Petfood: how to prevent insect infestation
Insect contamination of petfoods is a potential problem in tropical countries; this paper offers an overview of the situation and how the risks can be minimized
Issue number 33.2 Other Scientific
Published 13/09/2023
Also available in Français , Deutsch , Italiano , Português , Español and ภาษาไทย
Vomiting, alone or in combination with other clinical signs, is a common presenting complaint in cats; in this paper the authors share their recommendations for a systematic clinical approach for both acute and chronic vomiting cases.

Signalment, a detailed history, and clinical findings must all be considered before formulating likely differential diagnoses and a diagnostic plan for the vomiting cat.
The acute vomiting cat should firstly be triaged to determine if immediate stabilization measures are required before any diagnostics are performed.
Therapeutic or dietary trials can be considered before diagnostic investigations in clinically stable cases, but any inadequate nutrition should be promptly recognized and addressed.
Differentiation of inflammation from neoplasia is particularly important in the feline gut, as inflammatory bowel disease and lymphoma have a differing prognosis and treatments.
Both acute and chronic vomiting can be the manifestations of primary gastrointestinal (GI) disease or extra-GI conditions in the cat. The list of potential differential diagnoses for vomiting is extensive, and other criteria from both the history and physical examination, followed by appropriate laboratory and diagnostic imaging tests, can often help to narrow those down. For acute vomiting, the two most important decisions to be made following initial triage are to decide if relatively swift supportive care (particularly replacement of fluid losses and correction of electrolyte or acid-base imbalances) is needed, and if surgical intervention may be necessary (which could be diagnostic, therapeutic or both) when stabilization is achieved. Once the patient is cardiovascularly stable and there is no immediate indication for surgery, a more complete list of differential diagnoses for acute vomiting can be considered (Table 1).
For chronic vomiting, consideration of possible causes and diagnostic tests can usually be at a slower pace, and can depend on any additional presenting complaints, physical examination findings, and (not uncommonly) an owner’s preferences. It seems reasonable to assess more common conditions first and to proceed from there in a stepwise manner. For a cat with chronic GI signs and no evidence of extra-GI or infectious disease, the two most common remaining differential diagnoses are idiopathic Inflammatory Bowel Disease (IBD) and Low-Grade Alimentary Lymphoma (LGAL), and more invasive tests (e.g., biopsies) are commonly performed relatively early in the diagnostic workup compared to dogs. This article will describe a stepwise diagnostic approach for cats presented with either acute or chronic vomiting, and discuss management of the more common underlying conditions.
Table 1. Differential diagnoses for acute vomiting in cats.
Extra-gastrointestinal diseases
Gastrointestinal diseases
|
AKI = acute kidney injury, GIST = gastrointestinal stromal tumor, NSAIDs = non-steroidal anti-inflammatory drugs, UTI = urinary tract infection
A complete clinical history is extremely useful to refine the list of differential diagnoses. It is particularly important to establish that the owner does not describe regurgitation, retching or even coughing instead of vomiting, as these can be difficult to distinguish from one another in cats. Once it is established to be vomiting, the respective signalment and the presence or absence of additional clinical signs can help prioritizing some conditions over others. Therefore, the clinician should consider:
Cats with dietary indiscretions, food hypersensitivities or food-responsive chronic enteropathies (FRE) tend to be younger than those with other conditions, especially other forms of chronic enteropathies (CE) 1. Conversely, hyperthyroidism is a common cause of chronic vomiting, as is neoplasia, with both being more likely in older cats. Siamese cats are at an increased risk of GI adenocarcinoma 2, whilst longhair breeds are more likely to have trichobezoars, which can lead to partial or complete GI obstruction.
Any recent food changes increase the likelihood of intolerances. Hunting behavior raises the possibility of an infectious disease (especially in conjunction with diarrhea and/or pyrexia), and the clinician should ask if the cat is free-roaming, with possible access to toxins.
The differential diagnoses, diagnostic approach and therefore management of acute (< 1 week) vs. chronic (> 3 weeks) are considerably different. Intermittent vomiting can be “physiological” in some cases, but some cats with IBD or food-responsive enteropathy (FRE) can have mild and intermittent GI signs for months to years without significant clinical deterioration 1. Progressive, or more frequent chronic intermittent vomiting might warrant more prompt investigations, especially in adult/older cats.
The presence or absence of diarrhea can help narrow differential diagnoses down to potential infectious or obstructive disease (especially in acute vomiting), or to diseases of the GI tract and adjacent organs (pancreas, liver) when chronic, especially in the absence of other signs. Some cats with CE or IBD will not present with diarrhea, but only with vomiting +/- weight loss 1. Concurrent polyuria/polydipsia (PUPD) in chronic vomiting might raise a suspicion of chronic kidney disease (CKD) or diabetes mellitus. The presence of jaundice suggests hepatobiliary disease or pancreatitis (although pre-hepatic causes (i.e., hemolysis) have to be ruled out). Weight loss and anorexia are non-specific signs, but when chronic (especially in the absence of other specific signs or abnormalities) they can be an indicator of primary GI disease, including lymphoma.
Ivan Montanes-Sancho
During physical examination special attention should be paid to the following aspects:
Triage should be targeted to identify weak pulses, dehydration (which can be difficult to gauge in cats with moderate to severe weight loss), hypo- or hyper-thermia, prolonged capillary refill time, or marked lethargy. In contrast to dogs, cats with shock do not generally compensate and can present with either tachycardia, a normal heart rate or even bradycardia (which would be a major concern in an ill animal). Such indicators of severe disease warrant supportive treatment before a more thorough evaluation.
Cats that are otherwise well, still eating and have a normal hydration status should be considered as having mild disease. Abdominal palpation in cats with vomiting is mandatory and can be very rewarding, as it is usually easy to evaluate intra-abdominal structures such as liver, kidneys, bladder, and intestines. Intestinal wall thickening, masses (intestinal, lymph nodes or others), intussusceptions and sometimes even intestinal foreign bodies (FB) can be palpated, unless the patient is obese or difficult to handle. However, normal palpation does not exclude abdominal pathology, especially chronic conditions like pancreatitis, IBD or a FB. The most common type of FB in cats are linear ones such as sewing thread 3, which cannot usually be palpated directly. As one end of the thread can remain attached to the base of the tongue 3, careful examination of the oral cavity should be performed where there is acute onset of vomiting, particularly if dysphagia or hypersalivation are present concurrently.
In the authors’ experience it can be challenging to assess cats for abdominal pain or discomfort, as it seems to be an infrequent finding with feline intra-abdominal conditions. For example, only 10-30% of cats with acute pancreatitis have been reported to show apparent abdominal pain 4, and neither is it commonly reported with chronic small bowel disease, lymphoma 1, or even intestinal foreign bodies.
Finding jaundice in a feline patient with vomiting is significant; not only because it can indicate primary hepatobiliary disease, but because it could suggest hepatic lipidosis and the need for nutritional support.
Finally, any cat with vomiting could be at risk of developing aspiration pneumonia, thus the respiratory system should be carefully assessed by auscultation and/or thoracic imaging (either via radiography or point of care ultrasound [POCUS]) as indicated.
A minimalistic approach is often sufficient for cats with acute vomiting and unremarkable clinical examination (i.e., mild disease). The term “non-specific gastroenteritis” refers to self-limiting vomiting ± diarrhea of unknown origin, often presumed to be secondary to infection or dietary indiscretion. Here symptomatic treatment is often sufficient, but cats failing to respond to this need a more thorough evaluation.
The absence of abnormalities on abdominal palpation does not fully exclude the presence of a pathology; therefore, abdominal radiography can be considered – even with mild disease – in case surgical treatment is required. If a FB is not directly visible (e.g., not radiopaque), the clinician should be mindful of typical imaging findings that are indirect indicators of obstruction, such as central “bunching” of the intestines (Figure 1), or abnormal “crescent” or “teardrop” shaped intestinal gas bubbles 5. The presence of a so-called “gravel sign” is indicative of chronic intestinal obstruction (Figure 2). While other types of intestinal FBs tend to produce complete obstruction, with dilation of small bowel loops cranial to the foreign material, it does not seem to be the case for linear foreign bodies 5. Likewise, some cats with chronic GI disease (especially young patients) may present with chronic/dynamic intussusception with no obvious dilation of intestinal loops. It is very important to take two, if not three abdominal radiographic views (left and right lateral and an orthogonal image), as key abnormalities may be evident only in one view (Figure 3).

Figure 1. Right lateral abdominal radiograph of an adult cat presented with acute onset of vomiting due to the presence of an intestinal foreign body. Bunching of small intestinal loops (arrow) is observed with associated stomach distension (asterisk), although the foreign body cannot be visualized directly.
© University of Edinburgh

Figure 2. Right lateral abdominal radiograph of an adult cat with a history of chronic vomiting and intestinal granulomas leading to partial intestinal obstruction. There is marked distension of small intestinal loops (asterisks) with a gravel sign (presence of small mineral opacities) ventrally (arrow).
© University of Edinburgh
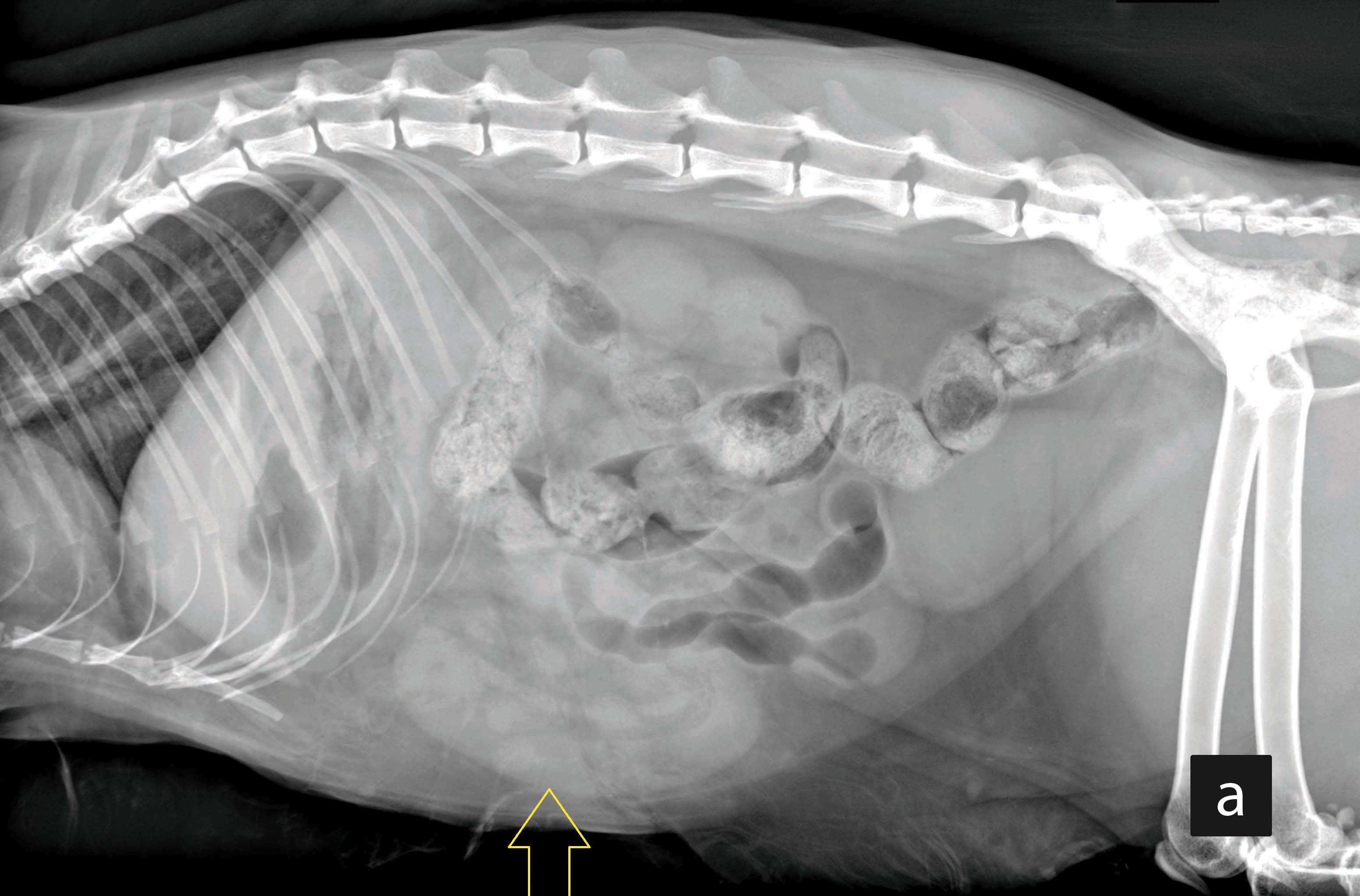
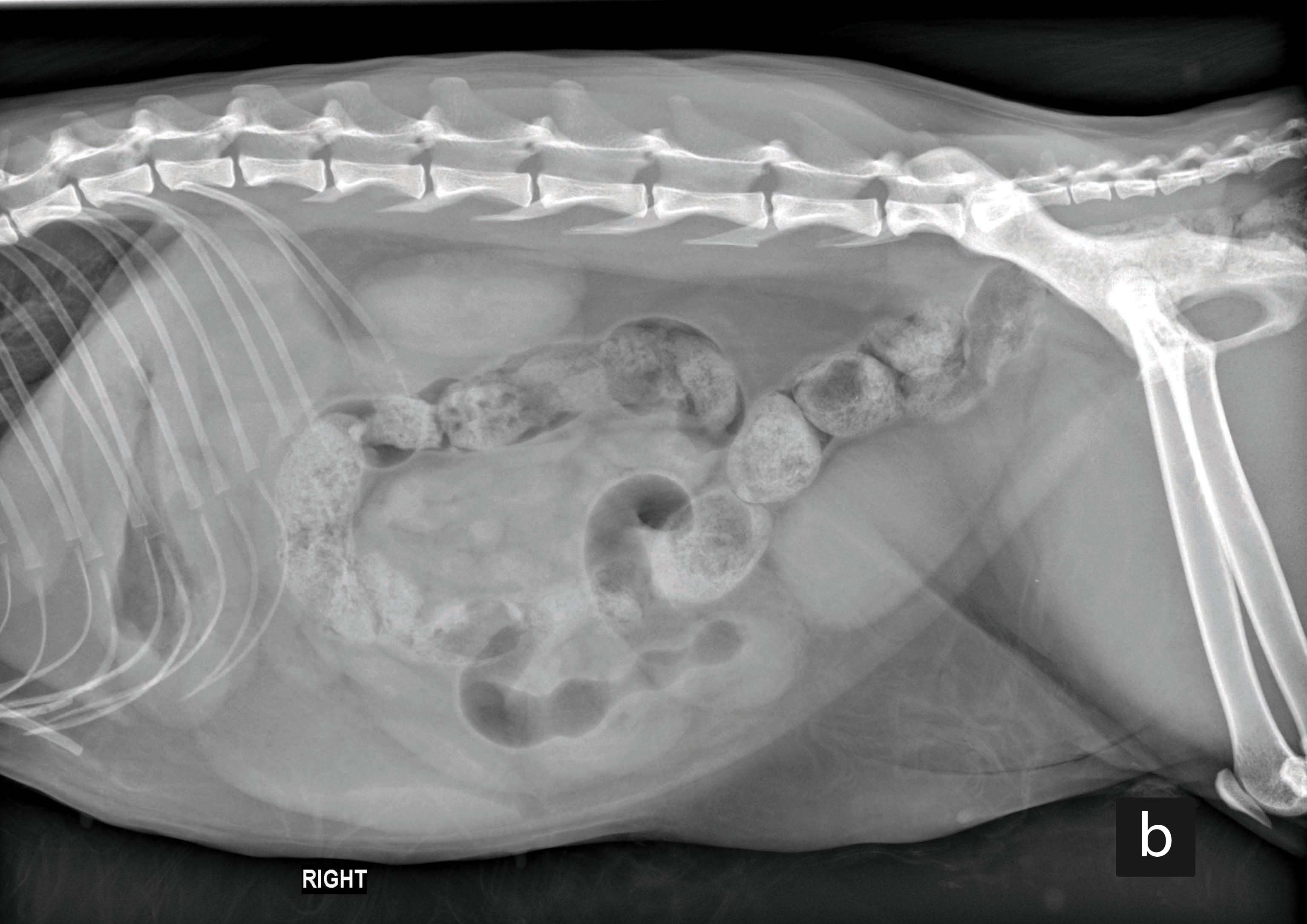
Figure 3. Left (a) and right (b) lateral abdominal radiographs from an adult cat presented with acute vomiting and diagnosed with intestinal intussusception. The intussusception is only visible in the left lateral projection as an intestinal loop with a “sausage-like” homogenous soft-tissue appearance (arrow).
© University of Edinburgh
Abdominal ultrasound can be a useful complementary tool in investigating causes of vomiting. For example, evidence of intestinal plication raises suspicion for a linear FB (Figure 4), although this can be misinterpreted as corrugation (Figure 5) which is a non-specific finding reported in cats with enteritis, pancreatitis, peritonitis or neoplasia 5. Architectural changes such as loss of layering or GI thickening can also be assessed. Overall, loss of layering raise suspicion for neoplasia, but severe inflammation can also lead to this. While ulcers are usually not detected by ultrasonography, a circumscribed thickening of the GI wall with an echogenic center (trapped gas) can be seen in some cases.

Figure 4. Ultrasonographic image of a small intestinal loop from an adult cat showing marked small intestinal plication (arrows). The cat was diagnosed with a linear foreign body.
© University of Edinburgh
Exploratory laparotomy can be considered if there is a high suspicion of obstruction (e.g., marked segmental intestinal dilatation on radiographs or ultrasound) even if a specific etiology (intussusception/FB) is not identified. The presence of peritoneal gas on an abdominal radiograph (suggestive of GI perforation) is another indication for emergency exploratory surgery.
For cats that are systemically unwell, or clinically stable cats who have failed symptomatic treatment, establishing a laboratory minimum data base (MDB) or a comprehensive complete blood count (CBC) and serum biochemistry (SB) is indicated. The MDB should include packed cell volume (PCV) and total solids (TS), electrolytes (with or without venous blood gases if available), and basic biochemistry parameters (glucose, creatinine, blood urea nitrogen (BUN), liver enzymes, albumin and total bilirubin).
Findings such as sudden onset azotemia that are consistent with acute kidney injury (AKI) warrant investigations to identify specific treatable conditions, e.g., urinary tract infection (UTI)/pyelonephritis (by urine culture and sensitivity (C&S) testing before antibiotic administration) or urolithiasis. However, in a large proportion of cats the cause of AKI is never identified.
Antemortem diagnosis of acute pancreatitis in cats is challenging and abdominal ultrasound seems to be insensitive to confirm the diagnosis, even in the presence of clinical signs 4,6. Specific feline pancreatic lipase immunoreactivity (fPLI) may aid in the diagnosis, and a combination of both techniques increases both sensitivity and specificity 4. Similarly, some cats with cholecystitis or cholangitis do not have abnormalities of the hepatobiliary system on ultrasound 7, while others show changes of the gall bladder wall or content, or extrahepatic bile duct obstruction (EHBDO). Aspiration of bile (for cytology and C&S) is strongly recommended when cholecystitis or cholangitis is suspected before antibiotics are administered, but this can risk gall bladder rupture in cases of severe edema or inflammation of the wall. Generally, the rate of complications associated with cholecystocentesis is low when performed under adequate sedation and appropriate technique; the gallbladder must be emptied as much as possible to minimize the risk of leakage. However, if deemed too risky, fine-needle aspiration (FNA) from the hepatic parenchyma just adjacent to the gall bladder (again for both cytology and C&S) is an appropriate alternative.

Figure 5. Ultrasonographic image of a small intestinal segment from an adult cat presented with acute onset vomiting and diarrhea diagnosed with pancreatitis. The picture shows intestinal corrugation (arrows indicate the intestinal lining).
© University of Edinburgh
In cats with chronic vomiting ± diarrhea but are otherwise systemically well, an elimination dietary trial and minimal diagnostic tests (for example fecal parasitology) should be considered before a more comprehensive workup. Conversely, cats with additional significant weight loss or that are systemically unwell need a more thorough evaluation (see below).
As for acute vomiting, differential diagnoses for chronic vomiting include various GI and extra-GI conditions (Table 2). Comprehensive laboratory work (CBC, SB, total thyroxin, urinalysis, fecal parasitology and other fecal pathogen testing [e.g., via PCR]) are considered initial relatively non-invasive diagnostic steps that can help exclude common extra-GI disorders. In most cases it is advisable to take extra serum samples that can be used later for additional laboratory examinations if required, based on the initial findings. For example, these can include tests for pancreatic and/or intestinal function (fPLI, fTLI, serum cobalamin), infectious diseases (e.g., toxoplasma titers, feline coronavirus (FCoV) titers), and hepatic function tests (baseline bile acids). The diagnosis of chronic feline pancreatitis can be especially challenging, because clinical signs are usually non-specific, and fPLI may be normal or borderline elevated. Depending on other clinical findings, the clinician may also consider further laboratory investigations (e.g., citrated blood to check clotting times in cases of hepatobiliary disease, or ionized calcium where neoplasia might be suspected).
Table 2. Differential diagnoses for chronic vomiting in cats.
Extra-gastrointestinal diseases
Gastrointestinal diseases
|
EHBDO = extrahepatic bile duct obstruction, FCoV = feline Coronavirus, FIP = feline infectious peritonitis, GI = gastrointestinal, FGESF = feline gastrointestinal eosinophilic sclerosing fibroplasia, LGAL = low-grade alimentary lymphoma, MCT = mast cell tumor.
Ultrasonography is the preferred diagnostic imaging modality, particularly in skilled hands, as it provides useful detail on the size and structure of intra-abdominal organs. Initial survey abdominal radiographs can be considered, but they are insensitive for the diagnosis of pancreatitis, cholecystitis/cholangitis, and assessment of changes in the GI wall consistent with inflammation/IBD (or triaditis, the three conditions combined) or diffuse GI neoplasia. Abdominal masses can only be identified on radiographs beyond a certain size, and their origin is rarely identified. Only ultrasound can provide a finer assessment of changes within the GI wall (i.e., thickness and structure, where the loss of layering raises suspicion for neoplasia), as well as assessment of abdominal lymph nodes (for size and echogenicity). Increased echogenicity of specific gut layers (e.g., mucosa) can be a sign of inflammatory or neoplastic changes, or (rarely) lymphangiectasia. Thickening of the muscularis layer is often observed in IBD, but can also be found in healthy cats. Skilled operators can reliably assess the pancreas with ultrasound, but sensitivity for chronic pancreatitis is poor and it may appear completely normal 4. The origin and inner architecture of any mass can also be characterized, but remember that in some cats with diffuse primary GI conditions such as FRE, IBD or even LGAL, imaging findings can be completely normal – so a “normal” ultrasound does not rule out primary GI disease.
Minimally invasive sampling of abnormal structures (e.g., FNA) should be considered, often in conjunction with an ultrasound scan, and hence ideally discussed with owners beforehand. The main indication for FNA is differentiation between inflammatory and neoplastic conditions, and whilst not always diagnostic, they are easy to perform, do not require specialist equipment, can be done under sedation, and are associated with extremely low morbidity. As for cases with acute hepatobiliary disease, cholecystocentesis should also be considered in suspected chronic hepatobiliary conditions, where changes on ultrasound might be more subtle. If cytology findings are not diagnostic, they can be repeated (apart from bile samples), or a biopsy of the relevant organ taken, e.g., a tru-cut biopsy for the liver, or laparoscopic, surgical or GI mucosal pinch biopsies.
Especially where there is a single abdominal mass (+/- enlarged lymph nodes), ultrasound and biopsy can help differentiate neoplasia from other possible diagnoses; these include granulomas of fungal or feline infectious peritonitis (FIP) origin, mycobacterial disease, or (if located in the gut) feline gastrointestinal eosinophilic sclerosing fibroplasia (FGESF) 8. FNAs are also useful in the characterization of neoplasia within or outside the GI tract, although some tumors (lymphoma, adenocarcinoma, mast cell tumors) exfoliate better than others (gastrointestinal stromal tumors (GIST), leiomyoma or leiomyosarcoma). For some of the more common cancers, FNAs can also be a tool for full staging (assessing metastasis to liver, spleen, lymph nodes, or other organs).
Finally, computed tomography (CT) is very rarely needed to aid in the diagnosis of primary abdominal disease leading to (chronic) vomiting. It can however be helpful for mesenteric torsions (extremely rare in cats), vascular abnormalities (portosystemic shunts), or assessing large abdominal masses before surgical removal (for invasion of surrounding structures including vessels, thrombus formation etc.).
The two main differential diagnosis for cats with chronic vomiting, ± diarrhea, ± weight loss but otherwise no specific findings on diagnostic investigation are IBD and LGAL 1,9, which can unfortunately look identical. Hence biopsies are often the only way to differentiate between these conditions with any level of confidence; these can be obtained either during endoscopy as mucosal pinch biopsies, or as surgical full-thickness biopsies. Both methods have advantages and disadvantages (Table 3), but diagnostic accuracy may also be dependent on sampling technique. For example, it has been suggested that full-thickness biopsies from the duodenum are more accurate than endoscopic biopsies in differentiating IBD from LGAL 10.
Table 3. Advantages and disadvantages of endoscopic and surgical gastrointestinal biopsies.
| Endoscopic mucosal biopsies | Full-thickness surgical biopsies | ||
|---|---|---|---|
| Advantages | Disadvantages | Advantages | Disadvantages |
|
|
|
|
When performing endoscopic mucosal pinch biopsies, current guidelines recommend a minimum of six samples from each section of the feline GI tract 11, although most clinicians collect at least 8-15 biopsies from each segment as some are expected to be of suboptimal quality. The submission and processing of samples are also important 12, which can be subject to the methods of a particular laboratory/pathologist. A recent study showed that mounted and orientated GI specimens were superior to biopsies floating free in formalin 12.
Decisions about the biopsy method should therefore be made for each case depending on the index of suspicion for a specific condition or combination of diseases. For example, if hepatobiliary and/or pancreatic disease along with a chronic enteropathy is suspected, it might be both medically and practically favorable to take biopsies from all three organs surgically, rather than endoscopic gut biopsies only. Ultimately, cost, invasiveness, potential risks and owner preferences also have to play a role.
While histopathology remains the gold standard to differentiate IBD from LGAL, it has its limitations, particularly in the context of sensitivity and specificity 1,9. This is likely due to the fact that – as opposed to most dogs with lymphoma – LGAL is a continuation from long-standing IBD in cats, hence there is variation from inflammation to neoplasia on a sliding scale, sometimes making diagnosis difficult. In addition, despite the availability of histopathological templates 11, there seems to be difficulty in differentiating both IBD and LGAL from healthy tissue. In a recent blinded study, 12/20 duodenal biopsies from supposedly healthy cats were classified as LGAL, but only 3 cats developed GI signs after a median follow-up time of 709 days 13. If clinical observations and histopathological diagnosis do not seem to match, the authors strongly recommend seeking a dialogue with the pathologist involved to discuss what more can be done to further the diagnosis. This can include advanced immunohistochemistry or clonality testing, but even these have limitations 9,14; for example, one study found 40% of cats with IBD to feature monoclonality in their GI biopsies 14.
Establishing a diagnosis of IBD or LGAL therefore remains a challenge, as clinical signs, laboratory results, imaging findings, histology, immunohistochemistry and clonality features may overlap between these conditions 1,9,14. There are other types of alimentary lymphoma, including intermediate/high-grade, large granular or epitheliotropic lymphomas, that more frequently present as focal intestinal mass lesion(s) characterized by a B or T cell immunophenotype 15,16. These can usually be diagnosed with less invasive tests such as assessment of cytology or flow cytometry performed from FNAs 15,16.
Silke Salavati
Clinically stable cats with acute vomiting usually have self-limiting gastroenteritis, especially if a foreign body has been ruled out. Here treatment can be limited to feeding a commercial “gastrointestinal” diet for few days, probiotics (e.g., Enterococcus faecium) and anti-emetics depending on the severity of the vomiting. Various anti-emetics are available (some off-license for cats) but the most frequently used is maropitant (a neurokinin-1 receptor antagonist with both central and peripheral action). It has been associated with bone marrow hypoplasia in kittens and should not be used in cats < 16 weeks of age. Metoclopramide is less effective as an anti-emetic in cats compared to dogs, especially with regards to central effects, because it is a dopamine (D2) receptor antagonist, yet α2-adrenergic receptors are much more important in controlling vomiting in the feline vomiting center. Ondansetron is also very effective (both centrally and peripherally) but is quite expensive and is unlicensed, so should be used as a last resort and only in very sick animals where other anti-emetics have failed. Phenothiazines (α2-agonists) such as chlorpromazine or prochlorperazine can also be very effective anti-emetics in cats, and are often more cost-effective.
Lack of adequate nutrition should always be a concern in vomiting cats. This is often due to the duration of inappetence or anorexia, as well as their propensity to develop hepatic lipidosis and associated problems, and additional supportive treatment should be considered. This can be in the form of increased medical treatment in mild cases, e.g., appetite stimulants as well as anti-emetics. Available options (not always licensed for cats) include oral or transdermal mirtazapine, capromorelin or cyproheptadine. In more severe or prolonged cases, or when appetite stimulation with drugs is unsuccessful, nutritional support should be provided via a feeding tube once the vomiting is adequately addressed. This can either be a naso-esophageal tube (for short-term usage and very liquid food) or an esophagostomy tube (O-tube), when tube-feeding is likely to carry on for more than a few days. An O-tube can also easily be placed during a pre-planned procedure (e.g., endoscopy, surgical biopsies).
Extra-GI disorders need to be treated according to the underlying etiology, and it is beyond the scope of this article to provide specific details about treatment for these disorders.
In cats with suspected food-responsive CE or IBD, an elimination diet trial should be performed using a commercial hydrolyzed diet or a novel protein diet. In contrast to dogs, cats with FRE usually respond very rapidly (2-3 weeks) to a food change, although it can take up to 6-8 weeks to see a full response. A short course of either anti-emetics or appetite stimulants can be considered to improve compliance with a new diet. In cats with partial response to diet change, a second dietary trial with a different type of appropriate food can be attempted.
Additional sequential treatments for IBD if elimination diet(s) are not successful include administration of probiotics or glucocorticoids. Ideally, GI biopsies should be obtained before administration of the latter if a diagnosis is desired (especially if lymphomas other than LGAL have not been excluded, as they may require alternative treatment). However, not only are feline IBD and LGAL treated similarly with glucocorticoids, they also have nearly the same prognosis and outcome – hence even if a final differentiation has not been made based on biopsy, it is often a pragmatic step to treat these cases the same. If glucocorticoids and diet alone do not improve the situation, a sensible additional immunosuppressive drug is chlorambucil; this will not only be beneficial in severe IBD, but is also standard treatment for LGAL. Cyclosporine or other immunosuppressants are not used as often for (suspected) IBD in cats compared to dogs. However, chlorambucil is not effective for the treatment of alimentary intermediate to high grade or large granular lymphomas, which is why differentiating these is important – these require either intravenous chemotherapy (COP or CHOP protocols) or oral lomustine (CCNU).
Vomiting can be the presenting sign of many different problems in cats, so signalment, history and clinical examination should all be taken into account before formulating a diagnostic plan. Initial assessment of acute vomiting should focus on identifying unstable patients requiring emergency treatment and and/or immediate surgical intervention, and (for both acute and chronic vomiting) should also consider the need for nutritional support before more extensive diagnostic tests are performed. In cats with chronic vomiting, diagnosis can usually be at a slower pace, and diet or symptomatic drug trials can be performed before advanced diagnostic testing. Supportive treatment is part of managing most etiologies that cause vomiting, but clinicians must make an effort to identify specific causes to be able to perform the appropriate diagnostic tests in order to construct a targeted and effective treatment plan.
Ivan Montanes-Sancho
Dr. Sancho graduated from Zaragoza in 2016 and worked in small animal first opinion practice before undertaking a rotating internship at the Autonomous University of Barcelona Read more
Silke Salavati
Dr. Salavati graduated from the Justus Liebig University Giessen in Germany, and subsequently become a ECVIM diplomate Read more
Insect contamination of petfoods is a potential problem in tropical countries; this paper offers an overview of the situation and how the risks can be minimized
Mistakes happen to all of us in veterinary practice; this article looks at how different people react in different ways when things go wrong and – importantly – discusses how we can best cope with mistakes.
Cats are living longer and better lives; how can we ensure that the healthcare we offer them is optimal? This article offers some hints.
OA in cats remains underdiagnosed and undertreated, despite its widespread prevalence; this paper looks at how we can surmount the challenge of early diagnosis, leading to better treatment interventions.
