Veterinary internships in the USA
Thinking about doing an internship in the USA? This article delivers a brief guide to the advantages and disadvantages as to what one can offer.
Issue number 32.3 Other Scientific
Published 21/12/2022
Also available in Français , Deutsch , Italiano , Português and Español
Feline pyothorax is a potentially fatal condition that requires prompt recognition and adequate treatment in order to ensure a good outcome for the patient, as Chiara Valtolina describes.

Pyothorax is characterized by the accumulation of septic exudate in the pleural space, and can be life-threatening if not rapidly recognized and treated.
Affected cats present with a variety of clinical signs; these can range from tachypnea, anorexia, lethargy and fever to severe dyspnea and cardiovascular collapse.
Emergency medical stabilization of the patient is paramount, requiring oxygen supplementation, thoracocentesis, fluid therapy and broad-spectrum antibiotics.
Small-bore chest drains that allow thoracic drainage and lavage, along with broad-spectrum antibiotics, are the mainstay of treatment for most cases.
Pyothorax in the cat is characterized by the accumulation of septic purulent fluid within the pleural space. As the initial clinical signs of the condition are frequently nonspecific, diagnosis and/or presentation to the veterinary clinic is often delayed, but once the signs of dyspnea and cardiovascular compromise become more manifest, affected cats are often presented as an emergency. Prompt recognition of the problem and adequate treatment are mandatory to ensure a good outcome for this life-threatening condition 1,2,3,4,5. This article will review the available literature on pyothorax and discuss the recommended therapeutic options for treating affected cats.
The pleural space is lined by the parietal and visceral pleura and contains a minimal amount of fluid to prevent friction during the normal breathing cycle. In pyothorax, bacteria can enter the pleural space by a number of different routes (damage anywhere along the respiratory tract, via oropharyngeal contamination, or via the esophagus), initiating an inflammatory septic process. The presence of bacteria causes release of inflammatory cytokines and vasoactive mediators that alter local capillary permeability and lymphatic flow; this causes a thickening of the pleura, which facilitates development and accumulation of exudate within the pleural cavity. Systemic signs of generalized inflammation, sepsis, fever and general malaise also develop 1,6.
Due to the time lapse between the initial development and the actual diagnosis, the underlying reason for the pyothorax may not be determined in many cases, and indeed the principal cause is currently unknown. It is possible that different factors may play a role in the development of pyothorax. However, it is suggested that the two most likely causes are penetrating thoracic wounds secondary to a cat scratch or bite 1,4,5,6,7,8, and parapneumonic spread of oropharyngeal bacteria in animals with chronic upper airway infection 2,4,6, The cat scratch/bite theory has until recently been widely accepted as the most likely etiology, an idea supported by a retrospective study that noted external scratch or bite wounds had been reported in 14-40% of cases 7. Cats living in a multi-cat household were also found to be 3.8 times more likely to develop the condition, emphasizing the idea that pyothorax is secondary to fight wounds from inter-cat aggression 7. Further support for this theory came with the idea that pyothorax could have a seasonal predisposition, with more cats being presented during late summer/fall when aggression and fighting between cats for territory and mating are perhaps more likely, although the above study noted that outdoor access and gender were not risk factors for the development of pyothorax 7. The parapneumonic spread of oropharyngeal bacteria has been suggested as the most common route of infection of feline pyothorax in another retrospective study 8, a theory supported by necropsy findings from cats that died of pyothorax which noted numerous abscess formation in the lungs, suggestive of parapneumonic spread. Animals living in multi-cat households are probably also predisposed to develop chronic upper airway infection and are more at risk for developing pyothorax 4,6.
Organisms from the feline oropharyngeal flora are commonly isolated from pyothorax cases, with bacteria such as Pasteurella multocida, Clostridium spp., Fusobacterium spp., Bacteroides spp., Actinomyces spp., Peptostreptococcus spp., and Prevotella spp. being most commonly identified 2,6,9. Nocardia and Actinomyces spp. are also frequently isolated, and in fact polymicrobial infections with both obligate anaerobic and facultative aerobic bacteria are commonly found on culture 4,6,9.
Although cats of all ages can be affected, pyothorax is more commonly found in younger and middle-aged animals, with a mean age at diagnosis between 3 and 6 years 2,3,4,6,7. Younger animals seem to be more affected due to their behavior and the tendency to fight and explore outdoors when compared to older animals. Male animals seem to be over-represented in some studies, although no sex predisposition has been found to be statistically significant 7,8. In addition there does not seem to be a breed predisposition 6,7.
The duration of clinical signs prior to diagnosis can be from days to weeks 4,6. Non-specific clinical signs include pyrexia, lethargy, anorexia and weight loss 5,7. In one study only half of the cats with pyothorax presented with pyrexia, demonstrating that the absence of fever, especially in this species, does not exclude the presence of pyothorax 3,5,8 – and indeed cats that also have cardiovascular compromise (see below) often present with hypothermia 7. The most common clinical sign is dyspnea due to the presence of pleural effusion (which is often bilateral); affected cats often present with rapid, shallow respiration, indicative of a restrictive respiratory pattern. Other common signs are tachypnea and muffled heart sounds, but coughing is not usually an indicator of pleural effusion in cats 4,5,6,7. One retrospective study noted that hypersalivation was not uncommon; this may be caused by difficulty swallowing due to the presence of a large volume of pleural effusion 7.
Cardiovascular compromise can be present in the form of distributive shock due to the release of inflammatory cytokines and bacterial toxins from the pyothorax. Cats suffering from distributive shock secondary to sepsis present with pale mucous membranes, hypothermia, tachycardia or bradycardia and hypotension. One study reported that cats which did not survive pyothorax had a significantly lower heart rate and were often hypothermic compared to cats that survived 7.
A presumptive diagnosis of pyothorax can be made based on the clinical presentation (lethargy, depression, fever, anorexia, restrictive dyspnea and pleural effusion) and supporting clinical history (Box 1). The definitive diagnosis is made by combining macroscopic assessment of the pleural effusion with a cytological and cultural evaluation 4,6. Macroscopic evaluation of the effusion often reveals a malodourous opaque, cloudy, sero-hemorrhagic fluid, often containing white/yellowish floccules (Figure 1). Final confirmation is via cytological evaluation of the fluid, which will reveal polymorphic inflammatory cells, predominantly degenerate neutrophils with intracellular bacteria (Figure 2). Multiple bacterial species are often recognized on cytology, although if a patient has been pre-treated with antibiotics before diagnosis intracellular bacteria may not be seen; because of this, the effusion should always be cultured 4,6,7,9.
Box 1. Diagnostic pointers for feline pyothorax.
|

Figure 1. Septic pleural exudate will appear turbid and flocculent on macroscopic examination.
© Chiara Valtolina

Figure 2. A direct smear of septic pleural exudate under the microscope (x100 magnification); note the numerous degenerate neutrophils and macrophages with intracellular bacteria.
© Chiara Valtolina
The hematological and biochemical abnormalities in feline pyothorax are often non-specific and reflect the presence of the underlying inflammatory septic process and the patient’s overall clinical compromise. The most predominant hematological abnormality is a neutrophilic leukocytosis with or without a left shift, which occurs in 36-73% of cats 5,6,7. A mild normochromic anemia is also often reported. The most common biochemical abnormalities are hypoalbuminemia, hyperglobulinemia, hypo- or hyperglycemia, serum electrolyte imbalances and a mild elevations in serum liver enzymes 5,7.
Thoracic radiographs are often performed to evaluate the degree of pleural effusion, determine unilateral versus bilateral involvement, and to highlight any possible underlying cause, such as pulmonary or mediastinal masses and pneumonia 4,6. Radiographs are also useful to evaluate correct placement of a thoracostomy tube, as well as monitoring the effectiveness of the chosen therapy. Ultrasound can estimate the degree of effusion, determine the echogenicity of the fluid (often hyperechoic for pyothorax) and facilitate thoracic drainage 4,6,10. Moreover, pulmonary abscesses, intrathoracic masses and foreign bodies can also be detected with this modality 11.
More advanced diagnostics, such as computed tomography (CT) are often not performed as an initial diagnostic for every patient; apart from the costs, general anesthesia is required, which implies that the patient should have a stable cardiovascular system. However CT is often suggested when there is no improvement with medical therapy alone, or when there is a suspicion of pulmonary disease on thoracic radiography and ultrasound 1,4,6,12.
Cats with a suspected pyothorax should be approached as any other emergency patient, with a focused evaluation of the major body systems (respiratory, cardiovascular and neurological) which will allow the clinician to both quickly determine the degree of respiratory and cardiovascular compromise and to start prompt stabilization measures.
Dyspneic patients should be offered oxygen therapy using whichever technique is readily available and which causes the least stress (e.g., flow-by, oxygen cage); such animals may benefit from a few minutes of this “hands off” approach, which also permits evaluation of the respiratory pattern and allows the cat to calm down. Severely distressed dyspneic patients may also benefit from administration of a mild sedative with minimal cardio-depressant effects (e.g., butorphanol at 0.2 mg/kg IM); this allows them to relax and removes the anxiety associated with the dyspnea. In any distressed patient thoracocentesis, placement of a peripheral catheter and any other stressful procedure should be performed only after administration of a sedative.
Physical examination will usually reveal signs linked to the restrictive dyspnea; decreased or absent lung sounds ventrally (uni- or bilaterally) on thoracic auscultation can be enough to diagnose the presence of pleural effusion. Thoracic radiography should not be performed in a dyspneic cat, as restraint can be fatal for the most compromised patient. The use of point of care ultrasound (POCUS) for rapid evaluation without causing too much stress to the patient has gained popularity in the emergency and critical care setting, and is a rapid and safer diagnostics procedure to detect the presence of pleural effusion and lung pathology 10,11.
Chiara Valtolina
If the cat’s stress levels allow, intravenous access should be gained to allow administration of fluid therapy for cardiovascular stabilization and medication, but if the patient is too dyspneic to allow placement of an IV line, thoracocentesis should be performed first.
Thoracocentesis should be both diagnostic and therapeutic. Depending on the location of the pleural effusion, either unilateral or bilateral thoracocentesis should be performed. The presence of fibrin and purulent exudate can make it difficult to completely remove the effusion, and an adequate size needle or butterfly is recommended. Collected fluid should always be sent for cytologic evaluation and culture.
Medical and surgical options are available for treating feline pyothorax, although in the available literature there is no agreement as to the optimal treatment 4,6. Prospective studies evaluating and comparing the efficacy of the two options are lacking; furthermore, the literature often reports and compares only limited number of cases, making it even more difficult to draw any meaningful conclusion. Despite this, the general consensus is that medical treatment should be considered, at least initially, as the mainstay of therapy 1,4,5,6,7. In a recent retrospective study, 85% of cats (47 animals) with pyothorax in the review were treated medically, with only 5 cats not responding and requiring surgical intervention 5.
However, thoracocentesis alone, even if repeated multiple times, is not adequate as a stand-alone treatment for pyothorax 1,4,5,6. Once the patient has a stable cardiovascular profile and any electrolyte abnormalities have been corrected, one or more chest drains must be placed – bilateral drains are often required. This should be done under sedation, and will allow drainage and lavage of the thorax, along with antimicrobial therapy. Intravenous antimicrobials should be started immediately whilst awaiting the results of fluid culture and susceptibility; a broad-spectrum antibiotic is usually chosen, with amoxicillin/clavulanic acid being the drug of choice as it also has some efficacy against Actinomyces spp. and anaerobes. Depending on the antibiotic regulations of the country, a fluoroquinolone antibiotic is also often suggested to improved gram-negative coverage 4,6,13. Antibiotic treatment should then be adjusted based on the culture results and must be continued for at least for 3-4 weeks beyond resolution of the clinical signs.
One or more indwelling thoracostomy tubes should be placed to drain and lavage the pleural space. Previously large-bore (14-16 Fr) chest drains were recommended for the exudative and fibrinous effusion seen with pyothorax 14, but these require general anesthesia for placement and cause pain and discomfort for the patient. More recently a small-bore chest drain (SBTT) (10-14 G) placed using a modified Seldinger’s technique is the preferred choice in both human and veterinary medicine. SBTTs are easy to place using only mild sedation, are well tolerated by the patient, and have minimal complication rates 15,16 – the most commonly reported complication being kinking or malposition of the tube. An on-line video demonstrating the procedure is available *.
* https://www.milainternational.com/index.php/videos_articles/
Chiara Valtolina
Note that it is important to determine how far the chest drain should be positioned within the thoracic cavity by pre-measuring; this should be from the insertion site (usually around the 8-9th intercostal space) to the cranio-ventral aspect of the chest cavity (2nd-3rd rib), and to ensure that all the side holes of the chest drain are within the thorax. The patient should be kept in sternal recumbency during placement to allow it to breath more comfortably, especially if only mildly sedated. The target area should be clipped, and asepsis maintained during the placement.
Drains should be placed unilaterally or bilaterally, depending on the location of the pleural effusion (Figure 3). The chest should be drained as soon as the tube is inserted, as it is not uncommon for placement to cause a mild iatrogenic pneumothorax. Once the chest is drained, the SBTT is secured to the skin; if the animal is stable, a thoracic radiograph should then be performed to evaluate the position of the drain. Radiography may show that that catheter is not in the intended position within the pleural space, but if it is working adequately and no other problems are noted, it should not be replaced (Figure 4). It may be beneficial to adjust the position of the animal during drainage to optimize aspiration of the effusion, and adequate analgesia is mandatory. In critical cases, the use of an opioid (methadone 0.2 mg/kg IV or IM every 4 hours or buprenorphine 20 µg/kg IV every 6 hours, possibly with ketamine at 30-50 µg/kg/min as a constant rate infusion (CRI) should be preferred over the administration of a non-steroidal anti-inflammatory analgesic. The latter option could however be considered for a normovolemic, well-hydrated and stable patient.
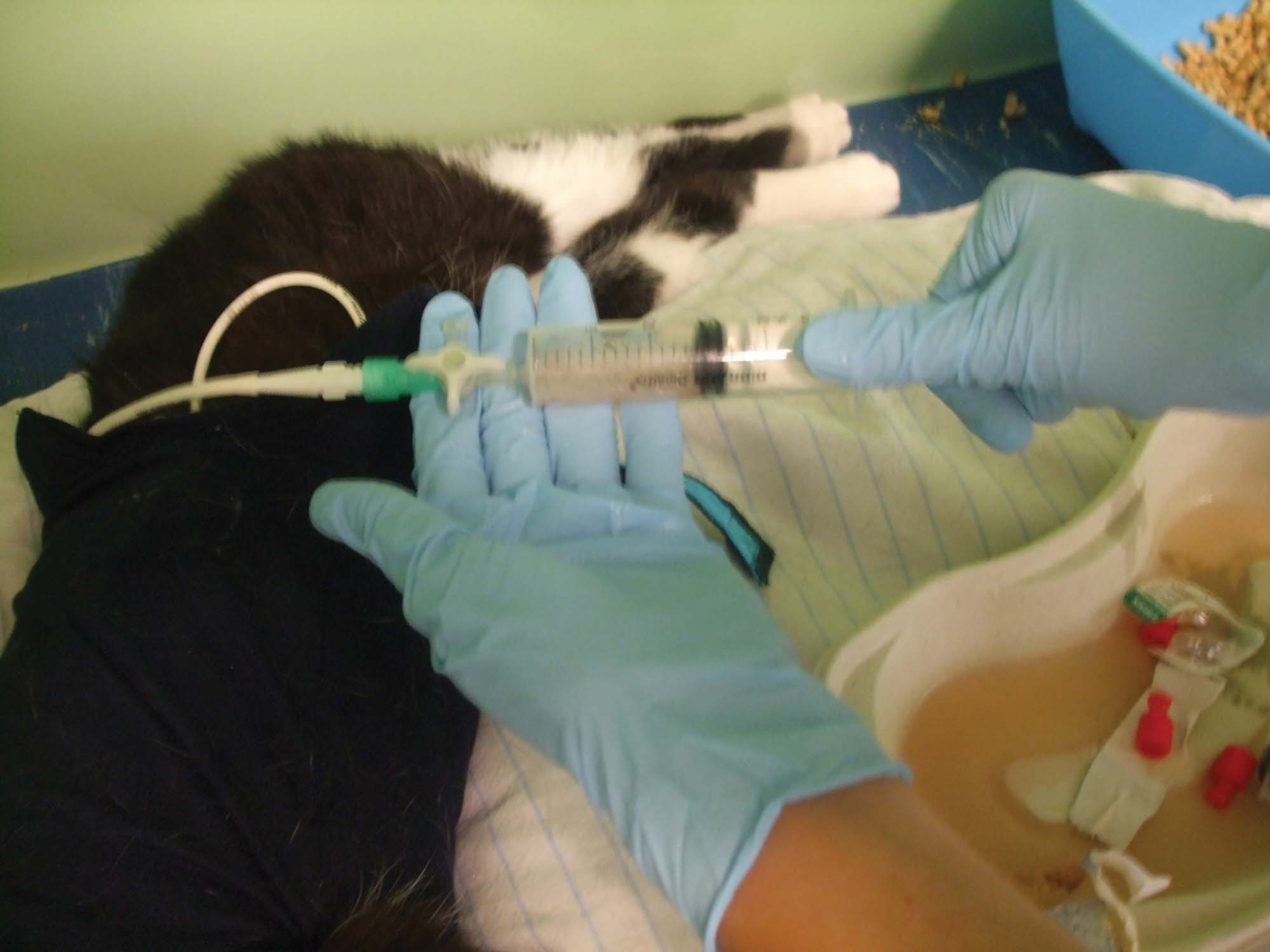 |
| a |
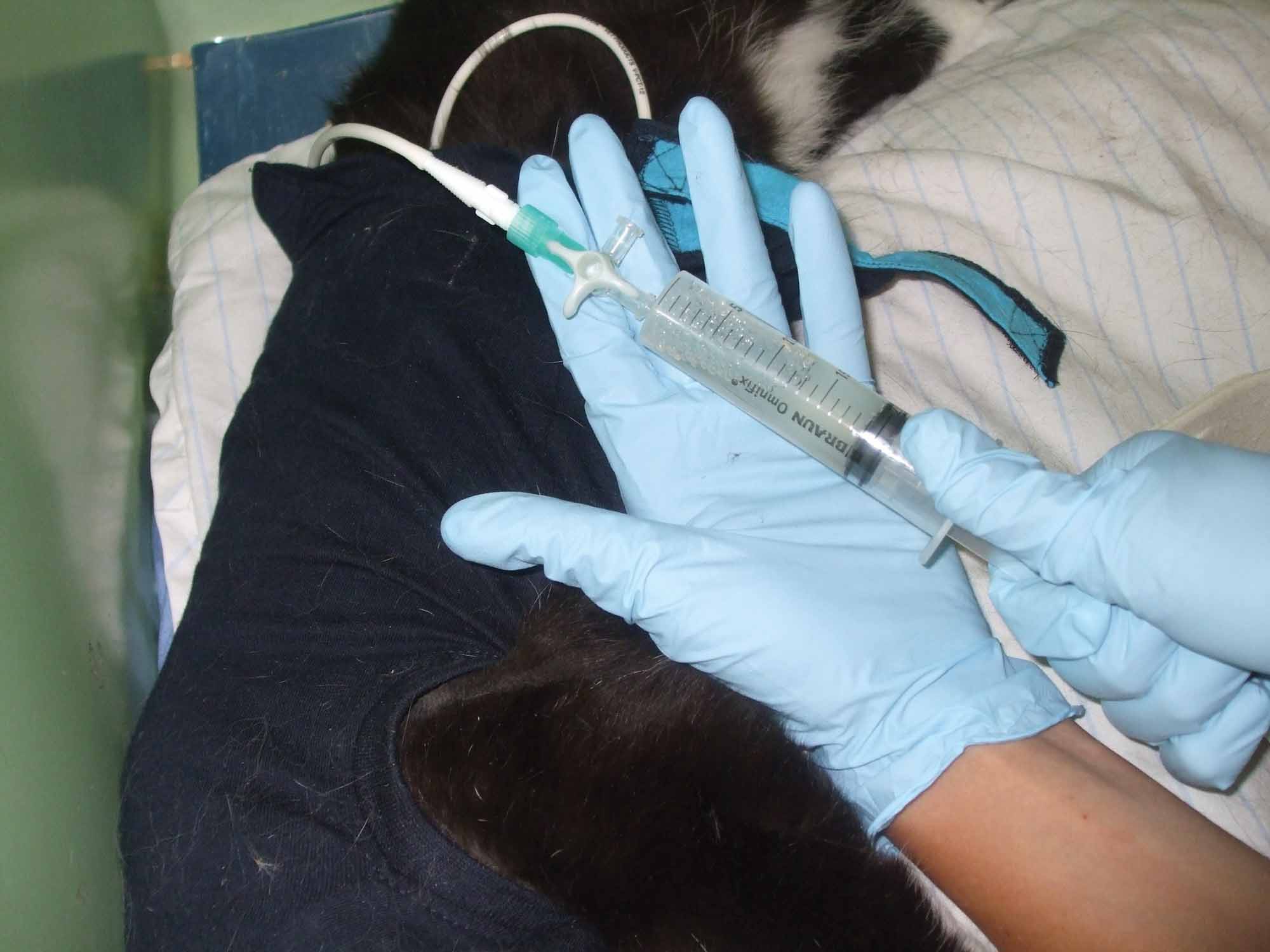 |
| b |
Figure 3. Thoracic drainage and lavage in a cat with pyothorax. (a) Once the syringe and three-way tap are attached to the drain, the pleural effusion is aspirated. (b) Lavage is then performed using a warmed isotonic crystalloid; lavage should be repeated until the drained fluid is clear.
© Chiara Valtolina
Various authors have stressed the importance of thoracic lavage to facilitate the removal of the purulent, fibrinous exudate, bacteria and inflammatory mediators 1,46. In a small retrospective study in dogs the benefit of pleural lavage over drainage alone was reported 17. A warm sterile isotonic crystalloid infusion should be used for lavage, initially at 5 mL/kg to ensure the patient tolerates the procedure well and to verify that the fluid can be withdrawn without any problem afterwards. If the procedure is well tolerated and the majority of the lavage fluid is retrieved, then the volume can be increased (10-15 mL/kg), with lavage repeated until the drained fluid is clear. Lavage should be performed as frequently as possible, especially when the cat is first hospitalized, and at least 4-6 times per day. Before performing lavage, it is important to first drain the chest to keep track of fluid production, and it is essential to record how much fluid is infused and retrieved every time and from which side of the chest. The addition of heparin or antibiotics to the lavage fluid is not recommended.
The chest drain should be kept in place until fluid production starts to decrease to around 2-3 mL/kg/day and the patent shows signs of clinical improvement. The chest drain itself and the pleuritis will cause some fluid production. Before removing the drain, it is very important to repeat cytologic evaluation of the fluid to check for the presence of degenerated neutrophils and intracellular bacteria. In the author’s institution a negative cytology result along with fluid production < 2 mL/kg/day are the criteria used for removal of the chest drain. The patient is usually monitored for 24 hours after the removal of the drain/s, and thoracic radiographs are repeated before discharge to allow comparison at recheck, which should be scheduled over the following 2-3 weeks.

Figure 4. A lateral thoracic radiograph of a cat with bilateral pleural effusion. A right-sided SBTT has been placed around the 8th intercostal space, with the tip of the chest drain positioned in the cranio-ventral part of the chest.
© Utrecht University/All rights reserved
Surgery should be considered if there is an inadequate response to medical therapy after 2-7 days – i.e., insufficient clinical improvement, continued fever, continued presence of pleural fluid or if fluid remains turbid or flocculent, and/or detection of an underlying cause on diagnostic imaging (e.g., abscess, suspicion of a foreign body, loculated (compartmentalized) effusions, thickened pleura) 4,5,6,15. Surgery consists of an exploratory thoracotomy; this is either via a sternotomy (if the effusion or the lesions are bilateral) or via lateral thoracotomy on the affected side 15.
Overall, pyothorax cases can have a fairly good prognosis. A recent retrospective study 5 highlighted both good short- (14 days) and long-term (1 year) survival rates, with 72% and 68% cats being alive at the respective times. In addition, the recurrence rate was low, with only 2% of cases developing a relapse.
If adequately treated, pyothorax has a good prognosis, but it is important to initially discuss with the owners likely costs of hospitalization (which on average is 5-6 days) and treatment, and the possibility of relapse. Pyothorax cases should be treated as potentially life-threatening, and the clinician should be alert to the need for immediate and aggressive intervention to ensure the best possible outcome for such animals.
Barrs VR, Beatty JA. Feline pyothorax – new insights into an old problem: Part 2. Treatment recommendations and prophylaxis. Vet. J. 2009;179(2):171-178.
Barrs VR, Beatty JA. Feline pyothorax – new insights into an old problem: Part 1. Aetiopathogenesis and diagnostic investigation. Vet. J. 2009;179(2):163-170.
Demetriou JL, Foale RD, Ladlow J, et al. Canine and feline pyothorax: a retrospective study of 50 cases in the UK and Ireland. J. Small Anim. Pract. 2002;43(9):388-394.
Epstein SE, Balsa IM. Canine and feline exudative pleural diseases. Vet. Clin. North Am. Small Anim. Pract. 2020;50(2):467-487.
Krämer F, Rainer J, Bali MS. Short‐ and long‐term outcome in cats diagnosed with pyothorax: 47 cases (2009‐2018). J. Small Anim. Pract. 2021;62(8):669-676.
Stillion JR, Letendre J. A clinical review of the pathophysiology, diagnosis, and treatment of pyothorax in dogs and cats. J. Vet. Emerg. Crit. Care (San Antonio) 2015;25(1):113-129.
Waddell LS, Brady CA, Drobatz KJ. Risk factors, prognostic indicators, and outcome of pyothorax in cats: 80 cases (1986-1999). J. Am. Vet. Med. Assoc. 2002;221(6):819-824.
Barrs VR, Allan GS, Martin P, et al. Feline pyothorax: a retrospective study of 27 cases in Australia. J. Feline Med. Surg. 2005;7(4):211-222.
Walker AL, Jang SS, Hirsh DC. Bacteria associated with pyothorax of dogs and cats: 98 cases (1989-1998). J. Am. Vet. Med. Assoc. 2000;216(3):359-363.
Lisciandro GR. TFAST Accurate diagnosis of pleural and pericardial effusion, caudal vena cava in dogs and cats. Vet. Clin. North Am. Small Anim. Pract. 2021;51(6):1169-1182.
Lisciandro GR, Lisciandro SC. Lung ultrasound fundamentals, “wet versus dry” lung, signs of consolidation in dogs and cats. Vet. Clin. North Am. Small Anim. Pract. 2021;51(6):1125-1140.
Eiras-Diaz A, Frykfors von Hekkel A, Hanot E, et al. CT findings, management and short-term outcome of dogs with pyothorax: 101 cases (2010 to 2019). J. Small Anim. Pract. 2021;62(11):959-966.
Lappin MR, Blondeau J, Boothe D, et al. Antimicrobial Use Guidelines for Treatment of Respiratory Tract Disease in Dogs and Cats: Antimicrobial Guidelines Working Group of the International Society for Companion Animal Infectious Diseases. J. Vet. Int. Med. 2017;31(2):279-294.
Farrell K, Epstein S. Pyothorax. In; Drobatz KJ, Hopper K, Rozanski EA, et al (eds) Textbook of Small Animal Emergency Medicine. Newark: John Wiley & Sons, Inc; 2018;291-296.
Del Magno S, Foglia A, Golinelli L, et al. The use of small-bore wire-guided chest drains for the management of feline pyothorax: a retrospective case series. Open Vet. J. (Tripoli, Libya) 2021;10(4):443-451.
Valtolina C, Adamantos S. Evaluation of small-bore wire-guided chest drains for management of pleural space disease. J. Small Anim. Pract. 2009;50(6):290-297.
Boothe HW, Howe LM, Boothe DM, et al. Evaluation of outcomes in dogs treated for pyothorax: 46 cases (1983-2001). J. Am. Vet. Med. Assoc. 2010;236(6):657-663.
Chiara Valtolina
Dr. Valtolina graduated in 2000 from the Faculty of Veterinary Medicine in Milan Read more
Thinking about doing an internship in the USA? This article delivers a brief guide to the advantages and disadvantages as to what one can offer.
Prescribing IV fluids for cats is not as straightforward as it may first appear; this paper offers a comprehensive overview of current knowledge.
Sepsis is a medical emergency in the dog; rapid identification and proactive intervention are essential for a positive outcome.
Acute heart failure in dogs carries a serious risk of death, and optimizing diagnosis and treatment is paramount, as discussed by Luca Ferasin.
