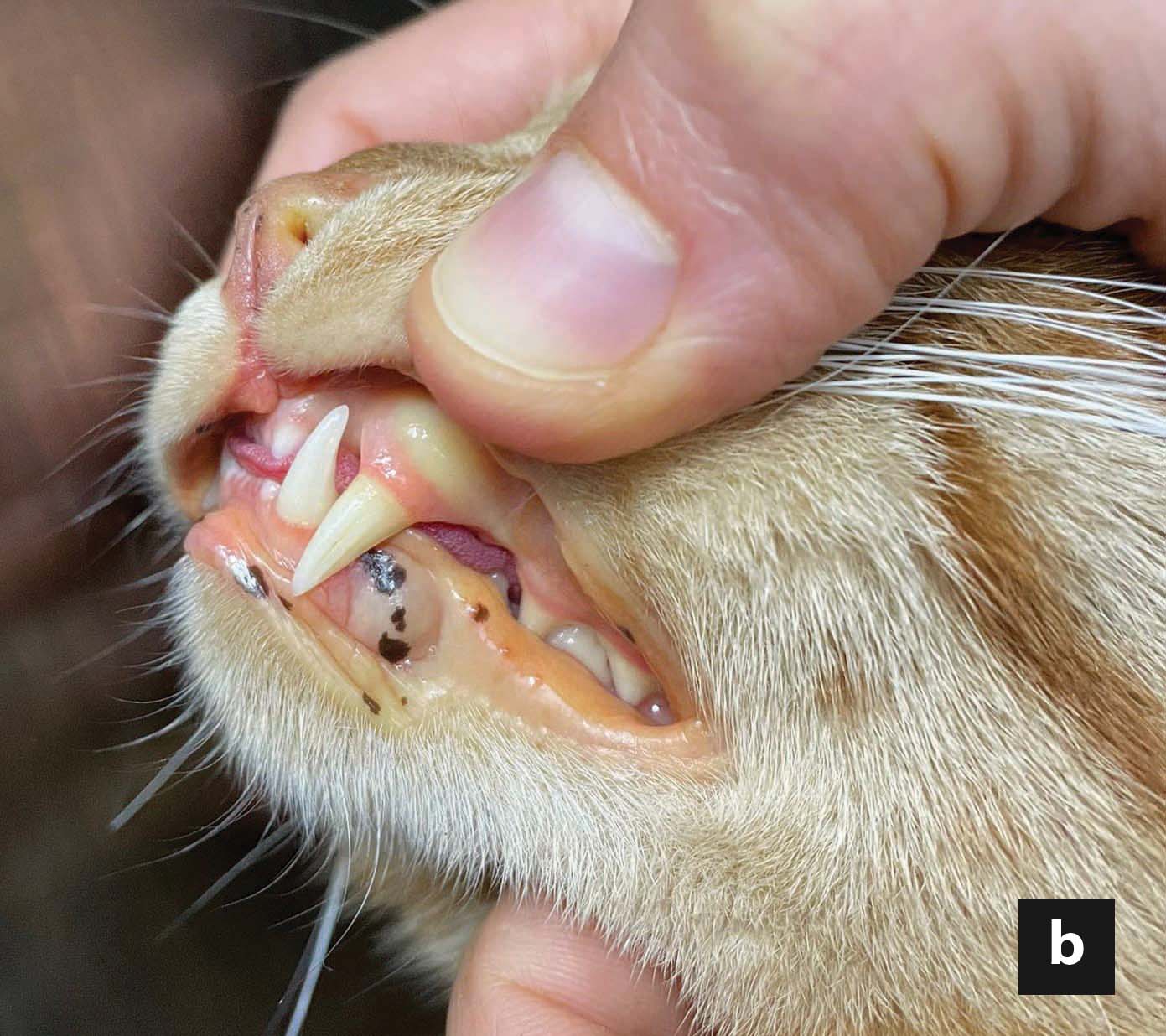
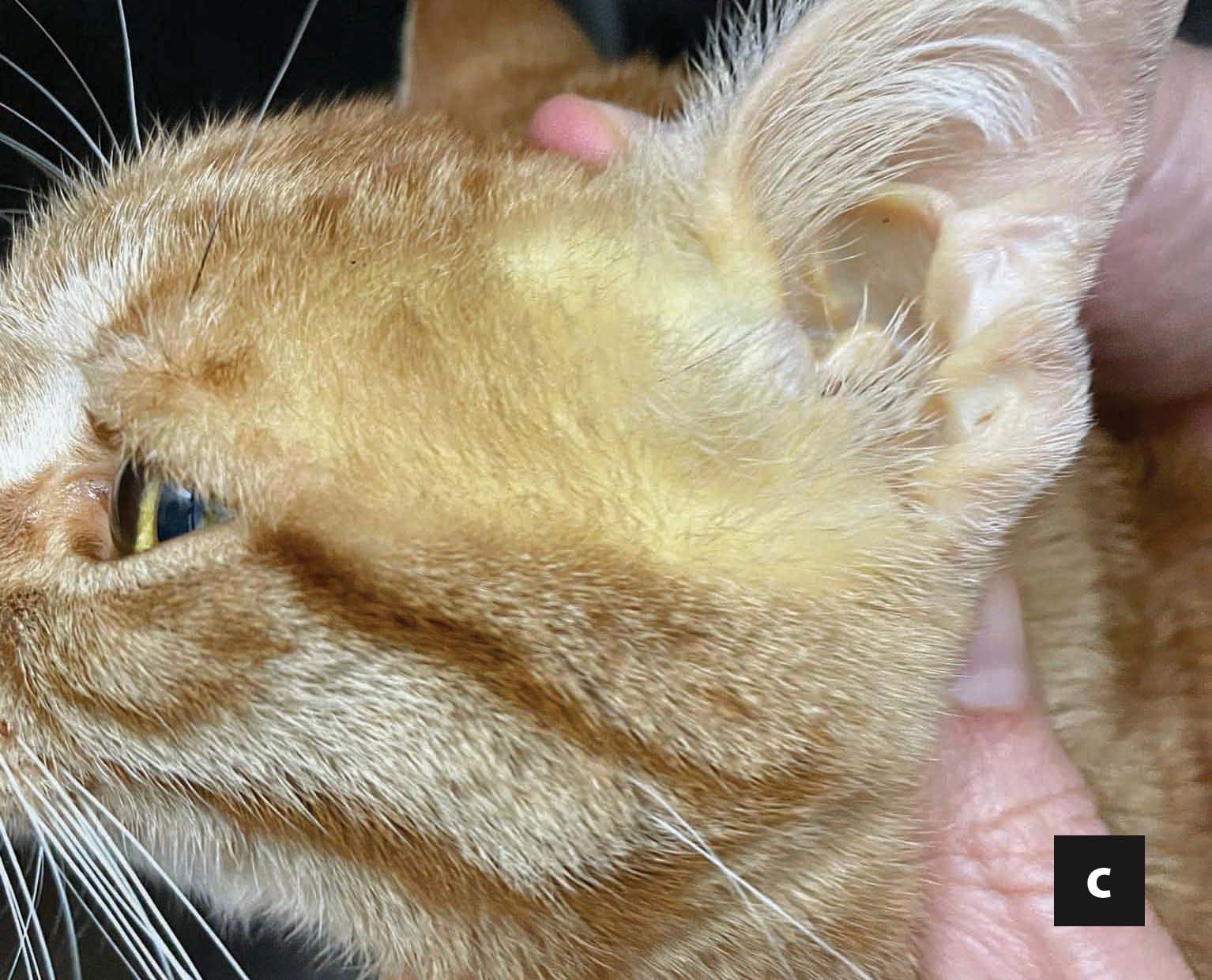
Figure 1. Jaundice in a 4-year-old, castrated male cat with idiopathic hepatic lipidosis. Jaundice is more readily discernible in the third eyelid (a) and sclera, compared to the gingival mucosa (b) in non-anemic cats. Jaundice can also be observed in the pinna (c) and other non-furred skin surfaces.
© Ran Nivy
Additional, less frequent clinical signs can arise secondary to FHL-associated complications (e.g., bleeding diathesis; HE; skin fragility syndrome) or the underlying etiology (e.g., fever with infections or inflammatory diseases such as pancreatitis; polyuria and polydipsia in cats with kidney disease or diabetes, etc.).
The biochemical hallmark of FHL is increased alkaline phosphatase activity (ALP), which occurs in > 80% of affected cats 7,9,11. Experimentally, increased ALP levels precede the development of hyperbilirubinemia 8, and this is considered a highly sensitive and rather specific marker of FHL. Conversely, gamma-glutamyl transpeptidase (GGT) activity is seldom increased, unless a concurrent cholestatic/biliary pathology coexists 11. A disparate increase in ALP activity in the face of a normal or slightly increased GGT activity is therefore suggestive of FHL. Nonetheless, additional differential diagnoses for increased ALP activity do exist, including hepatobiliary (e.g., cholangiohepatitis; biliary obstruction) and hormonal (i.e., hyperthyroidism) diseases.
Hyperbilirubinemia and increased alanine (ALT)/aspartate (AST) aminotransferase activities are common, non-specific findings 7,9. Hepatic fat accumulation, irrespective of etiology, causes cellular swelling and intrahepatic cholestasis, as well as oxidative damage and secondary inflammation, which result in increased transaminase activities – therefore, abnormal hepatic transaminase levels do not necessarily imply an additional underlying hepatic pathology. A higher-fold increase in ALT/AST activities (relative to their respective reference intervals) compared to that of ALP activity, might suggest an additional underlying disease 7.
Low urea concentration can result from decreased protein intake, and malfunction of the urea cycle. The latter is aggravated by arginine and vitamin B12 deficiencies, which commonly occur in FHL. Similarly, hyperammonemia can develop secondary to hepatic failure, vitamin B12 deficiency and inadequate dietary intake of arginine, and has clinical and therapeutic implications 1,2. Additional, infrequently observed derangements are non-specific and include hypoalbuminemia/hypoproteinemia from protein losing enteropathy and/or hepatic synthetic failure, hypocholesterolemia and cholestasis-associated hypercholesterolemia 7,9. Hypertriglyceridemia is common in obese cats in general, and during FHL in particular 12. Hyperglycemia can develop secondary to insulin resistance, pancreatitis or overt diabetes mellitus 7,9. Hypoglycemia, on the other hand, is seldom observed 9, but might arise secondary to hepatic failure, sepsis or pancreatitis, and constitutes a negative prognostic factor 13.
In terms of electrolyte derangement, hypokalemia is perhaps the most common and clinically significant finding in cats with FHL 1. Common complications include muscular weakness, inability to adequately concentrate urine with development of polyuria/polydipsia, gastric and intestinal ileus, exacerbation of HE, and cardiac dysfunction in more severe cases. The true magnitude of potassium depletion is often masked by dehydration upon admission, and instituting enteral and parenteral feeding can exacerbate hypokalemia 14,15. Therefore, close monitoring and correction of blood potassium concentration is crucial. Additional, less frequent electrolyte abnormalities include hypomagnesemia and hypophosphatemia 1,14,15. These may be present upon admission (secondary to intestinal and urinary loss), but often develop later in the course of disease following rehydration and commencement of parenteral or enteral feeding. Development thereof has deleterious ramifications, including muscular/cardiac and neurological manifestations (hypophosphatemia and hypomagnesemia), hemolytic anemia and thrombocytopenia (hypophosphatemia) and the development of refractory hypokalemia and hypocalcemia (hypomagnesemia).
Changes in the complete blood count are diverse and non-specific, and can develop secondary to the underlying disease or as a complication of FHL. Changes in erythrocyte morphology include poikilocytosis, microcytosis and Heinz bodies 1,6,7. The last is the result of increased oxidative stress and hypophosphatemia and might contribute to the development of anemia. Alternatively, severe hypophosphatemia can directly result in hemolytic anemia 14. Microcytosis, when severe, is suggestive of iron deficiency or hepatic vascular diseases (e.g., portosystemic shunts).
Coagulopathies are common in cats with HL 1,7 and in one report more than 90% of cases had abnormally prolonged prothrombin time (PT) and/or activated partial thromboplastin time (aPTT) 9. When comparing the proteins induced by vitamin K absence test (PIVKA) to PT/aPTT tests, the sensitivity of the former is significantly higher in detecting vitamin-K associated coagulopathies in cats with hepatic and intestinal diseases 16. However, since PIVKA measurement is currently commercially unavailable, and given the high prevalence of coagulopathies and vitamin K deficiency in FHL, vitamin K supplementation is advised (Table 2).
Table 2. Frequently used medications for feline hepatic lipidosis.
| Treatment | Dose | Comments | |
|---|---|---|---|
| Vitamin supplementation | |||
| Cyanocobalamin (B12) | 250 mcg/cat once a week SC/IM or 250 mcg/cat q24h PO; both for 6-8 weeks | Vitamin B complex preparations for IV or SC/IM injections might be considered, but the quantities of individual vitamins might be insufficient in some products | |
| Vitamin K | 1-1.5 mg/kg q12-24h SC/IM, (x3 injections) | ||
| Thiamine |
100 mg/cat q24h SC/IM |
||
| Vitamin E | 50-100 IU/cat q24h PO | As an antioxidant | |
| Antioxidants and nutraceuticals | |||
| Ursodeoxycholic acid | 10-15 mg/kg q12h PO | Minimal side effects (diarrhea, nausea); taurine depletion | |
| L-carnitine | 200-250 mg/cat q24h, PO | ||
| S-adenosyl-methionine (SAMe) | 20 mg/kg q24h PO on an empty stomach | Minimal side effects (inappetence, vomiting) | |
| Silymarin | 5-10 mg/kg q24h PO | Phosphatidylcholine conjugates are preferable owing to improved absorption | |
| N-acetylcysteine | 70 mg/kg (diluted with saline/D5W*) over 15-30 mins, q8-12h IV | Vomiting with rapid administration; use until oral treatment with SAMe/silymarin is feasible | |
| Taurine | 250-500 mg/kg/cat q24 PO | Most commercial diets provide sufficient taurine | |
| Potassium supplementation | |||
| KCL | Serum concentration (mEq/L) | KCL added (mEq/100 mL) | The final dose changes with the fluid rate; do not exceed 0.5 mEq/kg/h. If hypophosphatemia coexists, KPO4 can be used with KCl in varying combinations to achieve the same final potassium concentration in the fluid solution; often a KPO4:KCl ratio of 1:1 is used, unless severe phosphate deficits exist; avoid calcium-containing fluids with KPO4 |
| < 2 | 8 | ||
| 2.1-2.5 | 6 | ||
| 2.6-3 | 4 | ||
| 3.1-3.5 | 3 | ||
| 3.6-5 | 2 | ||
* dextrose 5% in water
Definitive diagnosis of FHL can often be achieved by ultrasound-guided fine-needle aspiration (FNA) of the liver 17, typically using a long, 22/23G hypodermic or spinal needle. Lipid vacuoles appear as sharply demarcated, clear vesicles within the cytosol, unlike the irregular, feathery glycogen-filled hepatic vacuoles (Figure 2). Special stains can help differentiate between the two but are rarely indicated. The presence of microvesicular and macrovesicular, distinct cytosolic vacuoles within hepatocytes from cytological specimens is often sufficient to establish diagnosis of FHL, and FNA is associated with a lower prevalence of significant complications (most notably bleeding) when compared to hepatic biopsies. Rarely, a liver FNA will reveal an underlying or concurrent hepatic pathology, but agreement between liver biopsies and FNA is poor 18,19 and cytological interpretation of inflammatory infiltrates in the liver is hindered by blood contamination. Furthermore, infectious hepatic diseases are seldom diagnosed cytologically in FNA samples, but conversely, neoplastic infiltrates, particularly round-cell tumors, can easily be diagnosed. Thus, the shortcomings of hepatic FNA must be recognized, and additional diagnostic procedures might be considered under specific circumstances.
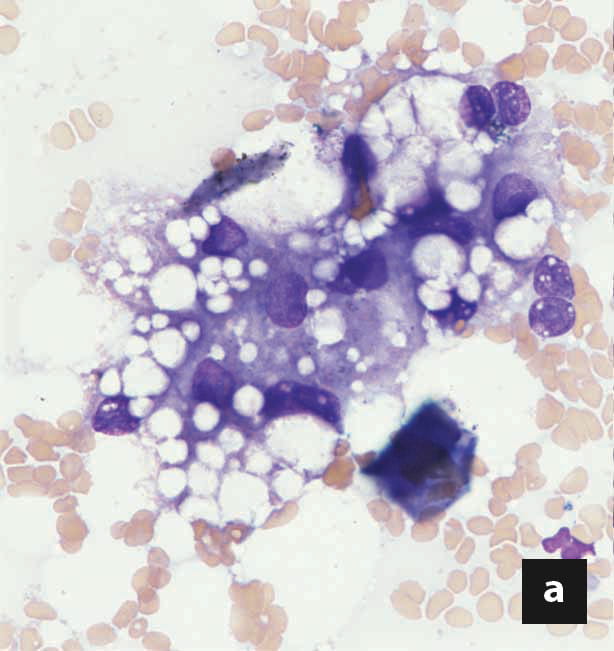
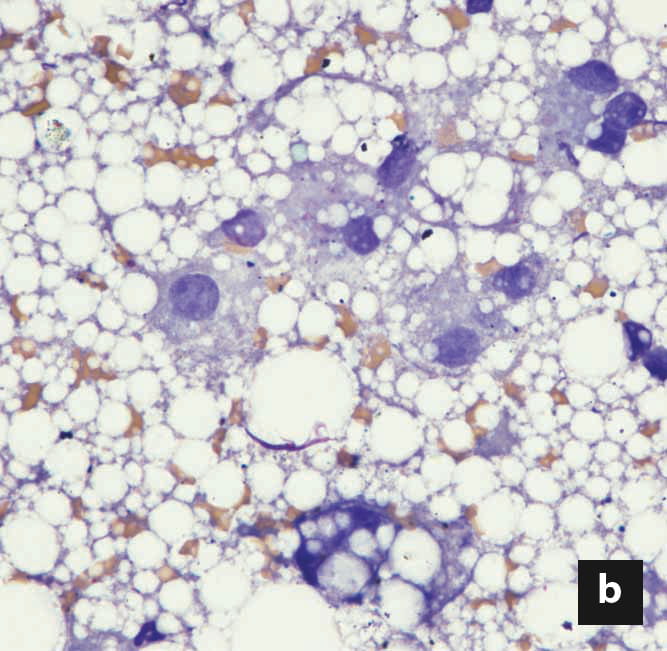
Figure 2. Liver cytology from a cat with hepatic lipidosis. Notice the distinct fat vacuoles of varying sizes within the hepatocytes, which displace the nucleus peripherally in more severe cases (a, b). Not uncommonly, free fat vesicles and adipocytes are also found in liver aspirates, especially when the adjacent adipose tissue is inadvertently sampled. (x60 magnification)
© Courtesy of Dr. Sharon Kuzi
Apart from its utility in guiding FNA sampling, abdominal ultrasound is a useful imaging modality to rule out concurrent or underlying diseases that might have precipitated FHL 17. Sonography of the liver in general is a non-specific, non-sensitive diagnostic tool 20; parenchymal changes in echogenicity have many disparate differentials, while hepatic masses and nodules might be overlooked. In FHL, hepatomegaly and increased hepatic echogenicity (compared to the falciform fat), with resultant decreased visibility of intrahepatic portal vein walls, are commonly documented, but alternative pathologies (e.g., infiltrative diseases) might also account for such changes or coexist 20.
The initial diagnostic work-up of a cat with FHL will include a complete blood count, biochemistry panel, PT/aPTT measurement and abdominal ultrasound with FNA of the liver. These tests provide a robust minimum database which will allow the diagnosis of FHL, complications thereof, and many underlying or concomitant diseases. Otherwise, further diagnosis is often halted at this stage, and initial treatment is instituted. When warranted, additional tests, directly related to FHL, might include the measurement of blood ammonia and B12 (cobalamin) concentrations. The former is indicated in cats that are severely depressed or have neurological signs, while the latter is indicated in any cat with FHL, since B12 reserves are rapidly depleted in anorectic cats. If a concurrent bacterial biliary infection is suspected, gallbladder cystocentesis can be performed for cytological and bacteriological evaluation of bile fluid. This procedure is considered safe, with minimal risk of gallbladder perforation/leakage or vagally mediated hypotension 21. Lastly, if laboratory and clinical improvement are not seen with treatment over the course of several weeks, a liver biopsy might be considered to rule out an underlying hepatic pathology.
Ran Nivy
Malnutrition is a hallmark of FHL. Regardless of the instigating cause and whether it has resolved, once HL develops, nausea follows, and a vicious cycle of anorexia and hepatic lipid accumulation ensues. The cornerstones of treatment therefore include nutritional support and anti-nausea treatment. Secondary oxidative damage, inflammation and intrahepatic cholestasis are common sequelae of FHL, and additional medications are used to promote fat utilization, choleresis and antioxidant activity.
Nutritional management must address several factors, namely:
Cats with FHL rarely consume enough food during the initial stages of hospitalization, but force-feeding might result in food aversion and postpone resumption of voluntary feeding. Since recovery can take several weeks, and to facilitate the administration of medications, treatment of FHL often entails the use of a feeding tube. Nutritional support should be instituted as early as possible, but the presence of severe dehydration, electrolyte changes, acute kidney injury, hypotension or severe neurological deficits might delay the procedure. Partial parenteral nutrition (PPN) can be used in the first days of hospitalization until the cat is stable enough to undergo general anesthesia. Unlike total parenteral nutrition, PPN does not require central vein access, and has fewer complications. Amino-acid solutions may be used alone as a constant rate infusion if premixed PPN solutions are unavailable.
Once the cat is stable, a feeding tube can be placed under general anesthesia. Esophageal tubes are often the first-choice for several reasons: they are easy to insert and use; both liquid and blended food and medications can be administered; they are associated with a lower prevalence of significant complications (other than dislodgement or stoma site infection) and are easily managed by owners; feeding can be instituted shortly after recovery from anesthesia; and no complications arise with inadvertent, premature tube removal 22 (Box 1). Naso-esophageal and nasogastric tubes can be used until a longer general anesthesia is feasible, but are not suitable long-term: only liquids can be passed through them, and they entail significant discomfort to the cat. Lastly, a percutaneous endoscopically assisted feeding tube is rarely indicated, unless the esophagus must be bypassed owing to an esophageal pathology.

© Darren J. Berger
© Darren J. Berger
© Darren J. Berger
© Karen L. Campbell
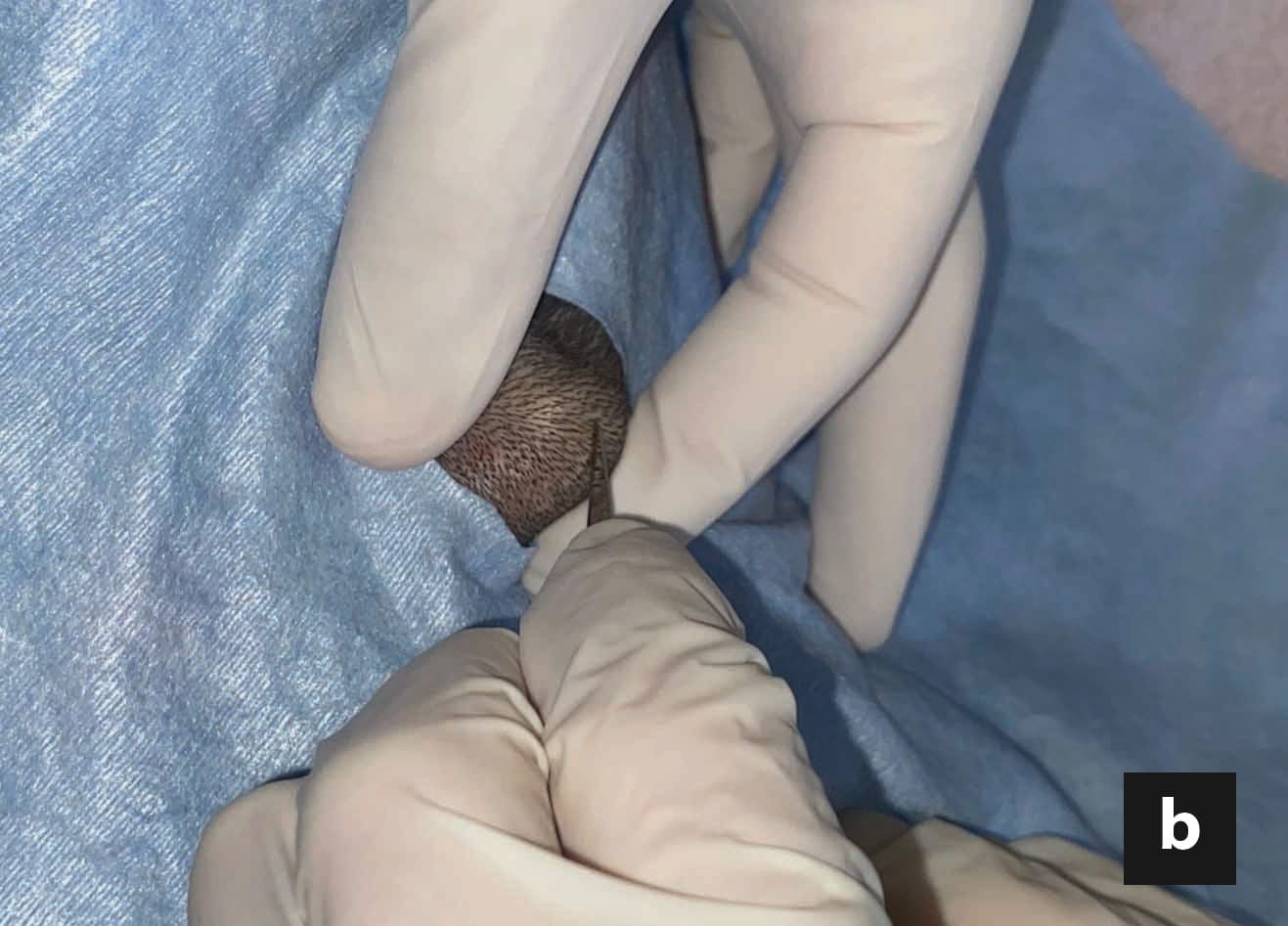
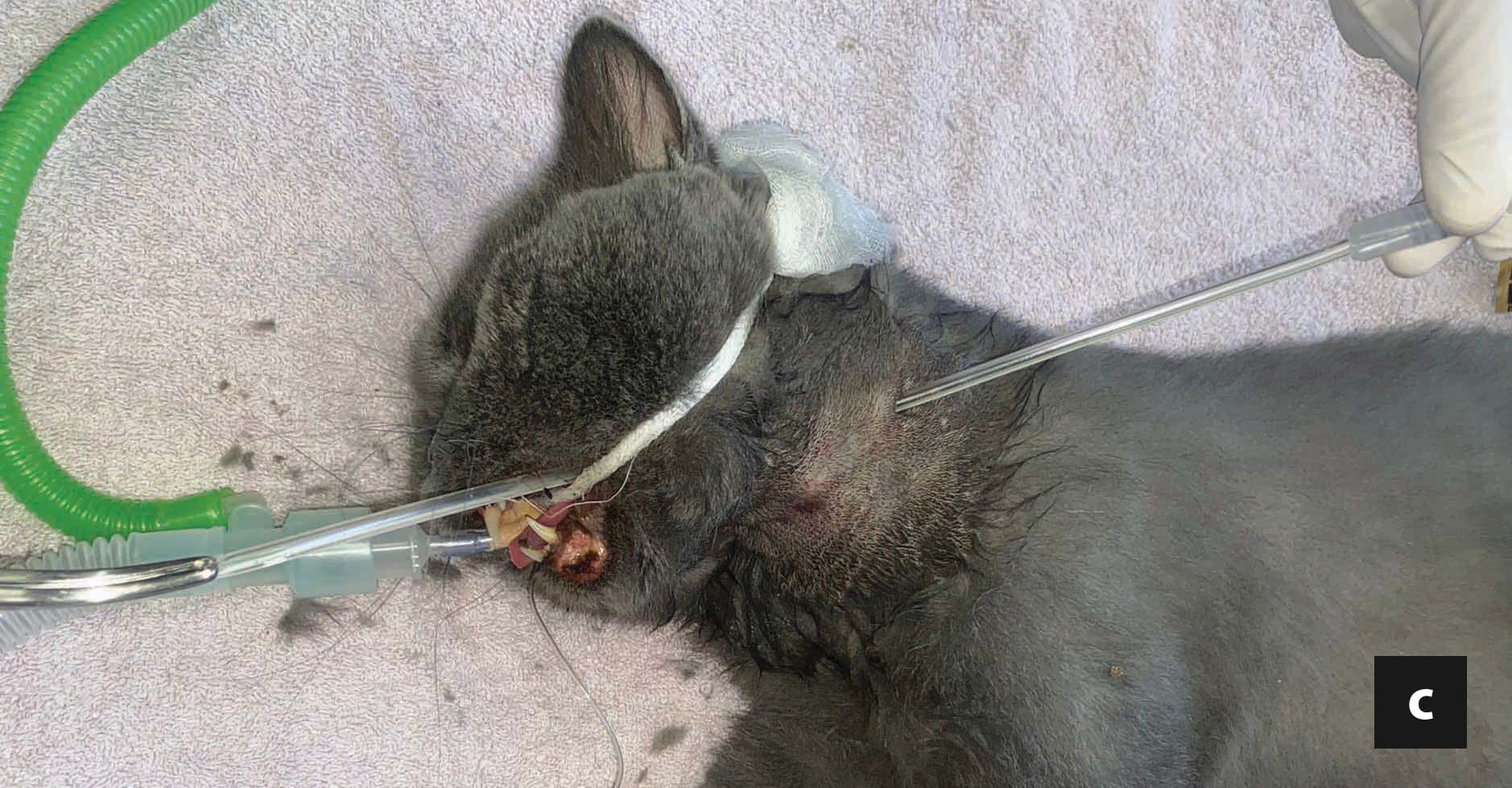
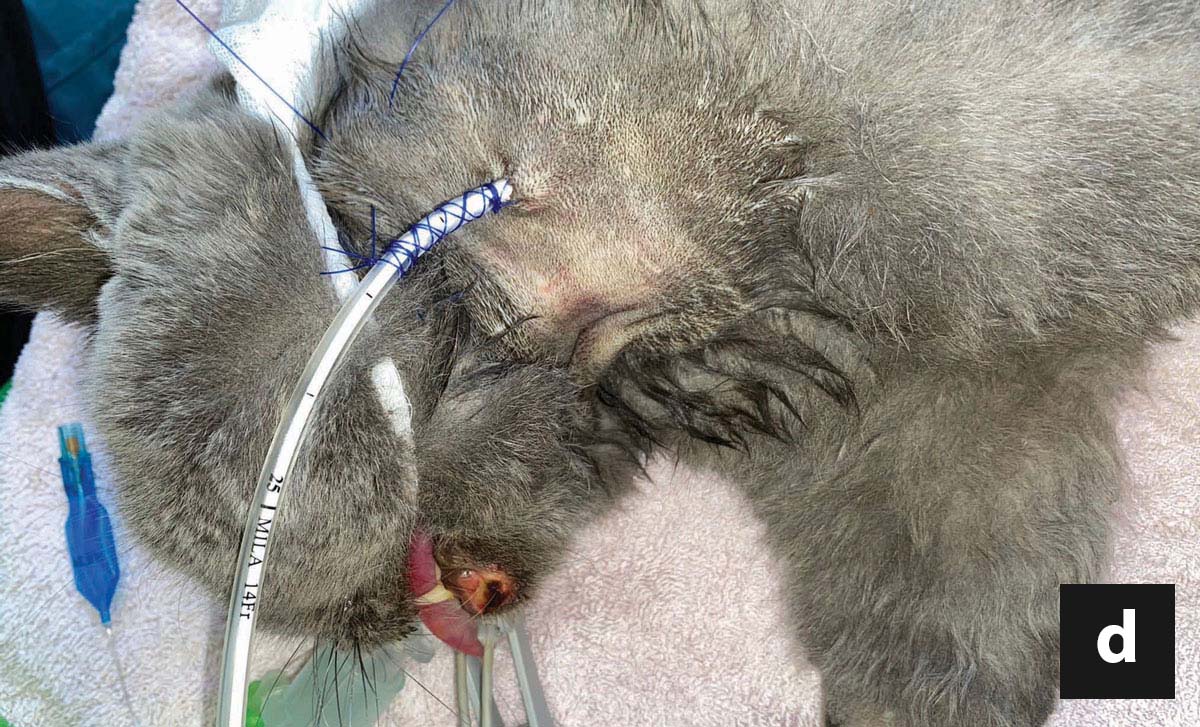
Box 1. Insertion of an esophageal tube in a 7-year-old, male cat with acute pancreatitis and secondary hepatic lipidosis. Cats with hepatic lipidosis are prone to anesthetic complications, and some drugs might be contraindicated owing to liver dysfunction, so an opioid will often be used for premedication, and etomidate, propofol or a low dose of ketamine for induction. Esophageal tubes are inserted only in intubated cats, using inhalation anesthesia, while fluid support (as well as colloids and/or vasopressor therapy, when indicated) is used to maintain blood pressure.
The cat is placed in right lateral recumbency, and the left cervical area clipped and scrubbed. The 14-G feeding tube is pre-measured from the mid-cervical point of entry to the 7th-9th intercostal space (a). However, any location caudal to the heart and cranial to the diaphragm might be tolerable to the cat. Curved, blunt-tipped forceps (e.g., Carmalt forceps) are inserted through the mouth, to the mid-cervical area, laterally and away from the adjacent jugular vein. A small cut is made over the bulge created by the tip of the forceps (b). The feeding tube is grabbed and retracted out of the mouth (c). The tip of the tube is then guided manually into the esophagus, until the outer part of the tube is flipped. After inserting the tube to its premeasured location, it is secured using a Chinese-finger trap technique (d). Using a purse string suture is discouraged, as it can interfere with proper drainage of secretions in case of stoma site infection. Location of the tube must always be ascertained by a thoracic radiograph at the end of the procedure.
Commercially available, calorie-dense, protein-rich recovery diets often suffice to provide the nutritional needs of the cat in terms of essential AA and fats. Protein restriction is rarely indicated unless severe HE is present. The calculated daily caloric intake (resting energy requirement: 30 x (body weight) +70 or (body weight)0.75 x70), is divided into 4-6 meals. The volume of each meal, including the water used at the end to flush the feeding tube, must not exceed 10-15 mL/kg/meal. A gradual upward titration of the food volume and the overall caloric intake is advised, usually over the course of 3 days. This is meant to ensure a gradual adaptation of the gastrointestinal tract to food, to monitor the development of gastric ileus which might preclude further feeding, and to reduce the risk of refeeding syndrome (see “complications” section below).
Both fat-soluble (e.g., vitamin D, vitamin K) and water-soluble (e.g., thiamine, cobalamin) vitamins are depleted in cats with FHL 2. Deficiencies are associated with a plethora of complications, including coagulopathies, villous atrophy, hyperammonemia, anemia and neurological deficits, rendering vitamin supplementation an integral part of treatment (Table 2).
Hydration status, maintenance needs, and sensible (e.g., vomiting/diarrhea) and insensible fluid losses are considered when calculating the daily fluid requirement. However, the potential for concurrent cardiac disease, and the amount of enterally administered water via the feeding tube, must also be considered. Potassium supplementation is often indicated, either as potassium chloride (KCl) or potassium phosphate (KPO4) when hypophosphatemia coexists (Table 2). If measurement of blood magnesium concentration is unavailable, a conservative approach might be to supplement the fluid with magnesium sulphate at 0.5 mEq/kg q24h for 2 days.
Antinausea and antiemetic therapy is indicated in all cats, with metoclopramide, maropitant and ondansetron being used in various combinations. Mirtazapine is often added owing to its antinausea and appetite-stimulating properties, especially to anorectic cats after hospital discharge. Metoclopramide has the added benefit of promoting gastrointestinal motility. If ileus develops despite metoclopramide treatment and resolution of hypokalemia, cisapride treatment (0.5 mg/kg q8h PO) should be considered.
Oxidative damage and decreased glutathione concentration are known complications of FHL, and antioxidant therapy is commonly prescribed to affected cats; these include SAMe and silymarin. SAMe is a ubiquitous molecule involved in a myriad of biochemical pathways 23,24, whilst silymarin (which contains several compounds, of which silibinin is the most abundant and active ingredient) exerts anti-inflammatory and choleretic activities, as well as antioxidant activity 23. Several veterinary products now provide a combination of silymarin and SAMe, but if oral or enteral supplementation is not feasible, n-acetylcysteine can be used intravenously to replenish hepatic cysteine concentration and subsequently glutathione levels. However, it does not possess the additional benefits of silymarin and SAMe, and can induce vomiting if administered rapidly 23. Lastly, vitamin E might be supplemented, as it is a free radical scavenger which protects against phospholipid oxidation in the cell membrane; it has minimal side effects, but there are no reported clinical benefits either 23 (Table 2).
Ursodeoxycholic acid, a hydrophilic bile acid, might prove beneficial in FHL owing to its choleretic, anti-apoptotic, anti-inflammatory properties, and rarity of side-effects 23 (Table 2). L-carnitine transports long-chain fatty acids into the mitochondria, thereby facilitating their utilization for energy production, and in obese cats undergoing food restriction it attenuates hepatic triglyceride accumulation, with clinical observations indicating a potential beneficial effect in FHL 10. Even though carnitine levels are increased in cats with FHL 25, a relative deficiency thereof might still exist, and supplementation is recommended (Table 2). Taurine deficiency is associated with various cardiac, neurological, reproductive and developmental pathologies, and contributes to hepatic fat accumulation. Commercial diets will provide adequate amounts of this amino acid, but enteral supplementation is occasionally advocated, especially when ursodeoxycholic acid is used, as it exacerbates biliary loss of taurine 1. Lastly, lactulose might be considered when severe signs of HE and hyperammonemia exist, or in constipated cats, while pantoprazole/omeprazole are used when gastric ulceration or reflux esophagitis are suspected.
A myriad of complications can arise secondary to FHL and following treatment. Refeeding syndrome, a constellation of metabolic changes which develop after reinstitution of feeding following protracted malnutrition, is a frequently mentioned but poorly documented complication in cats. It is associated with severe hypophosphatemia, and occasionally hypokalemia, hypomagnesemia, and thiamine deficiency. A gradual increase in caloric intake, with carbohydrate restriction, pre-emptive electrolyte supplementation and close monitoring will help avoid the deleterious consequences of this syndrome 1,14,15.
Diffuse villous atrophy is another complication which develops with prolonged anorexia, resulting in malabsorption and diarrhea following reinstitution of enteral feeding. Gradual introduction of enteral feeding, and the use of highly digestible diets, can alleviate diarrhea. Concurrent pathologies are common in cats with FHL, and include pancreatitis, kidney injury, cardiomyopathies, bleeding/thrombotic complications, and insulin resistance. Additionally, cavitary effusions might develop secondary to hypoalbuminemia, cardiac disease, or pancreatitis, and these constitute a negative prognostic factor 9.
Finally, immunosuppression from malnutrition can predispose to secondary bacterial infections, and antibiotic treatment is indicated if an infection is documented (e.g., cholecystitis). Under specific circumstances antibacterial therapy might also be considered when severe neutropenia/neutrophilia, and/or hypoglycemia occur, even if there is no cytological or bacteriological proof of infection.
Feline hepatic lipidosis is the most common liver disease in cats, and can have a highly variable outcome because of the prognostic implications of different underlying diseases and comorbidities. Whilst overall survival rates range from 50-85%, acute pancreatitis, ptyalism, cavitary effusions, hypoalbuminemia, and an older age at onset are but a few of the reported negative prognostic markers. Nevertheless, whilst the condition is associated with significant morbidity, requires intensive treatment, and entails considerable financial expenditure, treatment is often successful, and recurrence seems rare.
Valtolina C, Favier RP. Feline Hepatic Lipidosis. Vet. Clin. North Am. Small Anim. Pract. 2017;47(3):683-702.
Verbrugghe A, Bakovic M. Peculiarities of one-carbon metabolism in the strict carnivorous cat and the role in feline hepatic lipidosis. Nutrients 2013;5(7):2811-2835.
Hall JA, Barstad LA, Connor WE. Lipid composition of hepatic and adipose tissues from normal cats and from cats with idiopathic hepatic lipidosis. J. Vet. Intern. Med. 1997;11(4):238-242.
Pazak HE, Bartges JW, Cornelius LC, et al. Characterization of serum lipoprotein profiles of healthy, adult cats and idiopathic feline hepatic lipidosis patients. J. Nutr. 1998;128(12 Suppl):2747S-2750S.
Blanchard G, Paragon BM, Sérougne C, et al. Plasma lipids, lipoprotein composition and profile during induction and treatment of hepatic lipidosis in cats and the metabolic effect of one daily meal in healthy cats. J. Anim. Physiol. Anim. Nutr. (Berl.). 2004;88(3-4):73-87.
Webb CB. Hepatic lipidosis: Clinical review drawn from collective effort. J. Feline Med. Surg. 2018;20(3):217-227.
Center SA, Crawford MA, Guida L, et al. A retrospective study of 77 cats with severe hepatic lipidosis: 1975-1990. J. Vet. Intern. Med. 1993;7(6):349-359.
Biourge VC, Groff JM, Munn RJ, et al. Experimental induction of hepatic lipidosis in cats. Am. J. Vet. Res. 1994;55(9):1291-1302.
Kuzi S, Segev G, Kedar S, et al. Prognostic markers in feline hepatic lipidosis: a retrospective study of 71 cats. Vet. Rec. 2017;181(19):512.
Center SA, Harte J, Watrous D, et al. The clinical and metabolic effects of rapid weight loss in obese pet cats and the influence of supplemental oral L-carnitine. J. Vet. Intern. Med. 2000;14(6):598-608.
Center SA, Baldwin BH, Dillingham S, et al. Diagnostic value of serum gamma-glutamyl transferase and alkaline phosphatase activities in hepatobiliary disease in the cat. J. Am. Vet. Med. Assoc. 1986;188(5):507-510.
Brown B, Mauldin GE, Armstrong J, et al. Metabolic and hormonal alterations in cats with hepatic lipidosis. J. Vet. Intern. Med. 2000;14(1):20-26.
Nivy R, Kaplanov A, Kuzi S, et al. A retrospective study of 157 hospitalized cats with pancreatitis in a tertiary care center: Clinical, imaging and laboratory findings, potential prognostic markers and outcome. J. Vet. Intern. Med. 2018;32(6):1874-1885.
Brenner K, KuKanich KS, Smee NM. Refeeding syndrome in a cat with hepatic lipidosis. J. Feline Med. Surg. 2011;13(8):614-617.
Boateng AA, Sriram K, Meguid MM, et al. Refeeding syndrome: treatment considerations based on collective analysis of literature case reports. Nutrition 2010;26(2):156-167.
Center SA, Warner K, Corbett J, et al. Proteins invoked by vitamin K absence and clotting times in clinically ill cats. J. Vet. Intern. Med. 2000;14(3):292-297.
Griffin S. Feline abdominal ultrasonography: what’s normal? what’s abnormal? The liver. J. Feline Med. Surg. 2019;21(1):12-24.
Willard MD, Weeks BR, Johnson M. Fine-needle aspirate cytology suggesting hepatic lipidosis in four cats with infiltrative hepatic disease. J. Feline Med. Surg. 1999;1(4):215-220.
Wang KY, Panciera DL, Al-Rukibat RK, et al. Accuracy of ultrasound-guided fine-needle aspiration of the liver and cytologic findings in dogs and cats: 97 cases (1990-2000). J. Am. Vet. Med. Assoc. 2004;224(1):75-78.
Feeney DA, Anderson KL, Ziegler LE, et al. Statistical relevance of ultrasonographic criteria in the assessment of diffuse liver disease in dogs and cats. Am. J. Vet. Res. 2008;69(2):212-221.
Byfield VL, Callahan Clark JE, Turek BJ, et al. Percutaneous cholecystocentesis in cats with suspected hepatobiliary disease. J. Feline Med. Surg. 2017;19(12):1254-1260.
Breheny CR, Boag A, Le Gal A, et al. Esophageal feeding tube placement and the associated complications in 248 cats. J. Vet. Intern. Med. 2019;33(3):1306-1314.
Webster CR, Cooper J. Therapeutic use of cytoprotective agents in canine and feline hepatobiliary disease. Vet. Clin. North Am. Small Anim. Pract. 2009;39(3):631-652.
Center SA, Warner KL, Erb HN. Liver glutathione concentrations in dogs and cats with naturally occurring liver disease. Am. J. Vet. Res. 2002;63(8):1187-1197.
Jacobs G, Cornelius L, Keene B, et al. Comparison of plasma, liver, and skeletal muscle carnitine concentrations in cats with idiopathic hepatic lipidosis and in healthy cats. Am. J. Vet. Res. 1990;51(9):1349-1351.
Ran Nivy
After completing a bachelor’s degree in Life Sciences in 2005, Dr. Nivy continued to veterinary school and graduated in 2009 Read more
Insect contamination of petfoods is a potential problem in tropical countries; this paper offers an overview of the situation and how the risks can be minimized
Mistakes happen to all of us in veterinary practice; this article looks at how different people react in different ways when things go wrong and – importantly – discusses how we can best cope with mistakes.
Cats are living longer and better lives; how can we ensure that the healthcare we offer them is optimal? This article offers some hints.
OA in cats remains underdiagnosed and undertreated, despite its widespread prevalence; this paper looks at how we can surmount the challenge of early diagnosis, leading to better treatment interventions.
